Document Type : Original Article
Authors
Department of Biotechnology, Science College, University of Baghdad, Baghdad, Iraq
Abstract
This study aimed to demonstrate the biosynthetic process of iron oxide nanoparticles (Fe2O3-NPs) by using extracellular components produced by environmental isolates of Lactobacillus plantarum as reducing and stabilizing agents studied in the laboratory of the university of Baghdad. Add 1 g of iron sulfate to 10 mL of extracellular nanoparticles for synthesis. Biosynthetic Fe2O3 nanoparticles have broad application prospects in catalysis, biosensing, anticancer, and biomedicine. Optimal conditions for synthesizing Fe2O3 were investigated using UV-VIS, AFM, XRD, FT-IR, and FE-SEM techniques. The UV-VIS wavelength of NPs within the nanoclusters. Susceptibility testing of P. aeruginosa showed resistance to tetracycline, trimethoprim-sulfamethoxazole, ceftazidime, and chloramphenicol, whereas sensitive to Amikacin, Norfloxacin, Meropenem, and Ciprofloxacin and the effect of Fe2O3 NPs from extracellular component on bacteria Pseudomonas aeruginosa on an inhibition zone 18 mm.
Graphical Abstract
Keywords
- Fe2O3 NPs
- Antimicrobial activity
- Biosynthesis nanoparticles
- Pseudomonas aeruginosa
- Lactobacillus plantarum
- Susceptibility test
- Extracellular
Main Subjects
Introduction
The category of lactobacilli known as facultatively heterofermentative includes L. plantarum. It is a diverse and adaptable species found in a wide range of environmental niches, such as dairy, meat, fish, and numerous plant or vegetable fermentations. Numerous types of cheese have also been reported to contain L.plantarum strains. In addition, L. plantarum strains have a history of successfully colonizing the intestines of mammals, including humans [1]. Lactobacillus plantarum can be distinguished from other bacteria because of its characteristic of Extracellular and Intracellular products; Extracellular and Intracellular are components composed of (Enzymes, proteins, polysaccharides, peptides, pigments, toxins, etc.) [2]. These ingredients act as reducing agents and bioprotectants against various stressors, such as phage attacks, toxic metal ions, and desiccation. These components reduce agents and biological defenses against various stressors such as phage infection, toxic metal ions and desiccation [3]. Iron particles that are smaller than a micron are known as nanoscale iron particles. They have a large surface area, which makes them very reactive. They quickly oxidize to produce free iron ions when oxygen and water are present. They are extensively employed in medical and laboratory applications, and their potential for cleaning up industrial areas contaminated with chlorinated organic chemicals has also been investigated [4]. Pseudomonas is a common rod-shaped gram-negative enveloped bacteria that can harm humans, other animals, and plants. Positive for oxidase, catalase, and citrate [5]. P. aeruginosa, a species of major medical significance, is an opportunistic bacterium resistant to several medicines and disinfectants that causes severe acute and chronic nosocomial infections in immunocompromised, catheterized, or burn patients [6]. Biofilms are created when bacteria proliferate and attach to the surfaces of biomaterials. The bacterial cells are shielded by the biofilm growth phase from the host defense mechanism and antibiotic [7]. Because of its multitude of scientific and technical uses, including biosensors, iron oxide has attracted particular interest such as antimicrobial activity [8, 9]. Additionally, its biocompatibility and magnetic characteristics have been extensively used in biomedical research [10].
Materials and Methods
Species by taking samples of Lactobacillus spp were isolated from a fermented food product. Isolates were cultured in MRS broth, By the manufacturer's instructions [11]. All bacterial species isolate was identified via conventional biochemical assays and procedures through a VITEK2 system.
Extracellular production
Lactobacillus plantarum isolates were grown in MRS broth, incubated at 28 °C for 48 hours, and centrifuged at 8000 rpm for 10 minutes to collect the extracellular solution [12].
Synthesis of iron oxide nanoparticles from the extracellular space
Fe2O3 nanoparticles were synthesized by a modified biosynthetic method (biological synthesis method) using iron sulfate Fe2(SO4)3 (India) to generate iron oxide nanoparticles from extracellular for Lactobacillus SPP. [13]. Typical procedure: Dissolve 5 g of ferric sulfate Fe2(SO4)3 in 50 mL of Lactobacillus SPP extracellular product solution, dispersed by an ultrasonication bath for 10 min to more mix component and kept in the dark condition overnight on the shaker. The resulting solution was centrifuged at 8000 rpm for 10 minutes and washed twice with deionized distilled water to remove residual extracellular cells. It was then dried in an oven at 40°C overnight to obtain a brown powder and stored in the dark for later use.
Pseudomonas aeruginosa isolate
The clinical specimens of P.aeruginosa collection are 121 clinical isolates and took place from burns and wounds from November 2021 to January 2022. It is important to note that the samples contain burns and wounds. Under the specific condition, Cetrimide agar prepared according to the manufacturer was used to streak specimens taken from the hospital (Al Shula Teaching, Al-Kadmia Teaching, and Baghdad/Medical City) [14]. Other identification tests included biochemical and morphological characteristics, and the Vitek-2 system was performed [15].
Antibacterial activity of Fe2O3 Nanoparticles
Pseudomonas aeruginosa was cultured on Muller-Hinton agar and using the good method (MIC) of microbial inhibition by using Fe2O3 NPs synthesis from extracellular sources [16].
Detection of Biofilm (quantitative method)
After incubating the P. aeruginosa isolates in BHI medium at 37 °C for 24 hours, added (200 μl) of P.aeurginosa broth was to each well in a microtiter plate, and then (200 μl) of BHI medium was added to each well in a microtiter plate. After the incubation period, each well was washed three times with phosphate-buffered saline. Plates were then air-dried at room temperature for 15 minutes before adding 99% methanol (200 µL) over 15 minutes. Plates were then air-dried at room temperature for 15 minutes and stained with 200 μl of 2% crystal violet for approximately 15 minutes. After completely removing the stain, add 200 µL of 95% ethanol to each stained well and incubate at room temperature for 15 minutes. The optical density of the plate’s wells was measured using a micro-ELISA auto reader at 520nm. Then they used Sterile BHI broth as a negative control of the test. To compensate for background absorbance, the mean of (OD) is optical density, and the reading value of the control mean (C) was taken from the test (T) values [17]. Table 1 measures the intensity of biofilm:

Results and Discussion
Bacterial isolation in culture media
Lactobacilli samples were obtained from various sources, including dairy products (milk, buffalo, cheese, yogurt) [18]. The samples were cultured on MRS agar plates as selective media for isolation and incubated at 28 °C for 48 hours with the presence of (3-5 %) CO2 using Candle Jar. These bacteria can be examined using morphology, microscopy, and biochemistry testing to validate their identity (Figure 1).
Antibiotic susceptibility test of P.aeruginosa
Of the 121 isolates, only 63 were tested for susceptibility to 12 different antibiotics using the disc diffusion method recommended by the Clinical and Laboratory Standards Institute (CLSI, 2021) [19]. guidelines and the results shown in Figure 2 showed varying levels of resistance; in this study, P.aeruginosa showed the highest percent of resistance to tetracycline, followed by Trimethoprim-Sulfamethoxazole, Ceftazidime, and Chloramphenicol.
Absorption maxima were detected in extracellular applications using a UV-Vis spectrophotometer (Shimadzu, Japan) developed from Lactobacillus plantarum [20]. Figure 3 Shows extracellular absorption at 289 nm.
Characterization of biological synthesis Fe2O3 NPs
UV–VIs spectral analysis Fe2O3 from extracellular
The optical properties of the nanoparticles were studied by exploiting ultraviolet – Visible spectrometer. Figure 4 shows the absorbance of the sample in the nano range at room temperature. It has shown a peak at 290 nm wavelength was thus agreed with [21].

Figure 1: Lactobacillus plantarum cultured on MRS agar

Figure 2: The results of antibiotic susceptibility test of P.aeruginosa
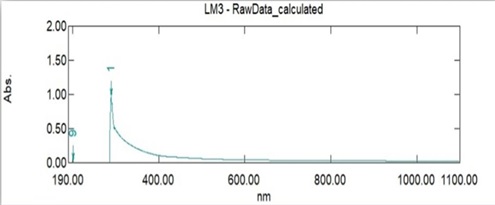
Figure 3: The UV-VIs of extracellular

Figure 4: The UV-VIS of Fe2O3 nanoparticles synthesis using extracellular
Atomic force microscopy (AFM) analysis of Fe2O3 from extracellular
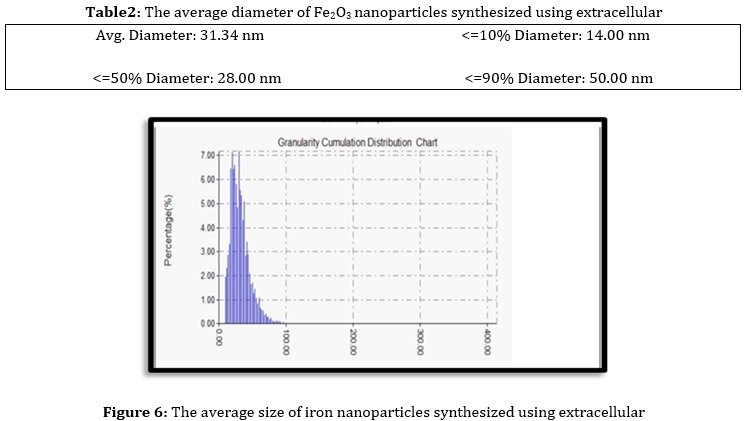
The surface shape formation of the Fe2O3 NPs was studied by atomic force microscopy to show that Fe2O3 NPs 2D and 3D (Figure 5) [22]. AFM images show that the biosynthesized Fe2O3 NPs are spherical. The size of an average diameter of 31.34 nm (Table 2) was also measured by AFM (Figure 6).

Figure 5: Atomic force microscopy (AFM) of Fe2O3 NPs synthesized using extracellular illustrate2D and 3D topological
Fourier transform infrared (FT-IR) analysis of Fe2O3 from extracellular
FT-IR spectrum has determined the functional groups of nanoparticles. Figure 7 represents the absorption spectrum of biologically synthesized nanoparticles in FT-IR. An intense peak at 3398.34 cm-1 was visible due to OH stretching mode. The peak properties at 1629.74 cm-1 suggested the presence of crystallographic H2O molecules, i.e., O–H bend. The wide peak at 455,17 cm-1 and 572,82 cm-1, respectively, represented the Fe–O band and Fe–O–Fe skeletal frequency [13].
X-Ray diffractometer analysis of Fe2O3 from extracellular
The X-ray deviation examination was performed to define the surface Morphology and the composition of the crystals Of Fe2O3 NPs, including crystalline size, lattice parameters, the thickness of the crystal, and textile factor. X-ray bias results for the Fe2O3 model calibrated to typical data from the Joint Commission Powder Diffraction Standard (JCPDS) #11-0614. X-ray diffraction test results for Fe2O3 NPs include declination and declination data format. The bias model was created using Origin 9.0 for Windows operating system (Figure 8). Deviation patterns are: It has seven diffraction peaks. The seven diffraction peaks represent electron diffraction processes occurring in the Fe2O3 nanoparticle diffraction planes, namely fields (220), (311), (400), (422), (511), (440), and (533). The diffraction pattern characteristic of Fe3O4 nanoparticles is according to JCPDS-11-0614 standard data [23, 24]. Similar to nanomaterials, particle size decreases with increasing peak width. Crystal structure parameters were obtained at b=0.3785 nm and c=0.9513 nm. The average crystallite size was calculated using the Debye-Scherrer equation [25]. The results show that the crystallographic planes (θ = diffraction angle) of Fe2O3 are 30.10°, 35.51°, 45.21°, 53, 44° and 57.31°, respectively, with six obvious strong diffraction peaks. 2θ and 62.81° are observed.
Field emission scanning electron microscopy analysis of Fe2O3 from extracellular
Through applying FE-SEM, images were taken of the sample at A magnification of 50kx. Focused on Figure 9. the whole sample has soft planes and a uniform shape in the form of Fe2O3 nanocluster centers [13].
Antibacterial susceptibility test
Iron oxide NPs antibacterial activity was investigated using Gram-negative bacteria (Pseudomonas aeruginosa). The minimum inhibitory concentration (MIC) of Fe2O3 NPs against microorganisms was calculated using the agar well diffusion method [26, 27].
Pour approximately 25 mL of sterile Mueller Hinton Agar into a sterile plate and let it to come to room temperature. Wells were prepared by transferring the growth of the test species onto agar and isolating the wells by pinching a sterile cotton swab. Then different ratios of Fe2O3 NPs from extracellular (500, 250, 125, 62.5, 31.25, 15.625, 7.8125, 3.90625, 1.953125) mg/mL. Plates seeded with Fe2O3 NPs were incubated at 37 °C for 24 hours. Assess the zone of inhibition around the well after incubation [13]. Results of Fe2O3 NPs from extracellular antibacterial activity were demonstrated in (Figure 10). The antibacterial activity was found to be directly dependent upon the Fe2O3 NPs concentration. Table 3 shows that at a concentration of 500 mg/mL Fe2O3-NPs from extracellular material, the maximum inhibition zone of P. aeruginosa was 18 mm, while the minimum inhibition zone was 15.625 mg/mL Fe2O3 NPs concentration. The difference in inhibitory diameter may be due to the different interactions between Fe2O3 NPs and microorganisms and the different susceptibility of the bacteria used in the current study. The main mechanism of toxicity of Fe2O3 NPs potentially associated with metal oxides carries the positive charge even though the microorganisms bear negative charges; this results in electromagnetic interaction between microorganisms and metal oxides, leading to oxidation and, finally, death of microorganisms. Extracellular Fe2O3 NPs were found to have MICs ranging from 500 to 1.953125 mg/mL by serial dilution methods described in CLSI [13]. The bactericidal action of Fe2O3 nanoparticles on bacteria is of extreme importance due to the ability of pathogenic bacteria to join the ecosystem’s food chain [28]. The antibacterial effect of Fe2O3 on bacteria was confirmed [28] and communicated in modern research.

Figure 7: FT-IR image of Fe2O3 NPs synthesized using extracellular (A) Fe2(SO4)3, (B) extracellular, (C) Fe2(SO4)3-extracellular, (D) Fe2O3NPs

Figure 8: XRD Pattern of Fe2O3 NPs synthesized using extracellular

Figure 9: FE-SEM images of Fe2O3 NPs synthesized using extracellular
Detection of biofilm production by microtiter plate assay
Under the same experimental conditions, all 20 Pseudomonas aeruginosa isolates showed different potential biofilm-forming abilities (Figure 11). Separation results were limited to three groups. Six isolates were high biofilm producers (30%), seven isolates were intermediate biofilm producers (35%), and seven isolates were low biofilm producers (35%). These results were obtained after 24 hours of incubation of Pseudomonas aeruginosa growth and biofilm formation.

Figure 10: Antibacterial activity of Fe2O3 NPs from extracellular on P.aeruginosa


Figure 11: biofilm formation of Pseudomonas aeruginosa after 24 h and 48 h
After 48 hours of incubation, 20 isolates were tested in microtiter plates. It shows that 15 isolates (75%) are strong biofilm producers, five isolates (25%) are moderate biofilm producers and 0 isolates (0%) are weak biofilm producers.
Conclusion
This study demonstrated the biosynthesis of iron oxide nanoparticles using extracellular as a reducing agent. Additionally, the attained Fe2O3 NPs were characterized using UV-Vis, AFM, XRD, FT-IR, and FE-SEM techniques. In particular, The XRD patterns showed the successful Fe2O3 NPs phase formation.
At the same time, the FE-SEM demonstrated that the prepared Fe2O3 NP exhibited spherical particles and plate-like structures with an average diameter size ranging between 30-50 nm. In contrast, the AFM revealed an average diameter of 31.34 nm. The antibacterial activity test found that the bio-synthesized has a strong antibacterial activity against the introduced bacteria. The maximum inhibition zone was found to be 18 mm at a concentration of 500 mg/Ml.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Authors' contributions
All authors contributed to data analysis, drafting, and revising of the paper and agreed to be responsible for all the aspects of this work.
Conflict of Interest
The author declared that they have no conflict of interest.
HOW TO CITE THIS ARTICLE
Maha A. Qasim, Laith Ahmad Yaaqoob. Evaluation of Antibacterial Activity of Iron Oxide Nanoparticles Synthesis by Extracellular Lactobacillus against Pseudomonas Aeruginosa. J. Med. Chem. Sci., 2023, 6(5) 1100-1111


