Document Type : Mini-Review Article
Authors
Department of Biochemistry, Dr DYPatil Medical College, Hospital and Research Center, Dr. D.Y.Patil Vidyapeeth, Pimpri, Maharashtra
Abstract
The most prevalent hemoglobinopathy is sickle cell disease, which is followed by β-thalassemia and thalassemia syndrome. The Mediterranean region, Middle East, Indian subcontinent, South east Asia, and the northern coast of Africa have been found to have the highest prevalence of both diseases. The majority of affected youngsters in many of these regions pass away before turning five without ever receiving a diagnosis. Our understanding of hemoglobinopathies' clinical and therapeutic features as well as their laboratory diagnosis has increased as a result of several study articles. Erythrocyte index, hemoglobin analysis by HPLC and capillary electrophoresis, and recently employed next-generation sequencing (NGS) to screen for thalassemia are the main biochemical and molecular procedures used in clinical laboratories. Preventive treatment and genetic counselling can be performed when SCD is detected and diagnosed early, which helps to lower mortality and minimize sequelae. Current diagnosis procedures are laboratory-based and call for expensive diagnostic tools and qualified employees. India has a vast population, making it impossible for them to pay for traditional diagnostics. The primary benefit of POCT is its low cost, and its effect on hemoglobinopathies may ultimately contribute to the improvement of India's health situation and other relevant fields.
Graphical Abstract
Keywords
Introduction
Red blood cells (erythrocytes) contain the tetrameric iron protein hemoglobin, which serves as an effective means of transporting oxygen from the lungs to the tissues of vertebrates. It originates in the bone marrow and is transformed into red blood cells. Two different types of polypeptide chains make up each hemoglobin molecule. The common peptide subunits are alpha, beta, and gamma they combine to produce several variations, such as Adult Hb (HbA), which includes (α2β2). The interaction of several structural differences in hemoglobin and thalassemia variants results in a broad spectrum of diseases with varying clinical severity. The most significant groups for which genetic counselling is suggested with the potential for prenatal diagnosis include thalassemia major (TM), sickle cell syndromes, HbE/thalassemia combinations, and -thalassemia syndromes, including the deadly Hb Bart's and HbH Hydrops Fetalis Syndromes [1]. Worldwide inherited conditions known as hemoglobinopathies include diseases like sickle cell anemia, hemolytic anemia, erythrocytosis, and polycythemia caused by structural changes to hemoglobin or changes in the synthesis of α- or β-globin (α- and β-thalassemia syndromes, respectively).
The pathogenesis of Sickle Cell Disease is due to polymerization of sickle hemoglobin. Hence, it shows lower affinity towards oxygen. Red blood cells' structure and cell membrane properties are altered as a result of polymerization, which causes the cells to become dehydrated and form chains. Hemolytic anemia is caused by increased polymerization, which also shortens the lifespan of cells and causes an increase in cell death.
The most prevalent kind of anemia is sickle cell, which affects several organ systems and manifests as both acute and chronic problems when fetal hemoglobin (HbF) levels fall to adult levels by five to six months of age [2]. The most prevalent hemoglobin disorder is sickle cell anemia, which is followed by thalassemia syndrome, which includes β-thalassemia with hemoglobin E and hemoglobin S (HbE/-thalassemia, HbS/thalassemia, Hb-Bart edoema, and HbH-disease) [3,4]. Hemoglobin disorders are a significant public health issue in India. Hemoglobinopathies can be prevented to some extent with prenatal diagnosis and counselling.
Materials and Methods
Information was gathered from the most significant databases like Google Scholar, Scopus, and PubMed. Likewise, it was collected from government documents, summaries of electronic conferences, and online archives of English-language publications that have been recognized as reliable sources.
The Mediterranean regions, the Middle East, the Indian subcontinent, Southeast Asia, and the northern African coasts are where the SCD and thalassemia are very common. Due to migration and mutations, there are also hemoglobin anomalies in northern European and American countries [5]. In India, hemoglobinopathies are the main contributor of nonferrous anemia. Indian tribal groups are particularly challenged by the topical enzyme-deficient dehydrogenase (G6PD) produced by hemoglobin S, D, E, -thalassemia, and glucose-6-phosphate.Tribal Health Expert Report for the Ministry of Health and Family Welfare shows that both adults and newborns in the Galacian community suffer from sickle cell anemia. Garasia and Bhil strains both exhibit thalassemia and a G6PD deficiency [6, 7]. We know more regarding the clinical and therapeutic features of hemoglobinopathies and their laboratory diagnostics due to a number of recently published articles. In locations where there is a high frequency of these diseases, many diagnostic tools have been established for screening programs. India has a broad range of hemoglobinopathies. A number of laboratory tests are necessary for hemoglobinopathies to be diagnosed. The red blood cell index, hemoglobin analysis by HPLC and capillary electrophoresis, and the recently utilized next-generation sequencing (NGS) to screen for thalassemia are the principal biochemical and molecular procedures used in clinical laboratories [8]. The sickle cell anemia (HbSS) mutation occurs in homozygosity with various illnesses that are categorized as sickle cell disease (SCD). The condition severity varies depending on the α-globin or β-globin variant, and it can potentially be life-threatening [9].
Hemoglobinopathy screening tool
The risk of invasive Streptococcus pneumoniae (pneumococcal) infection is lower when hemoglobinopathy is detected early [10]. In addition, early diagnosis of sickle cell disease allows medical professionals to inform the family about their SCD and offer proactive advice as part of the child's general treatment before symptoms and difficulties arise. Each screening program has a different approach to neonatal screening and diagnosis. Some techniques identify a wide range of variables, while others identify simply the most prevalent variables. While certain processes are intensive, others are automated. Applications based on DNA used in diagnosis call for smart gadgets. Isoelectric focusing (IEF) and high-performance liquid chromatography (HPLC) are now frequently combined as the main analytical techniques. To confirm samples with negative results, additional techniques including electrophoresis, HPLC, antibody testing, or DNA-based analysis are used [11, 12].
Isoelectric focusing (IEF)
The IEF test is the most effective way to check for hemoglobinopathies in infants. IEF is a frequently utilized, inexpensive, and extremely sensitive approach. IEF divides hemoglobin species with extremely high resolution based on their isoelectric points on gel media. The Hb variant migrates along the pH gradient until it reaches a net charge of zero. Alkaline electrophoresis and IEF both use a similar hemoglobin migration order. By separating HbS and HbD, HbG and HbC, HbE and HbO, and HbA and HbF, a distinct separation is achieved [13].
Compared to conventional electrophoresis, this technique offers more accuracy and precision, is time-demanding and requires effort yet offers high pressure and a good solution. To confirm or distinguish hemoglobin variations (bands other than HbF or HbA), further techniques are needed. The IEF can also be used to determine rapid movements (Hb Bart's, HbH) [14, 15].
High performance liquid chromatography (HPLC)
Using a preprogrammed buffer gradient that raises the ionic strength depending on ionic interactions to the analytical cartridge, hemoglobin is separated from the analytical cartridge by cation-exchange HPLC [16]. More samples can be processed faster and with fewer technicians due to the HPLC's automation.
Pan India surveillance and the systematic review carried by Kharche K, Bhake over 14 studies across four regions revealed that hemoglobinopathies must be analyzed in microcytic hypochromic anemia with a hemolytic component that is resistant to treatment. The HPLC is likely the best analyzer for finding the rare hemoglobin variants that the electrophoresis was unable to find [16]. Hbs A, A2, F, S, C, and D Punjab and G Philadelphia usually remain different from each other. For analysis, a minimal sample volume of 5 ml is adequate. Pediatric sufferers will especially benefit from this. However, it has the disadvantage that quantification cannot be offered and that it is challenging to differentiate between HbH and HB Barts. With numerous variants exhibiting comparable retention time, hemoglobin A2 is identical to hemoglobin E and hemoglobin Lepore [17].
Cellulose acetate electrophoresis
The negative charge that hemoglobin receives under alkaline conditions is used in cellulose acetate electrophoresis, often known as alkaline electrophoresis. Using pH 8.4 buffer (borate TrisEDTA), hemoglobin was isolated from these samples by electrophoresis after being put to cellulose acetate agar gel. Hemoglobin exists in several different varieties, each of which differs from the others in unique ways [13]. With little preparation required, this technique permits the quick and accurate isolation of HbA, HbF, HbS, HbC, and other hemoglobin variations. Its sensitivity is constrained; however, certain types of hemoglobin have distinct structures but are electrophoretically related [15, 18].
Capillary zone electrophoresis
In an alkaline buffer (pH 9.4), charged molecules are separated by electrophoretic mobility. Depending on the electrolyte's pH and the in osmotic or electroosmotic flux, this separation occurs. Hemoglobin emerges from the capillary's anodic end in a specific zone and moves to the cathodic end when a hemolyzed sample is injected into the capillary's anodic end. Results are visually examined for anomalies and a distinction between healthy and diseased patterns is made. HbS can be identified from other types by isolating the hemoglobin fraction, making sickle cell anaemia easier to spot.
HbE and HbA2 can be clearly separated using capillary zone electrophoresis, which also makes it easier to find Hb Bart and HbH [19].
Molecular methods
Common methods include DNA sequencing, restriction fragment length polymorphism (RFLP), and allele discrimination using real-time PCR endpoint data. For all PCR-based studies, whole blood DNA extraction in a DBS filter paper matrix is necessary [20].
Assays based on the Polymerase Chain Reaction (PCR)
The polymerase chain reaction is one of the most sensitive diagnostic tools for prenatal and neonatal diseases. Using particular primers and specialized enzymes, genetic material is amplified into millions of copies. It can identify numerous well-known genes in a single tube or well-known single genes. In this technique, denaturation, annealing, and elongation are carried out repeatedly over the course of 20-40 heat cycles. Gel electrophoresis, sequencing, melting curve analysis, or keeping an eye on variations in fluorescence can all be used to determine the outcome. Each PCR-based molecular test has a unique setup and amplification/analysis phase, a one-way process, and is susceptible to contamination by amplicons that have been aerosolized [21].
RFLP or restriction fragment length polymorphism
To determine if a gene's alleles are normal or mutant, RFLP uses restriction enzymes. Gel electrophoresis can reveal the size difference between the DNA fragments after the restriction enzymes restrict (cut) the DNA. A technique for identifying sickle cell disease called restrictions fragment length polymorphism (RFLP) relies on restriction enzymes that eliminate the gene's recognition site in mutants. Ddel I is a different restriction enzyme that has been employed to recognize sickle cells. The restriction site of Ddel I, 5′-GTNAG-3′, is removed by the mutation that causes sickle cell anemia (SCA). Consequently, depending on whether the sickle cell anemia mutation is present, bands of varying lengths emerge [22].
Allelic discrimination using real-time PCR
At the final stage of PCR, Allelic Discrimination (AD) measures fluorescence to find mutations. The hydrolysis probe and forward and reverse primers covering the region of interest are used in this method. The expensive cost of the probes and the requirement for a real-time PCR apparatus are the method's drawbacks. The restricted number of filters in PCR machines prevents higher levels of multiplexing, even though AD is considered as a multiplexed assay [23].
DNA sequencing
Sanger sequencing, which provides precise sequence throughout the entire range of primers used, is the most thorough approach for mutation detection. The key benefit of this approach is the ability to detect SNPs as well as small insertions and deletions in the region of interest. To further enhance the identification and treatment of hemoglobinopathies, including thalassemia’s, certain research facilities are looking into the use of tandem mass spectrometry and whole-genome sequencing [24].
The need for point-of-care SCD diagnosis
The SCD prevalence is rising globally, and there is an urgent need for POC diagnostic tools that are accurate and affordable for SCD in areas with limited resources. The vast majority of affected little ones in many of these locations die unrecognized before turning five. The diagnostic procedures used today are laboratory-based, expensive, and labor-intensive. Traditional techniques demand costly machinery and qualified technicians.With just a few microliters of unprocessed whole blood, this novel test can easily and quickly diagnose sickle cell anemia or thalassemia at a cheap cost and with high accuracy [25].
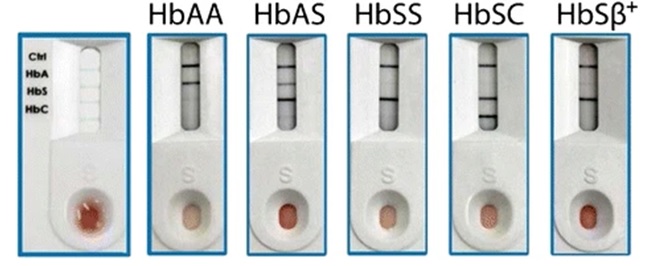
According to research done in Nigeria, Hemo TypeSC holds out a lot of promise, including high sensitivity, specificity, and low cost. This might aid in more accurate SCD diagnosis in India and the SSA majority. There are already a number of POCT devices on the market, each with pros and cons, but several of which have been demonstrated to be very good and useful in differentiating between various Hb phenotypes [26]. Sickle SCAN, another POCT that has been looked at earlier, also showed good specificity, sensitivity, and accuracy in HbSS and HbSC diagnosis [27]. Numerous POC test devices based on solubility test methodologies have been developed recently (Sickledex [Orthodiagnostic], Sicklecheck [TCS Biosciences]) and Polyclonal antibody-based capture immunoassays, HbS and HbA wicking through filter paper, and different red blood cell densities [28].
Lateral flow immunoassay
The Hemo TypeSC (Silver Lake Research Corporation) qualitative lateral flow immunoassay method uses monoclonal antibodies with very low detection limits for HbA, HbS, and the presence of HbC antigens. Because these antibodies are HbF blind, they can reliably identify neonates with high HbF and extremely low levels of HbA or HbS.
HemoTypeSC does not require any tools, power, or cooling. Test results are visually and qualitatively interpreted. Visual inspection can provide the test findings in 10 minutes [29, 30]. The low-cost, quick POC Hemo TypeSC HbS phenotyping equipment is best suited for remote locations where HbS is a serious public health issue and other facilities are not present. According to a study by Mukherjee and Roshan B. et al., Hemo TypeSC is a very accurate way to determine whether a parent is a carrier and can help parents make decisions that may lessen the likelihood that their child would be born with SCD in the future across India. Therefore, broad newborn and community screening in India for SCD and the sickle cell trait is appropriate using the Hemo TypeSC.
Density-based separation
In Density-Based separation, using an aqueous multiphase system based on cell densitometry (AMPS), can identify red blood cells with a crescent shape.
The biphasic AMPS system can discriminate between dense sickle cells and normal cells with 90% sensitivity and 97% specificity [31]. Based on the high density of sickle cells in hypoxic instances, a wearable smartphone platform was developed to identify sickle cells in blood samples less than 1 L. To dry and inflame the cells, sodium metabisulfite was combined with blood samples. Using a light-emitting diode (LED), the sample was illuminated. An optical lens is then used to magnify the image. The RBC maglev train takes pictures of the cells using permanent magnets. Benefits of using a maglev include avoiding the need for a microscope and centrifugation [32]. Quality control is very important aspect in POCT, especially preanalytical work like documentation, storage, etc. QC should be performed on every newly opened strip bottle. Initially all control and test strips bottles should be documented with opening date and expire date. The QC program should be followed as per manufacturer recommendation.
Conclusion
After Nigeria, India is considered to have the second highest SCD burden. Early supportive and preventive treatment and genetic counselling are essential for reducing mortality and minimizing morbidity associated with SCD. India has a vast population and cannot pay the price of conventional tests, thus the POC diagnostic equipment's low cost is a significant advantage. Chhattisgarh, West Bengal, Uttar Pradesh, Maharashtra, Madhya Pradesh, Jharkhand, Gujarat, Orissa, Kerala, and Rajasthan are Indian states with high rates of SCD. These are collectively referred to as sickle cell zones. It is easier for POCT to undertake timely investigation and detection in remote places since there is inadequate healthcare infrastructure and expertise among healthcare professionals. Over the next three and a half years, the government will screen 70 million people for the inherited blood disorder sickle cell anemia. Therefore, the hemoglobinopathies identification by POCT will undoubtedly boost the position of the medical and allied industries.
Acknowledgements
The authors would like to thank the staff and anesthesiology team at Maternity Teaching Hospital for their help and support during the research study. They appreciate their cooperation which was invaluable in enabling the author to conduct the research, and also, they are grateful to the women who participated in the study.
Disclosure Statement
No potential conflict of interest was reported by the authors.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Authors' Contributions
All authors contributed to data analysis, drafting, and revising of the paper and agreed to be responsible for all the aspects of this work.
ORCID
Anita Deepak Deshmukh
https://orcid.org/0000-0002-9778-8118
Umesh Kisanrao More
https://orcid.org/0000-0003-2005-1668
HOW TO CITE THIS ARTICLE
Sarita Anil Shinde, Anita Deepak Deshmukh, Umesh Kisanrao More*. Neonatal Sickle Cell Disease Assessment in Rural Indian Community: Demand for Point of Care Testing (POCT). J. Med. Chem. Sci., 2024, 7(1) 1-8.