Document Type : Case Report
Authors
Department of Clinical Pathology, Faculty of Medicine, Airlangga University, Dr. Soetomo General Academic Hospital, Surabaya, Indonesia
Abstract
Autoimmune hemolytic anemia (AIHA) is an infrequent etiology of anemia during pregnancy, and its occurrence in the postpartum period is even more uncommon. This study presents the case of a 27-year-old woman who exhibited severe anemia and thrombocytopenia six weeks after delivery. Hemolysis was confirmed upon evaluation, and a diagnosis of AIHA was established based on a positive direct antiglobulin test (DAT) and the presence of autoantibodies targeting red blood cells (RBCs). Treatment with steroids resulted in an improvement in the patient's hemoglobin levels. The demographic characteristics observed in our case align with prior research, which has documented mild anemia during pregnancy, lower gestational age, and decreased fetal birth weight in primary AIHA cases. Furthermore, thrombocytopenia can be manifested in AIHA during pregnancy, necessitating long-term monitoring of such patients. The AIHA rarity as a causative factor for anemia during pregnancy, particularly in the postpartum period, underscores the significance of including AIHA in the differential diagnosis for severe anemia. Moreover, extended follow-up care is essential for managing these patients. Employing a multidisciplinary approach to prenatal care facilitates the identification and management of rare complications, such as AIHA.
Graphical Abstract
Keywords
Introduction
In 2018, the prevalence of anemia among Indonesian women of reproductive age exceeded one million cases, with immune disorders and iron deficiency among the leading global causes [1, 2].
Within East Java, the incidence of anemia in pregnant women is reported to reach 37.1%, with 10% of all pregnant women receiving antenatal care in Surabaya presenting with anemia [3, 4].
Autoimmune hemolytic anemia (AIHA) during pregnancy is an uncommon condition, occurring at a rate of 1-3 cases per 100,000 per year, and may be manifested during both gestation and the postpartum period. The majority of cases are severe and necessitate treatment with steroids and transfusions [5-7].
AIHA is characterized by the destruction of red blood cells as a result of autoantibodies and can be classified into warm, cold, or mixed types based on the Direct Antiglobulin Test (DAT) [5].
Approximately 5% of all AIHA cases during pregnancy are present in the postpartum period [5, 8].
This report presents a case of AIHA diagnosed seven days after cesarean delivery, resulting in severe anemia compounded by delivery-related complications yet exhibiting favorable clinical outcomes.
Case presentation
Case Presentation A 27-year-old primiparous woman, gravida 3, para 3, was admitted to the emergency department with complaints of asthenia and abdominal pain seven days following cesarean delivery. The decision for emergency delivery was prompted by premature membrane rupture and fetal distress. The patient was at a gestational age of 32 weeks at the time of delivery. Preceding the procedure, the patient's hemoglobin level was measured at 10.8 g/dL, and her platelet count was 10,000/µL. The neonates exhibited a birth weight of 1,900 grams and APGAR scores of 7-8. The patient remained hospitalized for three days post-delivery and was subsequently discharged in a favorable clinical condition. Her obstetric history was unremarkable, characterized by regular antenatal care visits and the utilization of prenatal supplements without undergoing any laboratory examinations. Upon arrival at the emergency department, the patient was diagnosed with severe anemia (Hb 3.8 g/dL) and thrombocytopenia (platelet count 87,000/µL). The patient possessed blood type O Rh positive. Notably, her blood pressure was 89/52 mmHg, pulse rate was 140 beats per minute, and respiratory rate was 35 breaths per minute. Immediate resuscitation was performed, necessitating an emergency blood transfusion. However, blood products were unavailable at the previous hospital. Due to the risk of hemodilution and persistent hypoxia, the use of crystalloids was avoided. The crossmatch results revealed incompatibility, with +1 major, +3 minor, and +3 in the auto control. Subsequently, the patient received two units of packed red blood cells (PRC), with the major crossmatch positivity exhibiting a weaker reaction than the auto control. Blood transfusion was limited to a maximum of two units per day until the patient's hemoglobin levels reached 10 g/dL, accompanied by intensive monitoring.
The etiology of the patient’s severe anemia was investigated through a comprehensive examination. Physical assessment revealed anemic conjunctiva and the presence of a palpable mass in the abdomen, encompassing both sides of the cesarean incision, without alterations in abdominal circumference. No organomegaly was detected. Abdominal ultrasound unveiled a suspected subfascial hematoma measuring 9.4 × 2.41 cm. No free fluid was observed within the hepatorenal, splenorenal, Douglas cavity, or paracolic gutters. Gynecologic examination exhibited no discernible abnormalities. Laboratory findings, as listed in Table 1, indicated severe anemia, thrombocytopenia, reticulocytosis, hyperbilirubinemia, elevated LDH levels, positive ANA test, and normal C3 and C4 levels. No abnormalities were identified in PPT and APTT coagulation tests. Urine examination demonstrated hematuria (+3 erythrocytes) and trace proteinuria. The direct agglutination test (DAT) yielded a positive result.

Following an incompatible result in the initial crossmatch test, the patient underwent screening for irregular antibodies using three Hemascreen panels. The results revealed positive antigens for anti-k, anti-Kpb, and anti-Lub. Based on these findings, the provisional diagnosis for the patient was autoimmune hemolytic anemia (AIHA) with suspected subfascial hematoma associated with Systemic Lupus Erythematosus (SLE).
A laparotomy surgery was scheduled for the patient to address the bleeding from the subfascial hematoma. Preceding the surgery, the patient's hemodynamic status was stabilized, and she received blood transfusion therapy consisting of two units of Packed Red Cells per day until her hemoglobin level reached 10 g/dL, totaling 12 units of PRC.
In addition, the patient was administered intravenous methylprednisolone at a dose of 125 mg three times daily for the initial five days. On the sixth day, with the patient's hemoglobin level measuring 11.1 g/dL, laparotomy surgery was successfully performed. Although the patient's hemoglobin level slightly decreased to 10.6 g/dL after the surgery, no complications were encountered. Regular monitoring was conducted, and on the seventh day of treatment, the patient reported no complaints. The steroid dosage was gradually tapered, starting with 125 mg of methylprednisolone intravenously once daily for three days, followed by a further reduction to 62.5 mg of methylprednisolone intravenously for two days.
The patient exhibited remarkable clinical improvement and was discharged from the hospital with a hemoglobin level of 12.1 g/dL. Subsequently, the steroid treatment was transitioned to an oral form, commencing with 16 mg of methylprednisolone.
Results and Discussion
The demographic characteristics of this case align with findings from previous studies [5, 9].
The patient is 27 years old, which is the median age for AIHA during pregnancy. The patient's age of 27 corresponds to the median age for AIHA during pregnancy.
Furthermore, mild anemia during pregnancy and lower gestational age were observed, with the termination of pregnancy typically occurring around the 32nd week [10, 11]. Notably, primary AIHA was associated with fetal low birth weight in most cases.
AIHA is defined as a hemoglobin level below 10 g/dL along with a positive Direct Antiglobulin Test (DAT) [9].
In this patient, evidence of AIHA was supported by a markedly low hemoglobin level, reticulocytosis, elevated bilirubin, and LDH levels, positive DAT, and the presence of spherocytes in the blood smear evaluation.
The presence of polychromasia indicates reticulocytosis, while the occurrence of spherocytes is frequently observed in hemolytic processes. Severe cases may also exhibit normoblasts and agglutination, as seen in this case [12].
The DAT, also known as the Coombs test, detects the presence of IgG and complement (C3) bound to red blood cells, which can lead to their destruction. Most cases of AIHA are DAT positive. However, 5-10% of AIHA cases may be DAT negative [13, 14].
Autoantibodies to RBCs rarely develop in pregnant women, with anti-c and anti-K antibodies being particularly associated with significant hemolysis compared to other antibodies.
In this patient, the autoantibodies identified were anti-k, anti-Kpb, and anti-Lub. These antibodies may have arisen due to micro-transfusions during pregnancy, leading to autoimmunization against RBCs. Nonetheless, these autoantibodies pose no harm to the pregnancy [15].
In patients with AIHA, the presence of autoantibodies can mask the presence of alloantibodies. Knowledge of the patient's phenotype can guide RBC transfusion strategies [16].
Thrombocytopenia is a common finding in AIHA during pregnancy, and it is often accompanied by immune thrombocytopenia (ITP) in 0.8-3.7% of cases, a condition known as Evans Syndrome [17, 18].
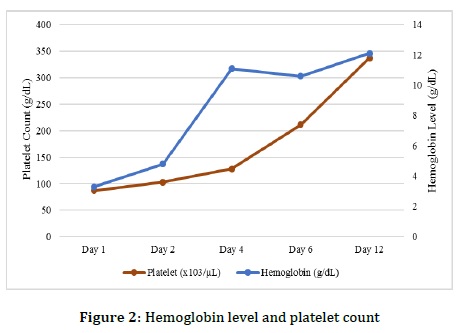
In this patient, the thrombocytopenia could potentially be attributed to gestational thrombocytopenia, as the platelet count increased during the treatment course, as depicted in Figure 2. However, long-term follow-up is recommended to monitor the platelet count and ensure the absence of thrombocytopenia recurrence.
Warm AIHA, mediated by IgG, accounts for approximately 80-90% of cases, while cold AIHA, mediated by IgM, is more common in children. Each type of AIHA can be further classified based on the presence of underlying diseases [12].
A previous report by Maroto et al. demonstrated that warm, IgG-mediated AIHA exhibited normal clinical findings and did not result in adverse perinatal outcomes [19, 20].
In this patient, Warm AIHA secondary to SLE remains a possibility. Although the ANA test was positive, the C3 and C4 levels remained within the normal range. SLE can also manifest during pregnancy, increasing the risk of pregnancy-related complications [21, 22].
However, primary AIHA due to the pregnancy itself, which is exceedingly rare, remains the primary etiology of AIHA in this patient, as there was no history of other illnesses before or during the pregnancy.
Conclusion
Anemia is a prevalent complication during pregnancy. While there are numerous reports on the incidence of AIHA in pregnant women, the AIHA occurrence in the postpartum period is relatively rare. This case underscores the significance of adopting a comprehensive approach to detecting anemia during gestation, as AIHA can be manifested as either mild or severe anemia. It is imperative to recognize the potential risks of AIHA during antenatal care visits.
Acknowledgements
Miftah Hasanah, M.D., contributed to data collection, performed laboratory examinations, obtained images, drafted the case report, and revised the manuscript and Dr. Yetti Hernaningsih, M.D., Clinical Pathologist, provided expertise in hematology and contributed to the revision of the paper.
Disclosure Statement
No potential conflict of interest was reported by the authors.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Authors' Contributions
All authors contributed to data analysis, drafting, and revising of the paper and agreed to be responsible for all the aspects of this work.
Orcid
Yetti Hernaningsih
https://orcid.org/0000-0001-8773-8267
Miftah Hasanah
https://orcid.org/0000-0002-9791-847X
HOW TO CITE THIS ARTICLE
Yetti Hernaningsih, Miftah Hasanah. Hemolytic Anemia in Pregnancy: A Rare and Challenging Case in Indonesia. J. Med. Chem. Sci., 2023, 6(11) 2679-2685.


