Document Type : Original Article
Authors
1 Department of Prosthodontics, College of Dentistry, University of Baghdad, Baghdad, Iraq
2 Department of Prosthodontics, College of Dentistry, University of Baghdad, Bab-Almoadham, P.O. Box 1417, Baghdad, Iraq
3 Dean of College of Dentistry, The Iraqia University, Baghdad, Iraq
Abstract
Background: Despite the fact that the mechanical properties of PMMA appliances and goods are not ideal, the goal of this research was to reinforce high impact acrylic dentures with rice husk-derived silica in various concentrations to improve some of the mechanical qualities of the denture base.
Materials and Method: Based on the pilot study made in College of Dentistry University of Baghdad, to create the specimens, silica particles from a rice husk source were added to high impact heat cure resin at 1 wt.% and 3 wt.% (by weight). On the basis of the three tests conducted the transverse strength, impact strength and hardness tests, the prepared samples were evaluated using Fourier-transform infrared spectroscopy (FT-IR), particle size analysis, and a field emission scanning electron microscope (FE-SEM).
Results: The results revealed that the addition of silica particles yielded a significantly improvement in transverse strength test (P-value= 0.001), and non-significantly increase in both impact strength and hardness values compared with the control group.
Conclusion: Silica particles prepared from natural resource waste was successfully added into the high impact acrylic resin. It seemed that adding 1 wt.% and 3 wt.% silica particles. The most beneficial effects were obtained by improving the mechanical properties of high impact acrylic dentures.
Graphical Abstract
Keywords
Introduction
Poly(methylmethacrylate) most widely used materials in prosthetic dentistry is (PMMA). Its inexpensive cost, straightforward manipulation method, and outstanding aesthetic results are all strengths. Dentures made of the PMMA materials are less expensive and bulky and they may be easily altered and fixed as needed. The most common complaint of denture users that doctors hear is a broken denture. It may be caused by the denture base prosthesis becoming too stiff over time as a result of the long-term failure brought on by repeated masticatory pressures or by over load oral stresses produced by inadvertent prosthesis prognosis [1]. When used for an extended period of time in the oral environment, the dental prosthesis comes into the direct contact with the oral mucosa, thus the dental materials should have high performance and good biological activity [2]. Research reveals that 67% of dentures suffer damage within a few years of being made [3]. Recently using nanotechnology, many materials with superior mechanical and physical properties have been created. Superior and custom-made materials can be created by adding nanoparticles to base materials to create composite materials with distinct physical and mechanical properties that cannot be achieved with the base material alone. This cutting-edge technology is used to meet the needs of base materials intended for use [4]. Nanomaterials are special because of their small size, large surface area to volume ratio, and atomic level characteristics [5]. The denture base composite with nanofillers has stronger interfacial cross-links between the resin bulk and nanomaterials than the conventional resin bulk. By protecting the nanomaterials and creating a dense interface, this improved molecular bonding strengthens the links between the resins, makes polymers denser and raises their molecular weight [6]. PMMA and other polymeric dental materials have been successfully combined with amorphous crystalline silica derived from rice husk consider as nanoparticles. The mechanical and thermal properties of these materials have been significantly impacted by several experimental studies [7]. Reduced mechanical characteristics, however, can occur when nanoparticles are loaded at an inappropriate type or dose [8]. Natural nanoparticle silica powder has been chosen to improving the qualities of the PMMA material because it is biocompatible and has a high level of fracture resistance [9]. Recently, there has been interesting in using inorganic nanoparticles to improve polymer performance. Due to its mechanical and optical characteristics, silica-poly methyl methacrylate is the most studied nano-composite material [10]. Composites made of polymers produced from RHA have the potential to create new hybrid organic-inorganic materials with unique properties. As a result, the rice husk-derived material has big potential to be used as a low-cost precursor in the manufacture of highly valuable amorphous silica-based products for industrial and medical uses. Silica nanoparticle has biomedical application. Biofilms are created when bacteria proliferate and attach to the surfaces of biomaterials, the bacterial cells are shielded by the biofilm growth phase from the host defense mechanism and antibiotic, its biocompatibility characteristics have been extensively used in biomedical research [11]. Nowadays, along with the growing demand of mankind for new nanotechnologies, chemical nanotechnologies are also developing rapidly. Therefore, in the field of nanochemistry, the scientific literature pays great attention to the development of nanotechnologies and their application is of high significance in various fields of human activity, by "nanoscience", we mean the branch of science concerned with the characterization and study of nanomaterials in addition to determine their physical and chemical properties between (1-100 nanometers) [12]. The goal of this study was to see how different biological silica concentrations derived from rice husk affected a few mechanical properties of a high impact strength heat cure acrylic denture base.
Materials and Methods
Silica preparation from rice husk
Following milling, stainless steel mesh (500 um pore size) was used to filter the Iraqi rice husk. After thoroughly rinsing with deionized water and mechanically stirring for one hour, the rice husk was dried in a dry heat oven at 70 °C. To reduce metallic contamination, the rice husk was then treated for 2 hours at 90 °C with 1 M hydrochloric acid as a leaching agent, followed by rinsing with deionized water to achieve neutral ph. After that, the mixture was calcined by heating it to 700 °C for two hours. Following calcination, 1 g of rice husk ash was added to a glass beaker with 20 mL of 1.5 M sodium hydroxide and heated to 90°C for an hour to produce sodium silicate solution. After that, the sodium silicate was dissolved in 250 mL of pure ethanol and then diluted with 1000 mL of water for 10 minutes. The solution was gently titrated with 3 M orthophosphoric acid until a yellowish gel formed at pH neutralization. The gel was centrifuged for 15 minutes at 4000 rpm after being washed with warm distilled water to remove any remaining sodium phosphate or sodium silicate. The gel was then dried for two hours at 90 °C before being calcined in a furnace for 30 minutes at 550 °C to produce silica powder [13, 14]. By measuring the transverse strength, impact strength, and surface hardness of acrylic after adding natural silica. A pilot study was conducted to determine the most effective and dependable concentration of rice husk-derived silica. For each test, ten samples were made, with two concentrations of natural silica particles (1% and 3% by weight) and a control group (0%).
Sample's preparation
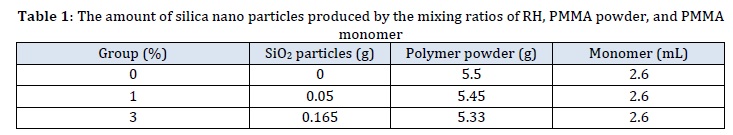
Cutting was done with a laser cutting machine. The plastic models based on the desired dimensions required for each test. To conduct tests on transverse strength and surface hardness, bar-shaped samples were created in accordance with American Dental Association Specification Number 12, measuring 65 mm in length, 10 mm in width, and 2.5 mm in thickness. In contrast, the bar-shaped specimens measuring 80×10×4 mm in length, width, and thickness were prepared for an impact strength test in accordance with ISO 179-1, 2000 [14]. These sample made of plastic disks were set into lower part of the dental flask, which was already fill with dental stone that had been recently mixed according to the manufacturer's instructions (W/P ratio of 25 mL/100 g). The excess stone material was removed and smoothed. After the stone had hardened completely, the plastic patterns and stone surface in the flask were coated with a thin layer of separating medium and allowed to dry. The upper half of the dental flask was then placed over the lower half, completely filled with dental stone (while vibrating to remove any incorporated air bubbles), and its lid was placed on top. Once the second layer of the stone had fully hardened, the flask was opened and the plastic disks were removed, leaving a hole in the stone. By combining acrylic liquid and powder according to the instructions of manufacture, control acrylic samples were created, while the RH-derived silica particles incorporated samples were prepared by measuring the amount of silica required and subtract it from acrylic powder weight use electronic balance (with accuracy of 0.0000), and then introduced to the PMMA monomer and blended with a probe sonication instrument to separate them into individual particles after three minutes of vibration at 120W and 60 KHz [15]. To avoid particle aggregation, the resulting silica-PMMA monomer suspension made with RH was combined as soon as feasible with PMMA powder, as provided in Table 1.
For the curing process, the clamped flasks were placed in a thermostatically controlled water bath at room temperature, after which the temperature was gradually increased to 70 °C for 30 minutes at this temperature in the water bath, and then gradually increased to 100 °C for 30 minutes at this temperature in the water bath. The flask was removed from the water route when the curing process was complete, and it was permitted to gradually cool at bench until it reached to room temperature. After that, the flask was opened and the acrylic samples were carefully removed from the molds. Each sample was wet ground after deflasking using silicon carbide paper with grit sizes of 120, 320, 400, and 600 in turn on a lapidary wheel following that, polishing was done with a wheel rug and pumice along with water at a speed of 1500 rpm to avoid overheating, which could have warped the samples. Prior to the test methods, after being stored in distilled water, all species were incubated for 48 hours at 37 °C [16].
Particle size analysis for RH-derived silica powder
To determine the size of the particles by measuring their diameters, RH-derived silica powder was examined by a particle size analyzer. This experiment was carried out in Iraq's Technology University, a hub for cutting-edge research in nanotechnology. The analysis showed that the RH-derived silica had particles as small as 575 nanometers.
Fourier transform infra-red (FT-IR) spectroscopy
A small amount of control and experimental samples were scratched and characterized by FT-IR spectrophotometer analysis (Shimadzu IRAffinity-1 japan) to recognize the existence of inorganic and organic substances in the sample by measuring the infrared spectrum's transmission or absorption.
Field emission scanning electron microscope (FE-SEM)
FE-SEM (INSPECT F50 FEI company Netherland) was utilized to scan the natural prepared silica powder and also the fractured surfaces of the control and experimental samples (1 wt%. and 3 wt.% RH-derived silica) were scanned to determine the distribution, size, and shape of MCC particles inside the acrylic matrix. The samples were made by cutting small pieces of acrylic and coating them with gold using a sputter coater device for 2 minutes. Then, the samples were mounted on the device pin stubs and examined.
Transverse strength test
Using a computer-controlled universal testing machine, the transverse strength of each sample was evaluated using a three-point bending test (Laryee technology Co., Ltd., China). The thirty acrylic samples were prepared and set on the bending fixture, which consisted of two parallel supporting arms spaced about 50 mm apart. The load's maximum was 50 kg, and it was applied via a rod placed in the middle of the sample at a cross-head speed of 1 mm per minute until fracture occurred. The transverse strength values were calculated using the following equation:

Where, T is the transverse strength (N/mm2), P is the maximum load applied to the samples (N), L is the distance between the supporting arms (mm), b is the sample width (mm), and d is the sample depth (mm) (ADA specification No.12, 1999) [17, 18].
Impact strength test
An impact strength test was performed in accordance with ISO 179-1 using a Charpy impact tester (Testing Machines Inc., USA) (2000). The thirty samples were set up horizontally at both ends and struck in the center by a 2 Joules free-swinging pendulum. The scale measures the amount of impact energy absorbed by the samples in joules. Using the following formula the impact strength was measured in kilo-joules per square meter in accordance with ISO No. 179, 2000, impact strength is calculated as follows:

Where, E is impact energy in Joules, b is sample width in millimeters, and d is sample thickness in millimeters.
Surface hardness test
The effect of silica addition on acrylic surface micro geometry was assessed using a portable digital roughness tester (Profilometer equipment, Mahr Federal, Germany). The stylus on this gadget, which is used to sketch the contour of surface imperfections, is a sharp, perceptible diamond needle. The test was carried out as instructed by the profilometer. The stylus made contact with three uniform, equal locations to provide three readings for each sample. In each area, the stylus makes 5 mm of contact. When the stylus was allowed to touch the first area of the sample, which was set up on a firm and stiff surface, the reading on the digital scale appeared on its own. Finally, a roughness value was determined by averaging the three values [14].
Results and Discussion
Pilot study
The findings of the impact strength test, surface hardness test, and transverse tests, which were all a part of the pilot study, are presented in Table 2.
According to the pilot study results, 1 wt.% and 3 wt.% of silica derived from rice husk were selected as the concentrations used in this investigation because they clearly improved transverse strength, impact strength, and surface hardness.
Fourier transform infra-red (FT-IR)
The infrared spectrum of Rice husk derived silica in Figure 1 showed the distinctive bands at 1009, 933, and 801 cm-1 which are credited to the stretching vibration of asymmetric Si-O-Si, Si-OH, and symmetric Si-O-Si, respectively. Furthermore, the measurement showed a band at 489 cm-1 which could be attributed rocking-type vibration. Moreover, the spectrum gave two bands at 3471 and 1631 cm-1 which are attributed the stretching and bending vibrations of hydroxyl groups in water adsorbed on the silica surface, confirming the nano-size effect, as presented in Table 3.


The infrared spectrum of high impact heat cure acrylic in Figure 2A showed a broad, high intensity main band at 3441 cm-1 which indicates the O-H stretching vibration in the COOH group or that of the crystallization water. Moreover, the FT-IR spectrum showed two high intensity bands at 2924 cm-1 and 2850 cm-1, which are credited to the stretching vibration of the methylene and methane groups. The spectrum also revealed the main characteristic band of poly acrylic at 1739 cm-1 which is attributed to the stretching vibration of C=O group, while the band at 1585 cm-1 is attributed to the stretching vibration of C=C group. Furthermore, the FT-IR spectrum revealed a medium intensity band at 1431 cm-1 which could be attributed to C-O stretching vibration, while the band at 1126 cm-1 is due to OH out of plane vibration, as presented in Table 3. Figure 2B revealed the infrared spectrum of high impact heat cure acrylic and rice husk-derived silica composite. The following bands at 3441, 3005, (2947, 2843), 1732, 1481, 1450, and 1280 cm-1 attributed to O-H stretching vibration in COOH, =C-H, C-H, C=O, C=C, C-O, and OH out of plane vibration, respectively. These bands confirm the presence of acrylic and the slight shift of these bands compared with the main material without adding silica is evidence of the success of adding silica. Furthermore, the spectrum showed the characteristic bands of SiO2 nanoparticles at 752, 983, and 1134 cm-1 attributed to the stretching vibrations of symmetric Si-O-Si, Si-OH and asymmetric Si-O-Si, respectively as listed in Table 3. It is worth noting that by increasing the percentage of silica, it was found that the intensity of the bands decreases, confirming the validity of the results obtained, as silica causes a decrease in the ability to vibrate the bonds and reduce their freedom of movement.
Field emission scanning electron microscope (FE-SEM)
The FE-SEM analysis of the cracked surfaces of the experimental (1 wt.% and 3 wt.% silica) and control groups is depicted in Figures 3. The acrylic matrix's consistent distribution was shown by the results.
Transverse strength test
According to the transverse strength test findings, the 1 wt.% RH-derived silica group had the greatest mean value, followed by the 0 wt.% control group and the 3 wt.% RH-derived silica group, which had the lowest mean value (Figure 4).

The results of Table 4 show that the 1wt. % RH-derived silica had the highest mean transverse strength test value (101.76 N/mm2), followed by the control group with a mean value of (96.09 N/mm2), and the 3 wt.% RH-derived silica had the lowest mean transverse strength test value (90.14 N/mm2). The calculated p-value of the One-way (ANOVA) test was 0.001. This showed a statistically significant difference between the study groups.
Further statistical analysis was utilized by using Post hoc tests (LSD) to confirm the statistical differences among groups. The results showed that a significantly lower values in transvers strength of 3 wt.% RH-derived silica group compared with 1 wt.% RH-derived silica (P<0.001) and control groups (P=0.013), while a non-significantly difference between control group and 1 wt.% RH-derived silica group (P=0.017), as provided in Table 5.
Impact strength test
The impact strength test results showed that the mean value was higher related to 3wt.% RH-derived silica (14.47KJ/m2) followed by 1wt.%.RH-derived silica with mean value of (14.38 KJ/m2), while the mean value of impact strength was the lowest obtained with the control group (14.14 KJ/m2), as indicated in Figure 5. The calculated P-value of One-way (ANOVA) test was (0.76) revealed a statistically no significantly differences among the studied groups (P˂0.01), as presented in Table 6.
Surface hardness test
According to the results of the surface hardness test, the highest mean value was related to 3 wt.% RH-derived silica group, while the lowest mean value was related to the control group 0%, as depicted in Figure 6.
Both experimental groups (1 wt.% and 3 wt.% RH-derived silica) revealed a higher mean values compared with the control group. The statistical analysis using one-way ANOVA test revealed a non-significantly differences among three studied groups, as presented in Table 7.
An good organic biocompatible polymeric substance used to make denture bases is poly(methyl methacrylate, or PMMA). Since 1937, it has been the material of choice for making dentures due to its advantageous physical and mechanical characteristics [19]. After a few years, approximately 68% of acrylic resin dentures fail. This occurs when the denture is mistakenly dropped onto a hard surface or breaks as a result of the extreme chewing pressure [20]. To address these issues, numerous attempts were made to change and improve the impact strength, flexural strength, and the PMMA hardness. One of these projects involves changing the chemical composition of acrylic resin via copolymerization with rubber. Butadiene-styrene co-polymer with rubber phase has higher impact strength but higher cost and lower transverse strength [21]. The most common nanomaterial in this discipline is silica nano-powder because of its intriguing and distinctive features [22]. Because of their rigidity, denture base polymer can be reinforced with silica nanoparticles [23]. Silica materials that are traditionally manufactured are being thoroughly evaluated for potential applications in the environment and in medicine. Because of their chemical inertness, biocompatibility, and exceptional physicochemical properties, bio-source RHA containing silica micro particles may be appealing as low-cost precursors for several pharmaceutical industries, such as drug delivery systems [24].
Impact strength
The impact strength is an essential property of acrylic denture bases as it predicts the resistance of the denture to fracture if it is placed under high, short-term stress, such as during dropping [25]. The impact testing machine was used in this study to quickly and affordably assess a material's relative hardness [26]. In the current research, incorporation of 1wt.% and 3 wt.% of RH-derived silica revealed an improvement in impact strength values as opposed to the control group.

Statistically, there is no discernible difference in impact strength values among 1wt.% and 3 wt.% RH-derived silica groups and control group. The FE-SEM finding revealed a uniform distribution of RH-derived silica particles as opposed to that, the large surface area, small size, and low percentage of RH-derived silica help so as to that the fillers are dispersed uniformly over the resin matrix, which has a favorable impact on the impact strength as explained by Gupta et al. [27]. Likewise, this result could be explained by the PMMA particles' gaps and pores having a micro-filling effect [28]. The findings of this study were consistent with the previous study made by Salman et al. [29]. in which the SiO2 powder were add to poly methyl methacrylate powder by three concentration (3%, 5%, and 7% by weight) with the addition of SiO2 powder to (PMMA) at the percentage of 3 wt.% and 5 wt.% SiO2 nano particles a highly significant increase in impact strength and transverse strength was observed, while at the percentage of 7 wt.% SiO2 nano particles a significant reduction occurred in both impact and transverse strength sample’s tests. This study's findings are in disagreement with [30], who revealed the highest impact strength values for the control group in comparison to 0.125 wt.% and 0.25 wt.% Sic particles. Perhaps as a result of nanoparticle agglomeration with high surface energies and a proclivity to adhere to one another.
Surface hardness
Surface hardness is a crucial property which concerning the material wear that may happen during routine denture brushing, creating roughness, and promoting microbiological adherence [31]. In the current study, a Shore D hardness tester appropriate for poly methyl methacrylate material was used. The Shore durometer type (D) hardness tester eliminates the issue of the elastic recovery because it measures the depth of the loaded indentation under loading conditions [32]. Surface hardness values were not significantly higher with addition of 1 wt.% and 3 wt.% of RH-derived silica compared with the control group; this improvement in hardness due to the strong abrasion resistance of silica Nano-composite. The hardness increase is concentration dependent, which means that a low concentration of silica nanoparticles (1 wt.%) compared with the control sample will result in a minor increase in hardness. This is because to the low network density, while an increase in silica nanoparticle concentration (3 wt.%) is linked to an increase in hardness values. According to Hudson et al., [33]. this observation might be explained by an increase in the accumulation of hard material particles in the acrylic resin matrix. Perhaps the random distribution of tough nano-SiO2 particles within the acrylic resin and on the surface of the samples is what is responsible for the hardness increase. The nano-SiO2 particles act to decrease the plasticizing effect and reduce water absorption by filling of inter polymeric spaces, led to improve the hardness as explained by Salman et al. [29]. The finding of present study is harmonious with Gad et al. [34]. who believe that the surface hardness might enhance following incorporation of different concentrations of nano-SiO2. In addition, findings of this study are consistent with a previous study by Safi & Moudhaffar [35], who found that adding different concentrations of SiO2 particles 3wt.%, 5 wt.%, and 7 wt.% increased the hardness in heat-curable poly(methyl methacrylate) denture base material, as compared with the control group.
Transverse strength
One mechanical strength test that is helpful in assessing the effectiveness of denture base materials is transverse strength test, because it represents the stress placed on the denture while chewing [36]. Compressive, tensile and shear strengths are combined to create transverse (flexural) strength, which is a measure of a material's resistance to fracture [37]. The transverse strength values in the current investigation significantly increased with the addition of 1 % by weight of RH-derived silica, while the transverse strength values in the 3 wt.% RH-derived silica group were lower significantly than in the control group. However, there is a statistically significant difference between the 1 weight percent, 3 % by weight and control group. In addition, the voids content was found to be higher due to the higher concentrations of nano-filler aggregate, resulting in inappropriate matrix distribution, as suggested by Kumar et al. [38]. The decrease in transverse strength depends on the particles distribution resulted in decrease in flexural strength. As stated by Balkees et al., [39] the silica nanoparticles may also function as contaminants during the reaction and reduce the degree of polymerization, which causes more unreacted monomer to remain within the matrix and lowers the values for flexural strength. The distance between particles will shrink as silica nanoparticle concentration rises, increasing the possibility of agglomeration. According to Jiangkongkho et al. [40] who discovered that a decrease in flexural strength was associated with an increase in nanoparticle concentration, agglomeration at the agglomerated region creates stress concentration, which reduces the mechanical properties.
Conclusion
Silica derive from Iraqi rice husk was successfully incorporated into the high impact heat-cured acrylic resin and resulted in the best improvement of the mechanical properties of the high impact heat cured acrylic (transverse strength, impact strength, and surface hardness) after the addition of 1 wt. % silica derive from rice husk of experimental group, as compared with the control group. Recently, there has been a lot of interest in byproduct and waste management for the creation of new products. The RH byproducts are regarded as renewable and sustainable silica supplies in this field because they contain significant amounts of amorphous silica. As a result, RHA has a lot of potential as a silica substitute in the production of various composites.
Acknowledgements
The authors would like to present their sincere gratitude to the University of Baghdad's College of Dentistry and Technology University, an Iraqi center for advanced research in nanotechnology, for its assistance.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Authors' contributions
All authors contributed to data analysis, drafting, and revising of the paper and agreed to be responsible for all the aspects of this work.
Conflict of Interest
The author declared that they have no conflict of interest.
ORCID
Azza walaaldeen khairi
https://www.orcid.org/0000-0001-6351-6424
HOW TO CITE THIS ARTICLE
Azza walaaldeen khairi, Ghassan Abdul-Hamid Naji. The Effect of Silica Particles Derived from Rice Husk on Some Mechanical Properties of a High Impact Heat Cure Acrylic Resin. J. Med. Chem. Sci., 2023, 6(7) 1646-1659


