Document Type : Original Article
Authors
1 Ministry of Science and Technology, Department of Materials Research, Baghdad, Iraq
2 Department of Chemistry, College of Science for Women, University of Baghdad, Baghdad, Iraq
Abstract
The current study aimed to evaluate the role and gene target of miR-126 in Iraqi patients with Coronary Artery Disease (CAD) and atherosclerosis (AS). By coverage average age (35 to 65) years old of both genders, a total of 40 patients with CAD and 40 healthy people as a control group were enrolled in this clinical study. The blood samples of patients were taken from Ibn Al-Bitar Diagnostic Center for Heart Surgery in Baghdad/Iraq in December 2020 to April 2021. Overall, the CAD patients had significantly higher (except HDL) total lipid profile (total chol., total Trig., LDL, and VLDL, age, and IL6 levels). Statistically no significant correlation was found between miRNA-126 and lipid parameters in all studied subject. The correlation was found between Triglycerides and his-Troponin (r=-0.521, pv=0.032). The ROC analysis of mRNA126 at UAC 0.976 had sensitivity of (0.80) and 1-specificity of (0.60) were total area under the curve = 759, pv=0.005; that means miRN-126 can be used as a screening test for CAD. Thus, miR126 can be considered as a biomarker on CAD in patient.
Graphical Abstract
Keywords
Main Subjects
Introduction
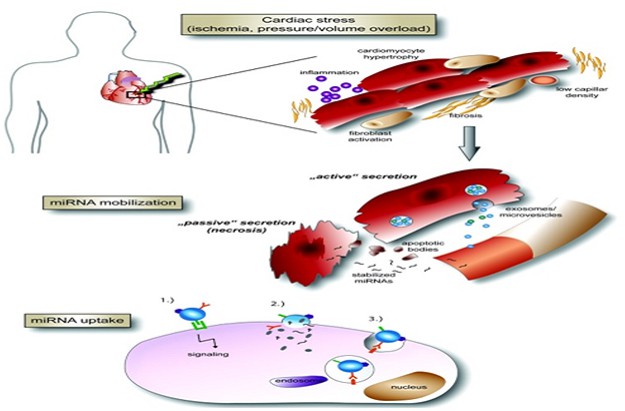
Coronary Artery Disease (CAD) is a type of human cardiovascular disease resulted from thrombosis or acute coronary atherosclerosis leading to sudden cardiac death, CAD is the greatest popular form of cardiac disease [1]. Atherosclerosis (AS) is an inflammatory condition of the arteries marked by the development of atherosclerotic plaques causing CAD. These plaques cause the artery's lumen to constrict, which can eventually result in the production of thrombi and total obstruction of the lumen [2]. MicroRNA (miRNA) families are powerful regulators in CAD among other various diseases [3], as a main target of the current study having 18–22 nucleotides in single-stranded nсRNAs binding to the 3′ -untranslated region of specific messenger RNA leading a decreasing in protein expression [4] because the particular miRNAs may be absorbed by the cells causing AS, these miRNAs can be considered as diagnostic biomarkers of AS [5] through whole blood, serum, or plasma. All related articles specified microRNAs linked to the particular molecular pathways that guide to AS, arterial remodeling, endothelial dysfunction, monocyte activation, arterial wall invasion, platelet activation, and vascular smooth muscle cell activation [6-8].
MiRNA-126 is abundant in endothelial cell may be treated as a biomarker for the CAD severity and complexity [9] with vascular integrity, angiogenesis, and wound healing. It exhibits several actions like atheroprotective, suppressing the inflammatory cascade, and mediating leukocyte adhesion in AS by reducing vascular cell adhesion expression [10, 11]. Low-density lipoprotein cholesterol (LDL-C) plays role in the progression and development of AS. Two forms of low-density lipoprotein cholesterol (LDL-C) controlled by miR-126 especially cholesterol metabolism of LDL-C [12] and regulated high-density lipoprotein (HDL) metabolism and reverse cholesterol transport with direct targeting of cellular cholesterol efflux [13, 14].
IL-6 plays role in the development and maintenance of the inflammatory response in AS, pro-inflammatory, and immune-regulatory cytokine [15, 16]. CAD-related miRNAs (miR126) are considered to be inflammatory-related miRNA influenced the inflammatory cytokines to coordinate the inflammatory processes [17]. Both cytokines and miRNA are elements of the signaling pathways that promote disease progression [18]. Also, higher sensitivity troponin (hs-Tn) levels were associated with multiple abnormalities of cardiac structure and function [19].
Therefore, this research aimed to study all of the above regulation elements and suggested them as good indictors of CAD in both genders of Iraqi patients compared with miR126.
Materials and Methods
Characteristics of patients
This study was carried out in Ibn Al-Bitar Hospital for Heart Surgery in Iraq from December 2020 to April 2021 for 8 patients of both sexes whose average age between 35-65 years old with CAD and 40 healthy people as a control group who have the same average age 35-65 year old. All patients were clinically diagnosed by the hospital doctor.
Ethical considerations of this study
The volunteers were aware of the research, their names were not revealed to anyone, their findings were unharmed by their participation, and the research was done in accordance with our scientific committee.
Anthropometric measurement
Body mass index (BMI) was determined by dividing the weight in kilograms by the height in square meters.
Exclusion criteria
Patients who have already had liver cirrhosis, alcoholism, tobacco use, hypertension, hyperthyroidism, hypothyroidism, leukemia, cancer, obese, kidney disease, and COVID-19 were excluded from this study.
Collection of blood samples
Serum was separated from blood collected from venous blood samples (10 mL) were collected of the studied subjects after fasting overnight. The serum is divided into three sections: a section for biochemistry measurements (lipid profile), ELISA (IL6), and high sensitive troponin (hs-Tn) measurements, respectively. Four Mls of the collected blood had been mixed with 0.5 mL of Trizole and kept under -70 °C to measure miRNA126.
Measurement of IL6
Enzyme Linked Immune Sorbent Assay (ELISA)/ sandwich technique were used for quantitative determination of Interleucken6, (IL-6). ELISA kits supplied by Shanghai Biological/China.
Assay of high sensitive troponin
An automated quantitative test that uses the ELFA method to determine the presence of human cardiac troponin in human serum or plasma on devices from the VIDAS family (Enzyme Linked Fluorescent Assay) [20].
Biochemical examination
Determine lipid profile
Serum lipid profile (Total cholesterol (TC), serum triglycerides (TG), high density lipoprotein (HDL), low density lipoprotein (LDL), and very low density lipoprotein (VLDL)) determination occurs by standard assays were used in hospital by villas. (TC, TG, and HDL) are supplied by Abbott/ Germany. The lipid profile assays were performed by using a multi-channel Abbott Spectrum auto-analyzer (Abbott Laboratories, C460276, USA) according to the manufacturer instructions as opposed to LDL, which was determined by using the Friedewald Equation. LDL = (TC-HDL-[TG/5]), and VLDL was calculated by dividing total serum TG by 5 [21].
Isolation of total RNA and real-time polymerase chain reaction (RT-PCR) analysis
-SYBR green quantitative real-time polymerase chain reaction assay (RT-PCR) by using the miScript SYBR Green PCR kit, was applied to quantify miRNA-126 (Qiagen, Hilden, Germany). Mature miRNA sequence and a particular forward PCR primer were the primers' sequences. (50CATTATTACTTTTGGTACGCG30) (50GGGGCATTATTACTTTTGG30).
The amplitude of miRNA expression change observed in patients in relation to control group was analyzed by the 22DDCt method [22].
RNA was isolated from the sample according to the protocol of TRIzol™ Reagent. For each tube, 0.4 mL of serum was added to 0.5 mL of TRIzol™ Reagent the lysate was homogenized. For each tube, 0.2 mL of chloroform was added to the lysate. All mixes were incubated, and then it was centrifuged for 10 minutes at 10.000 rpm. The mixture was separated into lower organic phase, interphase, and a colorless upper aqueous phase which containing the RNA.
Determination of RNA, cDNA yield fluorescence method [22]
QuantusFluorometer was used to detect the concentration of the extracted cDNA to detect the samples quality for downstream applications. For 1 µl of cDNA, 199 µl of diluted Quantifluor Dye was mixed. After 5 minute incubation at room temperature in dark place, cDNA concentration values were detected.
Gene expression by using pfaffi method [23] relative quantification:
Folding =2-ΔΔCT
ΔCT = CT gene - CT House Keeping gene
ΔΔCT = ΔCT Treated or Control - Average ΔCT Control.
Results and Discussion
Mean±SD, and the (P-value) to the result for gender, age, BMI, hs-Tn, IL6, Chol, Tri, HDL, LDL, VLDL, and mir126 were listed in Table 1. Based on this table, it is obvious that there is a high significant differences between the (age, IL6, Chol, Tri, HDL, LDL, and VLDL) of control and patient groups and non-significant for (gander, BMI, hs-Tropon, miR126) at p ≤ 0.05 level, as presented in Table 1. The anthropometric and biochemical parameters of the paticipants are indicated in Table 1, where Age, IL6, Chol, Tri, HDL, LDL, and VLDL in research group showed statistically significant variations.
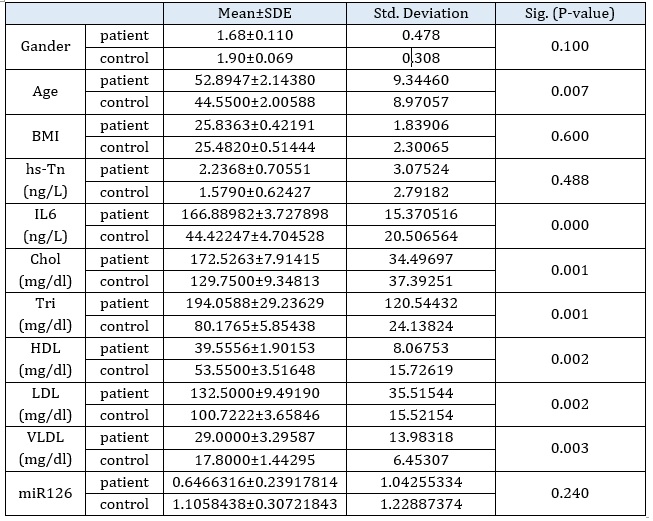
Control group of all these biochemical revealed a significant rise for results Age, IL6, Chol, Tri, HDL, LDL, and VLDL in the coronary artery (coronary heart) patients group that was consistent with Hedayatnia, M., et al. (2020) [24].
Heart disease patients' total serum Chol., Tri., LDL, and VLDL levels had been rising relative to the controls for all lipid profile indicators except HDL [25].
Likewise, the p-value for Il6 and Age showed an increase in the patient group compared with the healthy group. The pleiotropic cytokine IL6 has been found to be a prognostic indicator for many CVDs, including AF and CAD [26], while gender, BMI, hs-Tn, and miR126 indicated the apparent effect for both patients and other healthy people.
Patients serum Tri has a positive correlation with BMI (r=0.567, pv=0.018), but it was negatively correlated with gender (r=0.606, pv=0.01). VLDL showed a positive correlation with Tri (r=0.860, pv=0.000) and a negative correlation with HDL (r=-0.741, pv=0.001) (Table 2). In control group, Pearson correlation showed:
*A positive correlation between Chol and Age (r=0.515, pv=0.41), and between VLDL and Tri.
*Conversely, the negative correlation shows between Tri and hs-Tn (r= -0.521, pv= 0.032), Tri and IL6 (r= -0.754, pv =0.001), and between VLDL and IL6 (r= - 0.494, pv= 0.031).
Based on Tables 2 and 3 (The pearson correlation of patients and control groups) demonstrated miRNA-126 and lipid characteristics did not significantly correlate in any studies subject that were agreed with Ortega et al. [27] and they were disagreed with G. Musso et al. [28].
ROC curve and cute-off analysis
Receiver Operating Characteristic (ROC) curve and the best cut-off point for mRN126 at 0.976 had a sensitivity of (0.80) and 1-specificity of (0.60) were the total area under the curve = 759, pv=0.005. It means that miR126 at this cut-off point can be used as a screening test to detect patients with coronary artery diseases, as displayed in Figure 1 and Table 4.
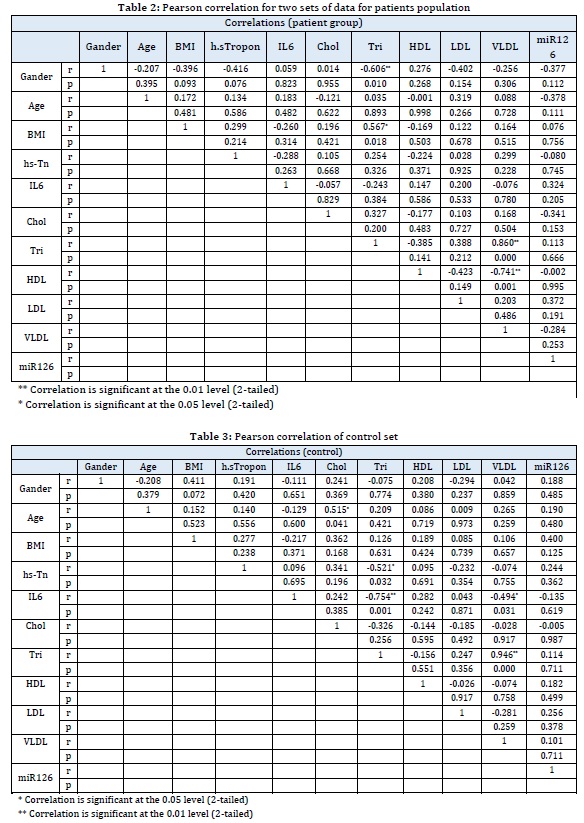

Figure 1:ROC curve
Table 4: The use of miR126 for detecting patients with coronary artery diseases

All the samples under this study (patients and healthy people) had the body mass less than 25. Therefore, the obesity and high weight were excluded, which had led body mass was not affected factor. Often associated with CAD are comorbidities such as abnormal lipid profiles which then leads to the dyslipidemia development and the well-known lipid triad: high levels of plasma triglycerides, low levels of high-density lipoprotein, and the appearance of small dense low-density lipoproteins (dsLDL). This triad is along with endothelial dysfunction, which can also be induced and contributed to atherosclerotic plaque formation. Likewise, lipid triad inhibits lipoprotein lipase activity in adipocytes leading to an increased release of free fatty acids and inflammatory cytokines such as IL-6. This explains the reason for the rise of lipid (Tri, Chol, VLDL, and dsLDL) and IL-6, and decrease of HDL in atherosclerosis [29]. Age showed an increase in the patient group (patient with CAD) in comparison with the healthy group; Since age is a risk factor for getting CAD, and includes age >45 years old in men and >55 years old in women [30]. Men are more likely to develop coronary heart disease (CHD) than women that they tend to do at a younger age than women. This is closely related to the estrogen deficiency, due to ovarian failure, endothelial dysfunction, where estrogen acts as an anti-arteriosclerosis in women in their early to mid-life. However, in this study, there was no significant difference between the gender in both patient group and control group. Because younger women accounted for 80% of all the healthy and patients [31, 32]. After myocardial damage begins, cardiac troponin I (cTnI) levels rise quickly and return to normal within 7 days [33]. Acute myocardial infarction (AMI), cardiac arrhythmias, and heart failure are acute and chronic cardiac disease states associated with elevated cTn concentrations. cTn is a reliable indicator of cardiovascular and all-cause mortality in the general population3-5, in people with known CAD, acute coronary syndrome (ACS), non-cardiac disease, and in people without these conditions [34]. The cases under this study all had CHD, but they had a history on CHD for a long time. Thus, there was no effect on troponin tests, which proves that troponin levels rise in blood when the heart attack is acute and immediately [35]. The miR-126 level did not change so no significant correlation was found between control and patient groups (p = 0.274). This might be brought on by the study participants' regular use of aspirin and -blockers, which could have an impact on the miR-126 level [36]. These results suggest that miRNA levels were unaffected by anti-platelet treatments [37, 38] that was agreed with Fiedler J (2011) [39] and disagreed with Mishra S [35]. It is possible to classify LDL cholesterol as a typical proatherogenic substance. Normally, LDL-receptors remove LDL particles from circulation (LDL-R). Different lipid metabolism-related molecular pathways are impacted by hyperlipidemia CAD. The LDL particles circulate in the bloodstream for an extended period of time [40, 41]. The reverse cholesterol transport or the cholesterol movement from peripheral tissues like macrophages in atherosclerotic plaques back to the liver is made by HDL particles. Thus, patients with hyperlipidemia and CAD may benefit from the enhancement of reverse cholesterol transport via HDL [41]. MiRNAs have been demonstrated to have important regulatory functions in lipid metabolism. They play a key role in coordinating the normal progression of the lipid metabolism. Since the use of apoptotic bodies caused the atherosclerotic plaques to regress, the miR-126 application also appears to be highly promising. Nevertheless, this is due to the possibility that it could have a proatherogenic effect by activating vascular smooth muscle cells [42]. In the present study, there is no significant correlation between plasma levels of miRNA-126 in diseased groups and the LDL levels and other lipid; uncertainty persists regarding the mechanism underlying the proportional drop in miR126 levels in the blood among CAD patients and the rise in LDL cholesterol. Despite the fact that high-density lipoprotein (HDL) cholesterol and miRNA are known to be connected. The transcription, processing, and stability of the miRNAs within circulating cells, as well as the capacity of these cells to release miR NAs into the plasma, may all have an impact on the amounts of circulating miRNAs. Circulating miRNAs may be delivered to the heart or blood vessel cells via microvesicles, exosomes, or apoptotic bodies. Since the current study evaluated miR-126, the current findings in plasma could be a result of modifications in miRNA transcription, or processing, and their release from circulating cells. It is yet unknown if down-regulation of miR-126 in CAD patients is a direct cause of inflammation or a response to it. Based on the observed variations in miR-126 level in relation to LDL cholesterol, it is possible that the levels of circulating miRNA are a result of a compensatory response to inflammation in the hyperlipidemia context. Endothelial miR-126 may become depleted as vascular disease processes progress and plasma levels may begin to drop. Transcoronary transit consumes the vascular miR-126. According to certain reports, specific miRNAs control cholesterol efflux. However, it is not anticipated that miR-126 will target any LDL component(s). Statin did not affect miR-126 in endothelial progenitor cells, despite the fact that statin was often administered to CAD patients in this study. The underlying processes that may connect LDL cholesterol and circulating miR-126 levels in patients with CAD need to be further investigated [43].
Conclusion
Micro RNAs as important regulators of the course of cardiac disease serve as the early indicators and therapeutic targets for cardiometabolic disease. Elevation of some miRNAs such as miR126 in cardiovascular patients can efficiently prognoses the CAD development in these patients. Therefore, miRNA126 can be used as a biomarker because its level was significantly decreased in patients with CAD.
Acknowledgments
My thanks and gratitude to all those who helped me carry out this research.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Authors' contributions
All authors contributed to data analysis, drafting, and revising of the paper and agreed to be responsible for all the aspects of this work.
Conflict of Interest
There are no conflicts of interest in this study.
HOW TO CITE THIS ARTICLE
Hibah Rajaa Taher, Perry Habib Saifullah. Role of MiRNA126 As a Biomarker for Coronary Artery Disease in Iraqi Patients. J. Med. Chem. Sci., 2023, 6(5) 1163-1172