Document Type : Review Article
Authors
- Muhammad Irfan Rahmatullah 1
- Asra Al Fauzi 1
- Nur Setiawan Suroto 1
- Muhammad Arifin Parenrengi 1
- Wihasto Suryaningtyas 1
- Dyah Fauziah 2
- Budi Utomo 3
- Billy Dema Justia Wahid 1
- I Wisnawa 1
1 Department of Neurosurgery, Faculty of Medicine, Airlangga University, Surabaya, Indonesia
2 Department of Pathology Anatomy, Faculty of Medicine, Airlangga University, Surabaya, Indonesia
3 Department of Public Health, Faculty of Medicine, Airlangga University, Surabaya, Indonesia
Abstract
Background: Cerebrospinal fluid (CSF) leakage is a well-documented and significant complication that can occur following neurosurgical procedures. The association between CSF leakage and various morbidities has been extensively studied. Research on different materials as potential dura mater grafts has been conducted since 1890. The search for the optimal material for dura mater grafts remains ongoing in current research. This investigation aims to assess the incidence of CSF leakage and infection subsequent to duraplasty using xenografts in comparison to autografts.
Methods: This study aims to systematically analyze the CSF leakage and infection rates following duraplasty procedures utilizing xenografts and autografts. The research methodology includes a comprehensive search of the PubMed electronic database and a manual search of Google Scholar, adhering to the Preferred Reporting Items for Systematic Review and Meta-Analysis (PRISMA) guidelines.
Results: Among the patients in the xenograft group, a total of 90 individuals out of 452 experienced complications in the form of CSF leaks and complicated pseudo-meningoceles. This study was conducted to investigate the incidence of CSF leaks and complicated pseudo-meningoceles in autografts, with a sample size of 540 patients. The results showed that 79 patients experienced these complications, with a statistically significant p-value of 0.05 and an odds ratio of 1.45 (95% confidence interval 1.00-2.10). Another aspect of the study examined the incidence of meningitis complications in xenografts, with a sample size of 400 patients. The results showed that 25 patients experienced meningitis. Among the 446 patients who underwent autografts, 11 patients (p=0.05; OR 2.10, 95% CI 1.01-4.36) experienced meningitis complications.
Conclusion: The analysis of duraplasty techniques shows that the use of xenografts increases the incidence of CSF leaks and pseudo-meningocele complications while raising the risk of meningitis.
Graphical Abstract
Keywords
Introduction
The incidence rate of Cerebrospinal Fluid (CSF) leakage is a significant concern following neurosurgical procedures, particularly in posterior fossa surgery and skull base surgery, where it is reported to be as high as 10%. The incidence of meningitis, pneumocephalus, longer length of stay, and death has been found to be associated with CSF leakage. In certain cases, a secondary surgical procedure may be necessary to address a dura mater leak in patients [1].
Inadequate closure of the dura mater constitutes a prominent etiology for morbidity in numerous neurosurgical procedures, necessitating the pursuit of advanced techniques and methodologies to enhance the safety and efficacy of dura mater closure during surgical interventions [2].
Since 1890, extensive research has been conducted to explore various materials suitable for dura mater grafts, including metallic sheets, autologous, homologous, or autograft body tissues, as well as biosynthetic substances. However, the search for an optimal material for dura mater grafts continues.
The ideal substitute material for dura mater should possess specific characteristics, including inertness, non-toxicity, non-carcinogenicity, water resistance, suture compatibility without causing adhesion to the cortex, ease of procurement, sterility, and cost-effectiveness [3].
In general, synthetic graft materials are rejected by the patient's body due to excessive inflammatory reactions from the local tissue and are characterized by irritation of the underlying brain cortex, excessive scar tissue formation, meningitis, and bleeding [4].
Moreover, these materials are relatively expensive. Dura mater from human cadavers as an autologous graft material has never been used as an alternative because it has been proven to transmit Creutzfeldt-Jakob disease [5].
Graft materials from autologous or autograft body tissues, such as the pericranium, fascia lata, and temporalis muscle fascia, do not pose a risk of infectious diseases but have the impractical disadvantage of requiring the additional incisions during surgery [6].
The amniotic membrane has long been used in the field of surgery, especially in plastic and eye surgery, as a suitable graft due to its characteristics including the ability to stimulate the epithelialization process, inhibit excessive fibrotic processes, inhibit inflammatory processes and angiogenesis, minimize the immune response, have antimicrobial and antiviral effects, and function as an effective wound dressing for adhesion, and moisturizing the wound surface, thereby accelerating wound healing.
In this study, the researchers aim to understand the incidence of CSF leakage and infection after duraplasty using xenografts. As a comparison, they also compared xenografts with autografts for dura mater grafting.
Materials and Methods
This systematic review aims to compare the incidence of CSF leakage and infection after duraplasty using xenografts and autografts. The search was performed using boolean operators in the form of AND to search for literature containing all keywords and OR to search for literature containing alternative keywords.
The research database consists of a literature search on PubMed and a manual search on Google Scholar. There is no limitation on the publication year of the literature.
The literature search was conducted following the Preferred Reporting Items for Systematic Review and Meta-Analysis (PRISMA) search flow based on suggestions from the Cochrane Review [7].
The inclusion criteria were cohort and case-control studies that compared xenografts, autografts, and documented complications such as CSF leakage, pseudo-meningocele, and meningitis. The exclusion criteria included case report studies and literature review studies.
The risk of bias for Randomized Clinical Trials (RCTs) was assessed based on The Cochrane Collaboration Tool for Assessing Risk of Bias in Randomized Trials published in 2011 [8].
The risk of bias for studies other than RCTs was assessed based on criteria and the Newcastle-Ottawa Scale for cohort studies according to the Cochrane Handbook for Systematic Reviews of Interventions Chapter, entitled: ''Assessing risk of bias in a non-randomized study.''

Statistical analysis was performed using SPSS 24 and Revman version 5.4 from the Cochrane Review.
Results and Discussion
The flowchart illustrating the research selection can be seen in Figure 1. The quality assessment of cohort studies was conducted using the Newcastle-Ottawa assessment (Tables 1 and 2). In this study, there were five retrospective cohorts and one prospective cohort, along with one case-control study. All 6 cohort studies and the case-control study were considered to be of good quality. In the selection section, all studies received high scores, as they were highly representative of the average target population. In the comparability section, each cohort study received two-star scores for controlling age, sex, marital status, and other relevant factors.
In terms of outcome, all studies utilized record linkage, had sufficient follow-up duration, and achieved over 80% response rate.
Based on the results of the meta-analysis of seven studies, the Odds Ratio (OR) was calculated for complications related to the dura graft procedure, such as CSF leakage and pseudo-meningocele, as summarized in Table 3.

For xenograft, 90 out of 452 patients experienced CSF leakage and pseudo-meningocele complications. In autograft, 79 out of 540 patients experienced these complications (OR 1.45, 95% CI 1.00-2.10). The Forest Plot (Figure 2) and Funnel Plot (Figure 3) present the meta-analysis results, indicating that CSF leakage and pseudo-meningocele are more likely to occur in xenograft cases. This result is supported by a significant p-value of 0.05 (p < 0.05).

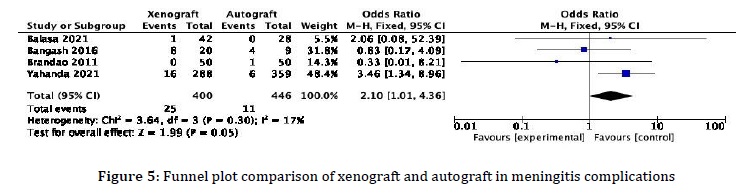
Regarding meningitis complications in duraplasty, four studies discussed this aspect. In xenograft, 25 out of 400 patients experienced meningitis. In autograft, 11 out of 446 patients had meningitis (OR 2.10, 95% CI 1.01-4.36). The meta-analysis results (Figure 4 and Figure 5) suggest a higher likelihood of meningitis occurrence in xenograft duraplasty. This result is supported by a significant p-value of 0.05 (p < 0.05).
This systematic review analyzed literature comparing the incidence of CSF leakage in duraplasty procedures using autografts and xenografts. The search yielded a total of ten relevant studies. Duraplasty is a reconstructive procedure aimed at closing the dura mater to prevent CSF leakage after surgery. The quest for an ideal dura replacement material remains a challenge in neurosurgery. Various materials including allografts, xenografts, synthetic materials, and muscle fascia have been tested and used as potential options [9].
Study characteristic
This study utilized data from the literature with a retrospective and prospective cohort study design. There were five literature sources with a retrospective cohort study design, one literature source with a prospective cohort study design, and one prospective case-control study. Risk of Bias Assessment was conducted using the Newcastle-Ottawa Quality Scale for cohort and case-control studies.
The Newcastle-Ottawa Quality Scale is a tool developed to assess the quality of studies for meta-analysis. It employs a star system to grade studies based on three main aspects: selection of study groups (Selection), comparability of groups (Comparison), and ascertainment of exposure or outcome of interest in cohort studies (Outcomes). Most of the cohort studies included in the literature exhibited good quality according to the Newcastle-Ottawa Quality Scale.
Patient’s characteristic
Balasa's study involved 70 patients who underwent posterior fossa decompression with duraplasty between January 2003 and December 2018. The study included 54 women and 16 men, with a mean age ranging from 18 to 66 years (mean age of 41.9 years).
The mean onset of symptoms varied from 3 months to 50 years, with suboccipital and general headaches being the main symptoms for Chiari malformation type 1 (CM-1) [10].
Bangash conducted a study on 45 patients who underwent craniotomy or craniectomy with dural closure. Among the participants, 18 (40%) were men, and 27 (60%) were women, with a mean age of 36.4 ± 22 years. The patients were followed up for nine months [11].
Brandao's study was a prospective case-control nonrandomized study conducted at Santa Casa Hospital Brazil from November 2009 to November 2010. A total of 100 patients who underwent cranial procedures were followed up for three months. The mean age of the patients in the dural substitution group with collagen matrix was 45.7 years; while in the control group (pericranium), the mean age was 46.6 years. Various cranial neurosurgical procedures were performed, including surgery for tumors (most common), aneurysms, craniectomy for ischemic stroke, and Chiari type I malformation [12].
Manzoor's study compared porcine small intestinal submucosal grafts (PSISG) with autologous tissue in multilayer lateral skull base reconstruction involving 45 patients. The average age of PSISG users ranged from 54 to 68 years, while for autografts, it was 44 to 68 years. All patients were followed up for six months after surgery, with assessments conducted for defect location, CSF leak, encephalocele, and dura erosion [13].
Al Melesy's research involved 24 patients who underwent hypothalamic tumor surgery using two types of dura graft: fascia lata graft or duragen.
In this study, 24 patients were recruited, consisting of 15 women and nine men, with a mean age ranging from 17 to 55 years. The patients were followed up for six months [14].
Totten's study examined 77 patients who underwent lateral skull base resection, comparing the use of dura grafts: porcine small intestinal submucosal grafts (SISG) and autologous fascia. Among the patients, 56 received dura graft autografts, while 21 received dura graft SISG. The average age for autograft patients ranged from 37 to 60 years, while for xenograft users, it was 45 to 62 years [15].
Yahanda conducted a retrospective study from July 2011 to October 2014, including a total of 781 pediatric patients who underwent posterior fossa decompression with duraplasty due to Chiari I malformation with syringomyelia.
The patients had an average age ranging from 1 to 20 years, with 327 male patients and 454 female patients included in the study. The non-autologous grafts were divided into bovine pericardium (n=63), bovine collagen (n=225), synthetic (n=99), and allograft (n=35) [16].
CSF leak and pseudomeningocele
The meta-analysis results of this study, which compared duraplasty using xenograft and autograft, revealed a higher risk of CSF leaks and pseudo-meningoceles associated with xenograft [17-22]. Among the seven studies included in the analysis, three reported a higher incidence of CSF leaks or pseudo-meningocele in the xenograft group. Xenograft dura grafts are derived from collagen-based materials obtained from living sources, with bovine pericardium being one of the commonly used tissues due to its higher type I collagen content compared to pigs or horses.
In Balasa's study, which involved 70 patients with Chiari malformation type 1 (CM-1) who underwent posterior fossa decompression with duraplasty between January 2013 and December 2018, two types of non-autografts (Duragen Integra, Duragen Plus Integra, and Durarepair Medtronic) were used in 42 patients, while autografts (fascia lata and pericranium) were used in 28 patients. The xenograft or non-autograft group had a higher incidence of pseudo-meningocele complications, observed in 22 (52.4%) of the 42 patients who underwent duraplasty with non-autografts [10]. In Bangash's study, 12 (60%) out of 20 patients who received xenograft dura grafts experienced CSF leaks [11, 19].
Al Melesy's study, comparing collagen matrix grafts (12 patients) and fascia lata grafts (12 patients), found that the collagen matrix group had the highest incidence of CSF leakage. The study also assessed the grade of CSF breaches in each group, with grade 2 leaks observed in one patient (8.3%), grade 3 in two patients (16.7%), and grade 4 in one patient [14, 19, 20].
Based on the conclusions of this meta-analysis, xenograft dura grafts are more likely to lead to complications of CSF leaks and pseudo-meningoceles. An important consideration when selecting a dura graft as a substitution is its ability to mimic the mechanical properties of the original dura. The mechanical properties of dura substitutes can be evaluated in terms of tension and length. If the length of the dural substitute is shorter than the original dura mater, it becomes challenging to suture. On the other hand, if it is too long compared to the standard, the dural substitute may stretch excessively, leading to CSF leakage when intracranial pressure increases. Aydin's research suggests that bovine pericardium, a type of xenograft, exhibits tension values of 6-18.96 MPa and a length of 20.67-39.5%, while human dura mater has tension values of 4.70-12.76 MPa and a length of 7-20%. Mechanical friction between the xenograft and surrounding tissues due to differences in length and stiffness may trigger chronic inflammatory reactions and result in intracapsular bleeding. In addition, the rigidity of graft material, if too dense or inflexible, it can create larger suture holes, potentially leading to CSF leakage [23].
Apart from that, other factors, such as poor surgical technique in wound closure, imperfect dural closure, and hydrocephalus, can also contribute to the development of CSF leaks and pseudo-meningoceles [12].
Yahanda's meta-analysis of 27 studies, focusing on complications associated with various dura graft materials in posterior fossa decompression surgery, examined five different types of dura grafts: autografts in 404 (27.6%) patients, synthetic grafts in 272 (18.6%) patients, bovine pericardium grafts in 181 (12.4%) patients, collagen-based grafts in 397 (27.2%) patients, and allografts in 207 (14.2%) patients. The study found a lower incidence of pseudo-meningocele complications in the autograft group than in collagen-based grafts, bovine pericardium grafts, and allografts [16, 18, 22].
In Totten's study conducted from 2016 to 2018, focusing on patients undergoing resection of various lateral skull base tumors via trans-labyrinthine approach using bone pate and fascia autograft or porcine small intestinal submucosal grafts (SISG), seven patients experienced CSF leaks and pseudo-meningoceles, all of whom had fascia autografts [15].
Yahanda's study, which covered patients from July 2011 to October 2014 undergoing posterior fossa decompression with duraplasty for Chiari I malformation and syringomyelia, involved patients with a mean age below 21 years, predominantly females. Autograft pericranium was used in 253 (70.5%) patients, cervical fascia in 40 (11.1%) patients, nuchal ligaments in 23 (6.4%) patients, and 43 (12%) patients had an unknown graft type. For xenografts, bovine pericardium was used in 63 (14.9%) patients, and bovine collagen was used in 225 (53.3%) patients. The study found no significant difference in the incidence of CSF leaks and pseudomeningoceles between xenograft and autograft duraplasty.
In the autograft group, complications of CSF leaks and pseudo-meningoceles occurred in 35 patients out of a total of 359 patients who underwent autograft duraplasty [17-19].
In the xenograft group, 33 patients out of a total of 288 patients experienced complications of CSF leaks and pseudo-meningoceles. Within the xenograft group, bovine collagen had the highest incidence of pseudo-meningocele complications, with 19 patients affected, while four patients experienced CSF leaks [16, 19, 20].
Meningitis
The results of a meta-analysis of four studies discussing complications of meningitis showed that the xenograft group had a higher likelihood of causing meningitis complications. In Balasa's study, one patient who received a Non-AutoG dura graft (Duragen Integra, Duragen plus Integra, and DuraRepair Medtronic) experienced complications of aseptic meningitis [24-28].
The study also highlighted the consideration of Non-AutoG dura grafts due to their high risk of aseptic meningitis and pseudo-meningocele formation [10, 25, 26].
In Bangash's study, comparing pericranium autografts and xenografts using Duraguard and Duragen in patients undergoing craniotomy with duraplasty, the xenograft type (Duraguard) had the highest incidence of postoperative infection, affecting 8 (42.2%) out of 19 patients [11, 17]. Yahanda's study reported that the xenograft group and bovine collagen had the most cases of meningitis complications, affecting 12 patients out of a total of 225 patients [16, 19, 20].
Complications of meningitis in xenograft materials may manifest as fibrosis and the formation of a fibrous capsule, leading to increased cortical adhesion, reduced naturalization, and increased recruitment of inflammatory cells. The continuous inflammatory response to xenografts in the body can cause meningeal irritation and symptoms of meningitis and aseptic meningitis [24-26, 29].
A meta-analysis of 27 studies conducted by Yahanda, which examined complications associated with different types of dura graft materials in posterior fossa decompression surgery, including autografts (404 patients, 27.6%), synthetic grafts (272 patients, 18.6%), bovine pericardium grafts (181 patients, 12.4%), collagen-based grafts (397 patients, 27.2%), and allografts (207 patients, 14.2%), reported a lower incidence of aseptic meningitis and meningitis infections compared to collagen-based grafts, bovine pericardium grafts, and allografts [19, 20].
Based on the data from meta-analysis results, it can be concluded that xenografts have a higher incidence of complications such as CSF leaks, pseudo-meningocele, and meningitis compared to autografts. This comparative study of xenografts and autografts in complications of CSF leaks, pseudo-meningocele, and meningitis is the first meta-analysis conducted, providing valuable insights into the complications of duraplasty.
Limitation
The systematic review and meta-analysis comparing complication rates between autograft and xenograft duraplasty included 6 cohort studies and 1 case series. However, there are several limitations to consider. These limitations include potential selection bias, publication bias, heterogeneity among the included studies, inadequate control of confounding factors, variations in the quality of the primary studies, data availability issues, and potential language and publication bias. These limitations should be taken into account when interpreting the findings of the study.
Conclusion
Duraplasty using xenograft carries a higher risk of CSF leaks and pseudo-meningocele compared to autograft, and duraplasty with xenograft is associated with a higher risk of meningitis complications compared to autograft.
Disclosure Statement
No potential conflict of interest was reported by the authors.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Authors' Contributions
All authors contributed to data analysis, drafting, and revising of the paper and agreed to be responsible for all the aspects of this work.
Orcid
Asra Al Fauzi
https://orcid.org/0000-0002-5155-2476
Muhammad Irfan Rahmatullah
https://orcid.org/0009-0001-8453-5621
Nur Setiawan Suroto
https://orcid.org/0000-0001-6706-2896
Muhammad Arifin Parenrengi
https://orcid.org/0000-0002-1327-8955
Wihasto Suryaningtyas
https://orcid.org/0000-0002-1187-3777
Dyah Fauziah
https://orcid.org/0000-0002-7661-1647
Budi Utomo
https://orcid.org/0000-0001-6060-9190
Billy Dema Justia Wahid
https://orcid.org/0000-0002-4093-9325
I Wayan Weda Wisnawa
https://orcid.org/0000-0001-5376-7326
HOW TO CITE THIS ARTICLE
Asra Al Fauzi*, Muhammad Irfan Rahmatullah, Nur Setiawan Suroto, Muhammad Arifin Parenrengi, Wihasto Suryaningtyas, Comparison of Complication between Autograft and Xenograft Duraplasty: A Systematic Review and Meta-Analysis. J. Med. Chem. Sci., 2023, 6(11) 2793-2803.


