Document Type : Original Article
Authors
- Clarisa Finanda 1
- Evelyn Komaratih 1
- Wimbo Sasono 1
- Nurwasis Nurwasis 1
- Citra Dewi Maharani 1
- Kautsar Abiyoga 1
- Djoko Legowo 2
- Hari Basuki Notobroto 3
- Willy Sandhika 4
1 Department of Ophthalmology, Faculty of Medicine, Universitas Airlangga, Dr. Soetomo General Academic Hospital, Surabaya, Indonesia
2 Department of Pathology Anatomy, Faculty of Veterinary, Universitas Airlangga, Surabaya, Indonesia
3 Department of Epidemiology, Biostatistics, Population Studies and Health Promotion, Faculty of Public Health, Universitas Airlangga, Surabaya, Indonesia
4 Department of Pathology Anatomy, Faculty of Medicine, Universitas Airlangga, Dr. Soetomo General Academic Hospital, Surabaya, Indonesia
Abstract
Patients with open globe injury (OGI) are higher risk of developing secondary retinal detachment. This study examines the effectiveness and safety of a single intravitreal of triamcinolone acetonide or bevacizumab to suppress the expression of VEGF and TNF-α in OGI rabbit eye. OGI is generated by inflicting a 5-mm posterior ocular lesion with blade no. 11 and 6-mm inferior the limbus in the supratemporal quadrant. Intravitreal injection with triamcinolone acetonide (TA) 4 mg/0.1 mL or bevacizumab 1.25 mg/50 µL 3 days (T+3 and B+3 groups) and 7 days (T+7 and B+7 groups) after OGI (n = 6). Another OGI group without treatment served as the positive control (CtrP) while five left eyes were randomly selected from the treated group to serve as the negative control (CtrN). Pretreatment and decapitation, a comprehensive anterior segment, posterior segment, and intraocular pressure (IOP) examination was conducted on days 3, 7, and 21. On day 21, the eyes were enucleated, and VEGF and TNF-α expression were assessed by immunohistochemistry. VEGF and TNF-α overexpression were observed in rabbit eyes after OGI (9.64 ± 1,646, p = 0.0002 and 10.44 ± 1,381, and p = 0.0553, respectively). Treatments significantly reduced these pro-inflammatory proteins compared with eyes in the OGI group. Posthoc Tukey’s multiple comparison test showed that TA gave better downregulation than that of bevacizumab. Clinical examination found no significant rise of IOP at the end of the study. The current study exhibits TA intravitreal injection after OGI significantly attenuated the overexpression of pro-inflammatory protein with a good safety profile.
Graphical Abstract
Keywords
Introduction
Ocular trauma is an under-recognized cause of unilateral blindness affecting approximately 19 million people around the globe [1]. Children, young adults, and the elderly were disproportionately affected, whereas men in their productive years had the most potential [2, 3]. Ocular trauma is classified as open globe injury (OGI) or close globe injury (CGI) based on the structure of the globe that has been damaged. OGI affects the entire thickness of the anatomical area, either the cornea or sclera [4, 5]. Severe OGI is expected to occur roughly 203,000 times annually globally, with hospitality incidence rates ranging from 27.7 to 35 per 100,000 people in China and Taiwan studies. Males and working-age groups have the highest risk [6]. This occurrence may result in economic strain [7]. OGI or severe CGI typically have a terrible prognosis, with proliferative vitreoretinopathy (PVR) developing as a significant consequence, especially if retinal detachment (RD) or vitreous hemorrhage (VH) is subsequent [7, 8]. PVR is triggered by excessive healing of a vitreoretinal wound caused by a retinal tear and an inflammatory response to the entrance of retinal pigment epithelial (RPE) into the vitreous cavity [8-10]. During wound healing, there are three concurrent and continuous phases: inflammatory, proliferative, and remodelling [10, 11]. The incidence rate of PVR in the general population with rhegmatogenous retinal detachment (RRD) ranges from 5 to 10%. However, the incidence rate in patients with OGI might reach 50%, leading to poor visual prognosis [12-15]. Retinal surgery has evolved significantly with successful anatomical attachment over the past decade [16]. To prevent the PVR development, research into pharmacological therapies and adjuvant surgical procedures for OGI is essential. Several pharmacological therapies, including anti-inflammatory, anti-proliferative, antineoplastic, antigrowth factor, and antioxidant agents, have been studied to treat PVR [17-19]. Triamcinolone acetonide (TA) is a synthetic glucocorticoid which possessed anti-inflammatory and fibroblast suppression properties [20]. It is one of the most frequently used steroids in ocular therapy [21]. Side effects had been detected in intravitreal steroid application, such as IOP spike [22], cataract [23, 24], and sterile endophthalmitis [25]. Therefore, alternative regiments are being reviewed. Bevacizumab, a monoclonal anti-VEGF that was initially developed for the treatment of colorectal cancer and commonly applied for ocular disease, has been expanded to retinopathy of prematurity, glaucoma, retinal vein occlusion, diabetic retinopathy, age-related macular degeneration, and other neovascular ocular diseases [26-29].
Due to the difference in ocular anatomy between humans and NZW rabbits, the NZW rabbit is unsuitable for directly extrapolating the pharmacokinetics to a human. However, animal models can provide an overview of the biomolecular event before human clinical trials. Current studies lack immunohistology retinal structure examination for PVR after OGI [4]. Our study was conducted to reveal whether bevacizumab and TA in different time points, which have been used widely in ocular field applications, are effective for this entity through intravitreal injection in rabbit OGI model experiments.
Materials and Methods
Study design
This study was an animal model experiment using a random control group design and post-test only.
Pharmacology
Bevacizumab (Avastin; Genentech/Roche, South San Francisco, CA) and Triamcinolone acetonide (Flamicort 40; Dexa Medica, Palembang, Indonesia) were acquired from the local pharmacy and prepared per the manufacturer's instructions.
Experimental animals
The right eyes of 27 male albino New Zealand rabbits (Oryctolagus cuniculus), (4–6 months old, weighing 2.0–2.5 kg) were carried. The animals were collected from Veterinary Faculty of Airlangga University farmer and treated following the Association for Research in Vision and Ophthalmology Statement for the Use of Animals in Ophthalmic and Vision Research. They were housed in the Experimental Animal Laboratory of the Veterinary Faculty of Airlangga University. The Care and Use of Laboratory Animal Care and Use Committee of Airlangga University authorized the studies (Registration No. 2.KEH.059.05.2022 dated May 24th, 2022). Pre-procedure, all rabbits were evaluated to ensure they were free of ocular disease. The animals were acclimatized for 1 week to prevent the spread of illness.
Open globe injury model
Rabbits were kept in separate cages, provided with food and water ad libitum. They were separated into five groups: OGI (CtrP), OGI with intravitreal TA 3 days after OGI (T+3), OGI with intravitreal TA 7 days after OGI (T+7), OGI with intravitreal bevacizumab 3 days after OGI (B+3), and OGI with intravitreal bevacizumab 7 days after OGI (B+7). Five left eyes were randomly selected as a negative control (CtrN). Intramuscular injections of ketamine and xylazine were administered as anesthesia prior to OGI. The operation was performed using microsurgery. The OGI model was begun by producing a 5 mm penetrating wound with blade no.11 circumferentially 6.0 mm behind the limbus in the supratemporal quadrant of the right eye (AAOM Brilliant Advent FS9; Appasamy, India) [14, 30]. The incision was sutured with 6-0 vicryl using an aseptic approach 30 min later. A single dosage of 1.25-mg bevacizumab (50 mL, Avastin; Genentech, South San Francisco, CA) was administered using a microliter syringe (Hamilton, Reno, NV, USA) for anti-VEGF therapy. Thirty-one tuberculin syringe was used to inject a single dosage of 4 mg TA (100 mL, Flamicort; Dexa Medica, Palembang, Indonesia). Post-OGI, the medicines were injected into the vitreous of the eyes at the indicated time points (3 or 7 days), with animals (n = 6) per time point. Before and after the eye damage and intravitreal injections, Levofloxacin eye drops were given 3 times daily for 3 days.
Ophthalmic examination
Before, 3, 7, and 21 days following OGI, bilateral routine ocular exams were conducted. The anterior region was inspected for inflammatory reactions using a biomicroscope. Both eyes' intraocular pressure (TonoVet Plus, iCare, Vantaa, Finland) was measured.
Histopathological examinations
In 10% buffered formalin, the globes were fixed. The anterior portion was excised 12 h later. The eyecups were then returned to the fixative. After 24 h, the eyecups were removed from the fixative, and a little portion of tissue containing the damaged site was cut. The tissues were fixed in paraffin and sectioned at 8-mm thickness for light microscopy examination. The examining physician and pathologist were double-blinded that the animal had been injected.
Immunohistochemistry staining
VEGF and TNF-α immunohistochemistry staining was conducted near the damage site. After heating the paraffin sections at 55 °C for 30 minutes, xylene dewaxing and gradient ethanol hydration were carried out. Using 1:50 dilutions of mouse monoclonal antibodies (clone C-1 and 52B83; Santa Cruz Biotechnology; USA), the expression of VEGF and TNF-α was observed. After counterstaining with Meyer's hematoxylin, the stained sections were seen using a light microscope (H600L, Nikon, Japan) equipped with a digital camera DS Fi2 300 megapixel and image processing software Nikon Image System. For the expression evaluation, five consecutive fields were counted using a modified semi-quantitative IRS scale of Remmele [31]. Each reading included positive and negative controls, and the slides were reviewed by two masked observers unaware of the identification group.
Statistical analysis
All statistical tests were conducted with statistical software (Prism version 9; GraphPad, San Diego, CA).
After the normality and homogeneity variance test with Shapiro-Wilk and Levene’s test, VEGF and TNF-a groups were examined with One-way ANOVA followed by Tukey's multiple comparison test. The correlation between each expression was studied with two-tailed Pearson correlation analyses. A p-value of <0.05 was considered statistically significant.
Results and Discussion
Twenty-seven right eyes were used, which was divided to the following groups: positive control (CtrP), OGI + triamcinolone D+3, OGI + triamcinolone D+7, OGI + bevacizumab D+3, and OGI + bevacizumab D+7. The treatment and positive control groups randomly chose five contralateral eyes (CtrN). These eyes were omitted from the research. During our study, one rabbit from each of the CtrP, B+3, and B+7 groups died at different times. Neither endophthalmitis, nor any other irritation of the front region was detected during the research.
Upregulation and downregulation of protein expression VEGF and TNF-α in the open globe injury model
Positive control group overexpression peaked with VEGF (9.64 ± 1,646, p=0.0002) (Table 1) and TNF-a (10.44 ± 1,381, p=0.0553) (Table 2). Interestingly, TNF-α in the negative control group from the contralateral eye (8.44 ± 2.64, p=0.0553) was higher than the VEGF expression (3.24 ± 1,322, p=0.0002). Although significant (p<0.000) expression was noted from the negative control group to the positive control group in VEGF, we found no significant difference between the positive control in TNF-α compared to the negative control (p=0.7974) (Table 1). Significant downregulation expressions of VEGF (Figure 1a) and TNF-α (Figure 1b) were observed in treatment groups, particularly the positive control group with T+3 group TA (p<0.000 and p<0.05, respectively). Nevertheless, there was no difference statistically (p>0.05) in the expression of VEGF (Figure 1a) and TNF-α (Figure 1b) of the positive control group to the bevacizumab treatment group.
Expression of VEGF and TNF-a (Figures 2a-F and 3a-f) by glial, Muller, and cell showed gradation across groups with brown chromogen colour in immunohistochemistry staining. All retinal layers were identifiable in all group sections with different thicknesses and contours as the effect of the treatment. Correlation between both expressions was analysed with linear regression analysis. P-value and Pearson’s correlation analyses displayed weak correlation and not significant statistically (Figure 4).
Clinical examination in an open globe injury model
There was no evidence of keratitis precipitate, hypopyon, hyphema, cataract, and RD.
Table 1: Comparison of VEGF expression in retinal tissue between treatment groups

Table 2: Comparison of TNF-α expression in retinal tissue between treatment groups
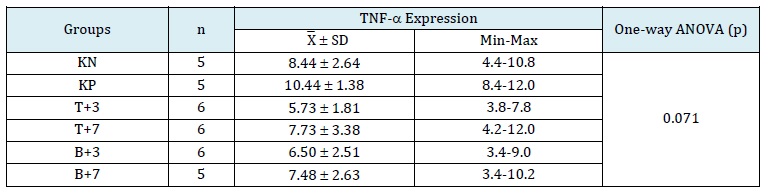
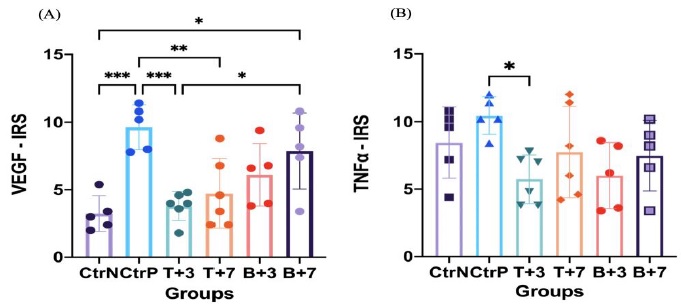
Figure 1: Expression of VEGF and TNF-α in retinal tissue from the rabbit open globe injury model. (A) The bar diagram showed a significant effect of OGI induction in VEGF expression from the negative control group compared to the positive control. Significant reduction of expression is marked in treatment groups T+3 and T+7. B+7 demonstrated a significant increase in expression compared to P1 and negative control. (B) TNF-α expression markedly decreased at positive control compared to treatment group T+3. Expression in the CtrN group from the contralateral eye showed high expression. Data are shown mean ± SD with a scattered plot of each individual, n = 5-6 for each group. *P< 0.05, **P< 0.001, and ***P< 0.0001 by One-way ANOVA with Tukey’s multiple comparisons test

Figure 2: VEGF expression with immunohistochemistry in all retina layers, especially in the INL and ONL layer (arrow). The negative control showed normal retinal architecture. Overexpression was exhibited in the positive control group with retinal oedema compared to the negative control. (A) negative control; (B) positive control; (C) OGI with TA D+3; (D) OGI with TA D+7; (E) OGI with Bevacizumab D+3; and (F) OGI with Bevacizumab D+7. GCL: Ganglion cell layer; IPL: Inner plexiform layer; INL: Inner nuclear layer; OPL: Outer plexiform layer; ONL: Outer nuclear layer; and OS: Outer segments of photoreceptors. Scalebar = 50 mm, objective lens 40x; microscope Eclipse E-i; camera DS Fi2 300 megapixel

Figure 3: Various TNF-α expressions with immunohistochemistry across groups in all retina layers, especially in the INL and ONL layer (arrow). (A) positive control; (B) OGI with TA D+3; (C) OGI with TA D+7; (D) OGI with Bevacizumab D+3; and (E) OGI with Bevacizumab D+7. GCL: Ganglion cell layer; IPL: Inner plexiform layer; INL: Inner nuclear layer; OPL: Outer plexiform layer; ONL: Outer nuclear layer; and OS: Outer segments of photoreceptors. Scalebar = 50 mm, objective lens 40x; microscope Eclipse E-i; camera DS Fi2 300 megapixel
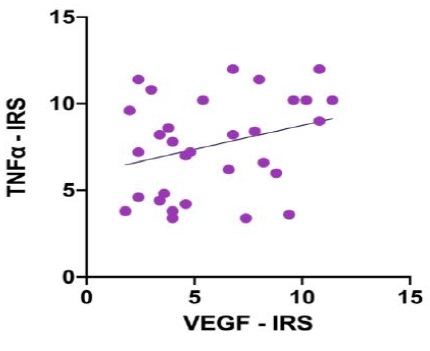
Figure 4: Scattergram showing simple regression analysis. Pearson’s Correlation analyses (r = 0.2950; r2 = 0.0870; 95% CI -0.05985 to 0.5837; p two-tailed = 0.1012)
The IOP results indicated that the mean IOP was comparable for all groups prior to the first day of the trial.
Following the introduction of the open globe injury, the IOP in the compromised eye decreased on day 3, with the positive control group displaying the lowest mean. All groups showed improvement on day 7 compared to day 3 measurements. Group T+3 had a higher mean than group B+3. Group 3 (T+3) had the same mean value as group 4 (T+7) despite group 4 had not yet been injected (Figure 5).
Tight connections between the retinal capillary endothelial cells (inner BRB) and the retinal pigment epithelial (RPE) cells (outer BRB) compose the blood-retinal barrier (BRB) as an immune privileged [32]. It prevents the passage of chemicals in healthy tissue that lack transporters or receptors on the luminal surface of the endothelial cells [33].

Figure 5: IOP pattern during the research with negative and positive control groups were included in graphs as the benchmark. The negative control group showed a relative linear line during the course. Another group exhibited various measurements as the result of the treatment. (A) IOP in injection three days after treatment group. The group with injection on day 3 showed a lower IOP than the initial data in the T+3 group and a higher one in the B+3 group, with the final IOP in group B+3 higher than T+3. (B) IOP in injection seven days after treatment group. Group T+7 and B+7 were observed with a relative linear line from day 7 to 21. Data are presented as mean ± SD in each examination time point before the study, day 3, day 7, and day 21 before decapitation
Ocular trauma typically compromises the blood-retina barrier integrity. The migration of inflammatory cells and the leakage of serum components caused by the disruption of this highly sophisticated barrier system might cause a significant biochemical shift. Microglia and RPE usually reside in the retinal layer and maintain retinal physiology. They will migrate to the vitreous following platelet and macrophage stimulation [32]. RPE will transform into a mesenchymal cell type that induces an extracellular matrix as the scaffold for PVR [9]. PVR would initially emerge due to excessive wound healing by these mechanisms [10]. Consequently, proper management and prophylaxis of PVR and tractional retinal detachment are crucial for achieving a favourable visual outcome. We found that a single dose of TA and bevacizumab intravitreal injection at different times inhibited the production of VEGF and TNF-a expression in the retinal layer. In the PVR pathophysiology, elevated expression of growth factors and cytokines in the vitreous has been implicated. VEGF is the primary growth factor involved in angiogenesis [34]. Although VEGF is prominent in vascular disease, VEGF concentration in PVR is high [35, 36]. Macrophage-secreted TNF-a recently appeared as a significant cytokine in the PVR induction following retinal apoptosis by inducing fibroblastic cell transformation of RPE cells [37, 38]. The absence of TNF-a in normal tissue contrasts with the concentration peak in PVR and RRD [34, 39,]. Although the precise function is unknown, and it is likely a measure of morbidity, our study showed that the expression of VEGF and TNF-a were elevated in all retinal layers due to PVR induction in the OGI eyes, with the positive group having the highest mean (9.64 ± 1,646, p 0.0002) and (10.44 ± 1,381, p 0.0553), respectively. The elevation of these expressions may contribute to the formation of retinal fibrosis from the wound site. Our investigation demonstrated a high TNF-a expression in the negative control. The stressful state following trauma influence the rabbit expression systemically and penetrate the ocular–retinal barrier [40, 36]. Our high expression level finding in the negative control group, which was harvested from the contralateral eye, might be induced by this condition, contrary to the normal group in a study of Chen et al. [41]. Pearson correlation was examined to determine whether VEGF and TNF-a. Few studies examine the histology of retinal tissue following an OGI [42, 14]. Mehdizadeh et al. showed from hematoxylin-eosin different grading of proliferative extension in the OGI model with and without Bevacizumab injection [30, 8]. However, recent studies have set forth the molecular role of PVR during OGI. PVR is generated by VEGF to suppress a non-PDGF-dependent route and to activate it from the PDGFR pathway, indicating that VEGF may have a role in nonangiogenic ocular entities [35]. Multiple cells, including the RPE, endothelial, and Müller glial cells, generate VEGF and TNF-a under ischemia conditions [43, 44]. Zhao et al. also discovered an increase in VEGF in vitreous specimens following open globe damage, particularly on days 10, 21, and 28 [14]. This observation accords with a review by Khaw et al. that VEGF has several peaks during wound healing and during vascular response, fibroblast phase, and remodeling [45]. Study showed that TNF-a after the OGI model had been expressed in retinal tissue from day 10 during the proliferative phase and remained stable after 30 days in a study of Chen et al. They also observed significant TNF-a expression after etanercept injection 72 h post-OGI induction [41]. Our study did not show significant down expression of TNF-a after treatment, which had different injection time points. Alternatively, the low expression of VEGF in the negative control group was interpreted as a result of the absence of hypoxia or the treatment's effect on a distant organ. The high expression in negative control eyes can support the hypothesis that TA and bevacizumab do not significantly affect TNF-a expression. This study found a weak relationship between VEGF and TNF-a expression, suggesting that the two cytokines do not affect each other during inflammation. TA is a synthetic glucocorticoid steroid in which the hydrogen atom has been substituted with fluorine. Favorable safety profile on IOP increase, cataract formation, and sterile endophthalmitis are the most common adverse effects that had been studied [22]. Clinically, IOP in the TA group was eventually managed with an antiglaucoma drop. In addition, it yielded superior final visual acuity compared with the control group [17]. Our investigation demonstrated a spike in IOP following IVTA, but IOP stabilization was observed at the end of the experiment. These results were inconsistent from Ghoraba et al., who reported an uncontrolled increase in IOP and other side effects, such as cataracts. Lang et al. demonstrated that the IOP spike was immediately detected following the injection, and the IOP returned to its preinjection level within 1 h [21]. Meanwhile, various anti-VEGF therapies, such as ranibizumab, concept, and aflibercept, have been developed specifically to inhibit PVR and have shown encouraging results [14, 46, 47, 30, and 17]. The precise method by which anti-VEGF drugs inhibit various cytokines is unclear. However, it may be a result of VEGF-A neutralization. It was thought that anti-VEGF therapies might assist recovery of retinal homeostasis by lowering the inflammatory and fibrotic response associated with ocular damage from the aforementioned pathway [48, 45, and 13]. Because there was no substantial downregulation in the bevacizumab treatment groups, it suggested that bevacizumab may have only a moderate effect on TNF-a. PVR is a complicated process, and a single mechanism may not be sufficient to halt the inflammatory reaction. Statistically significant down expression in the treatment group has been observed after TA injection, but the bevacizumab group gave no significant difference. TA decreases the expression of both VEGF and TNF-a, notably when administered 3 days after trauma. The half-life of TA intravitreally is ranged 6.08-7.995 days [49, 50]. Meanwhile, bevacizumab’s half-life in vitreous is 3.91-6.61 days [51, 52].
Although both agents have similar retention inside the vitreous, TA might give better expression due to extensive inflammation inhibition mechanisms. Zhao et al. showed that anti-VEGF treatment gave the best effect 3 days post-trauma than injection in 30 min and seven days. According to this study, our research showed better downregulation on day 3 than the day seven administration, although not significantly decreased. Better inflammation depression demonstrated in the previous study might be attributed to the smaller molecule of other anti-VEGF. Even though bevacizumab stays longer in the vitreous, its molecular weight is heavier. As a result, the substance is absorbed by retinal tissue at a lower rate than that in anti-VEGF. Weak correlation and statistical insignificance was found for both expressions. This finding suggests that both expressions do not possess a direct influence during wound healing process. This investigation has several limitations. The first limitation was serial termination to examine the IHC expression. Wound healing is a dynamic process, which involved milieu of cytokines. Our study was conducted from a tissue 21 days post-trauma. Day by day response is warranted in future study. Second, we applied the injection only on the third and seventh days after OGI. Future research could elaborate on the effect of different time points to evaluate the efficacy of earlier bevacizumab administration. Since trauma patients will have a delay from the trauma event to the primary repair of more than 8-24 h, injection in the first 24 h is warranted to be included in future research [3].
Lastly, we did not conduct a cytokine level assay on the blood sample. Consequently, we could not demonstrate the effect of increased TNF-a expression due to a systemic situation. Assessing the ocular and systemic cytokines can provide a complete picture. This study demonstrated the pathophysiology of TA and bevacizumab in the OGI eye. Currently, there is no standardized adjunctive treatment after OGI. Therefore, TA and bevacizumab inhibited the production of VEGF and TNF-a in the retinal layer produced by an OGI. The inhibitory effect of TA was greater than that of bevacizumab, primarily 3 days after OGI induction without IOP spike at the end of the experiment. Being male and having a poor income are the two risk factors for OGI. This patient population can economically achieve visual persistence and injection compliance with TA. Further research should be done in nonhuman mammal species for OGI to fill the gap of our knowledge regarding the drug effect to wound healing factors within retina tissue. This research has several limitations, such as this study is conducted on rabbits, which may not fully represent the complexities of the human eye. The results might not directly translate to human patients, and there could be differences in response to treatment between rabbits and humans. The follow-up period is limited to 21 days, which may not be sufficient to assess the long-term effects and safety of treatments. Retinal detachment and other complications could manifest beyond this timeframe.
The study examines the effect of a single intravitreal injection of each drug. Real-life treatments may involve multiple injections or different dosing regimens, and the long-term effects of repeated injections are not explored.
Acknowledgments
The authors would like to acknowledge “CaroVet Dokter Hewan” for supporting TonoVet during our research; staffs and residents of Ophthalmology Department Airlangga University/Dr Soetomo General Hospital for their endless support and constructive input.
Disclosure Statement
No potential conflict of interest was reported by the authors.
Funding
We would like to thank “Penelitian Unggulan Fakultas” research grant from Universitas Airlangga, Surabaya, Indonesia for supporting this research.
Authors' Contributions
All authors contributed to data analysis, drafting, and revising of the paper and agreed to be responsible for all the aspects of this work.
ORCID
Evelyn Komaratih
https://orcid.org/0000-0002-4821-4572
Clarisa Finanda
https://orcid.org/0000-0002-2856-4778
Wimbo Sasono
https://orcid.org/0000-0002-2339-1222
Nurwasis
https://orcid.org/0000-0001-8302-729X
Citra Dewi Maharani
https://orcid.org/0000-0002-8111-4055
Kautsar Abiyoga
https://orcid.org/0009-0004-3191-681X
Djoko Legowo
https://orcid.org/0000-0003-0099-8782
Hari Basuki Notobroto
https://orcid.org/0000-0002-3905-7413
Willy Sandhika
https://orcid.org/0000-0002-8499-1600
HOW TO CITE THIS ARTICLE
Evelyn Komaratih*, Clarisa Finanda, Wimbo Sasono, Nurwasis, Citra Dewi Maharani, Kautsar Abiyoga, Djoko Legowo, Hari Basuki Notobroto, Willy Sandhika, Effectivity and Safety after Intravitreal Triamcinolone Acetonide and Bevacizumab Injection in Open Globe Injury. J. Med. Chem. Sci., 2023, 6(12) 3121-3134.


