Document Type : Review Article
Authors
1 Laboratory of Pharmaceutics and Pharmaceutical Technology, Faculty of Pharmacy, Universitas Indonesia, Depok 16424, West Java, Indonesia
2 Department of Pharmaceutics, Faculty of Pharmacy, Universitas Surabaya, Surabaya 60293, East Java, Indonesia
3 Laboratory of Pharmaceutical and Medicinal Chemistry, Faculty of Pharmacy, Universitas Indonesia, Depok 16424, West Java, Indonesia
Abstract
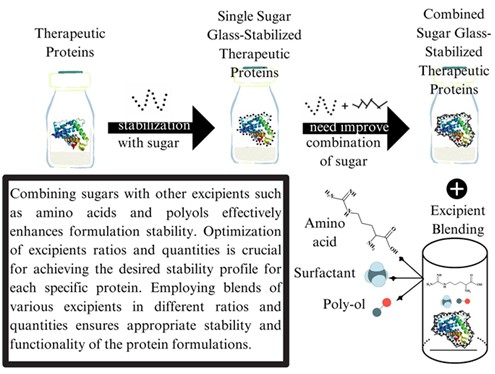
Sugars have long been used as stabilizing excipients for protein-based therapeutics during the development and production process. Incorporating stabilizing agents, such as sucrose and trehalose into dry formulations is a common strategy approved by the US FDA. However, single-sugar formulations may not be sufficient for diverse medicinal products, necessitating the blending of excipients to improve stability, dissolution, and patient acceptability. Our goal is to comprehensively understand the impact of excipients, focusing on the impact of various sugar types on formulation stability, and to explore the effectiveness of incorporating additional excipients such as amino acids, surfactants, and polyols, to further enhance protein stability.
The review begins with an overview of sugars’ used in proteins stabilization and the blending of excipients. It discusses the limitation of single-sugar formulations and explores alternatives such as oligo- or polysaccharides. The mechanism underlying sugar and excipient combinations is elucidated, emphasizing the benefits of blending multiple excipients for stabilizing therapeutic protein. Our findings demonstrate that excipients blending presents a promising strategy for improving the stability of sugar-stabilized therapeutic proteins. Combining sugars with other excipients such as amino acids and polyols effectively enhances formulation stability. Optimization of excipients ratios and quantities is crucial for achieving the desired stability profile for each specific protein. Considering the impact of excipients on stabilization is essential in therapeutic proteins development. Employing blends of various excipients in different ratios and quantities ensures appropriate stability and functionality of the protein formulations.
Graphical Abstract
Keywords
Main Subjects
Introduction
Over the past decade, therapeutic proteins have emerged as lifesaving treatments, with their high efficacy and specificity leading to a substantial increase in their usage worldwide [1]. As evidenced by projections, the global protein therapeutics market is anticipated to undergo substantial growth, with its size estimated at USD 516.79 billion in 2024 and expected to reach USD 761.80 billion by 2029, reflecting a robust compound annual growth rate (CAGR) of 8.07% during the forecast period (2024-2029) [2]. This projection underscores the anticipation of the global protein therapeutics market doubling in size over the next decade, driven by the increasing demand; there is an urgent need for these therapeutic agents [3]. However, ensuring the potency and stability of therapeutic proteins poses a significant challenge due to the complexity and instability of their physical and chemical structure. Degradation of these proteins not only compromises their efficacy, but can also lead to severe side effects [1]. Consequently precise formulation methods are required, necessitating a deeper understanding of the physicochemical properties of stabilizing agents, particularly sugars, which are commonly employed in protein formulations [4].
Addressing this challenge requires a thorough examination of the physicochemical properties of sugars, which play a crucial role in maintaining protein stability. Primarily, non-reducing sugars with high glass transition temperatures (Tg) are essential to keep the protein formulation in an amorphous state, thereby preventing denaturation or aggregation [5]. Next, maintaining low water activity is necessary to further prevent protein denaturation or aggregation. In addition, ensuring high solubility is essential to ensure uniform distribution across the formulation [6]. Finally, sugars with low reactivity are important to minimize the chemical degradation of the protein, while biocompatibility and non-toxicity are crucial to avoid any adverse effects on the biological activity of the protein [7]. Extensive development efforts have led to the registration by the United States Food and Drug Administration (US FDA) of thermally stable protein formulations. Although most therapeutic protein products are available in liquid formulation [8], transitioning proteins to a dry state is a promising approach to render them stable [7].
Storing proteins in a dry state may significantly reduce breakdown rates due to restricted molecular mobility, thereby enhancing their stability and shelf life [4].
However, proteins are subjected to multiple stresses during drying and subsequent storage, which may compromise the integrity of the protein. For example, in spray drying process, protein may encounter stress due to high inlet or outlet temperatures [9]. Furthermore, in freeze-drying processes, lyophilization involves two stress factors that cause protein denaturation: freezing and drying. Therefore, certain conditions are necessary to ensure that the stresses are not harmful to the protein [10].
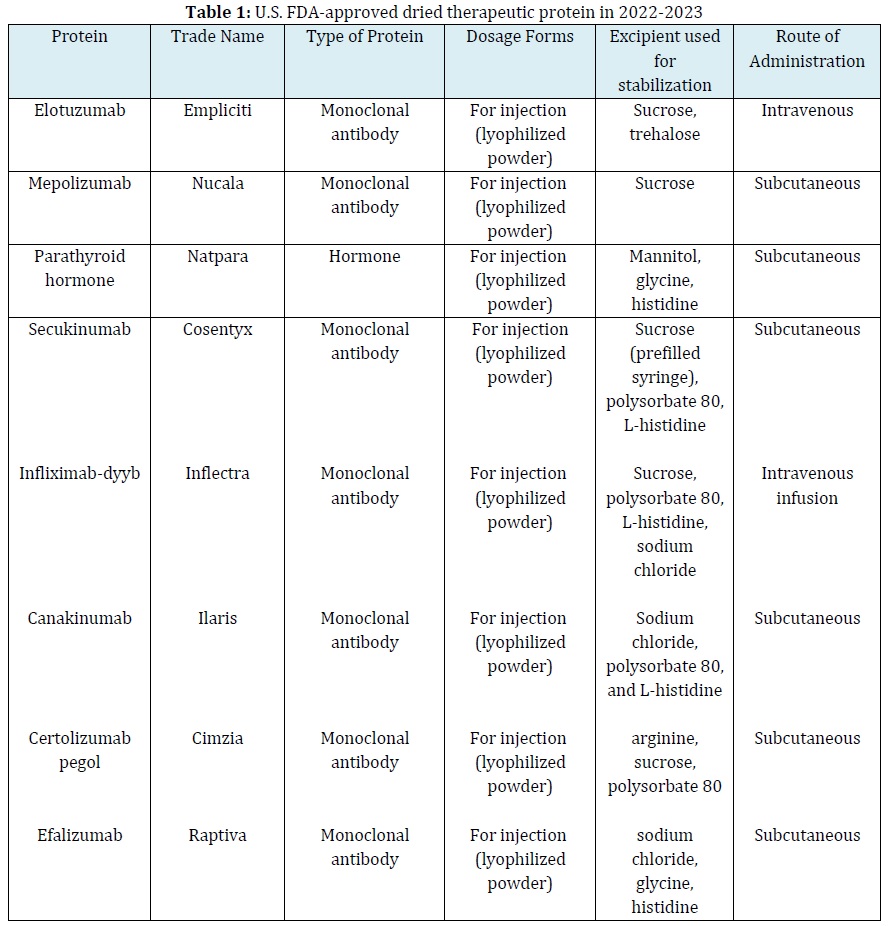
One strategy to protect proteins against deterioration involves incorporating stabilizing agents such as sugars and surfactants into dry formulations [10]. Sucrose and trehalose are the most commonly used sugars for stabilizing solid protein formulations. Dry sugar-stabilized therapeutic protein and other stabilizer-therapeutic protein products that have been approved and registered by the US FDA are presented in Table 1 [11, 12].
For several reasons, incorporation of a protein with a single sugar may not be sufficient for producing various medicinal products that can be administered through different delivery systems and routes. Initially, protein stability can be affected by factors such as pH, temperature, and light exposure during storage and administration, which can cause degradation or aggregation. Next, proteins may interact with other components in the formulation, leading to stability issues. Furthermore, the formulation may impact protein dissolution ability, resulting in difficulty in administering the product. To overcome these challenges, extra excipients, such as stabilizers, protectants, and solubilizing agents may be added to the formulation to stability, and usability [13]. Excipients play a crucial role in improving safety, efficacy, stability, and patient acceptability, underscoring the importance of evaluating their impact on sugar-stabilized therapeutic proteins in powder formulation. This review seeks to comprehensively evaluate the influence of different sugar types on protein stability, and analyze several excipients capable of further enhancing the stability of sugar-stabilized therapeutic proteins. Through this review, valuable insights into the development and optimization of protein formulations will be gained, ultimately improving the safety, efficacy, and usability of therapeutic proteins.
A systematic electronic search was conducted across various databases, including PubMed, Springer Link, Science Direct, ACS Publications, and Google Scholar, to investigate the influence of excipient blending on sugar-stabilized therapeutic proteins. The search utilized a range of keywords such as Therapeutic proteins, Excipient blending for therapeutic protein, Sugar for protein stabilization, Amino acid for protein stabilization, Polyol for protein stabilization, and Sugar-stabilized therapeutic proteins. Studies meeting the inclusion criteria were included in the analysis, which required (1) publication between 1999 and 2024, with English text, (2) availability as complete articles, and (3) provision of relevant data pertaining to therapeutic proteins and excipients for protein stabilization. However, studies published solely as proceedings were excluded from consideration.
The influence of sugar and mechanism underlying protein stabilization
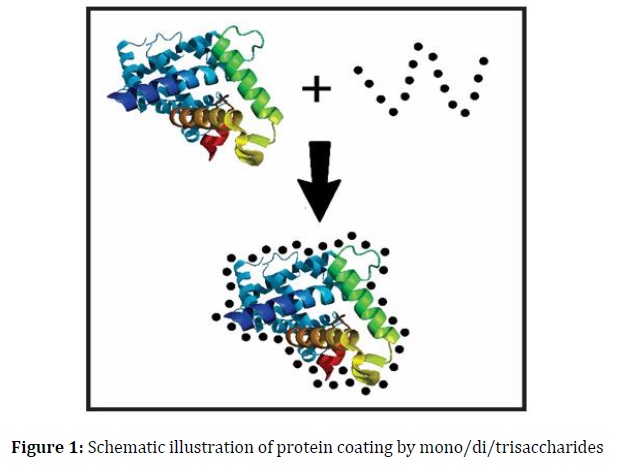
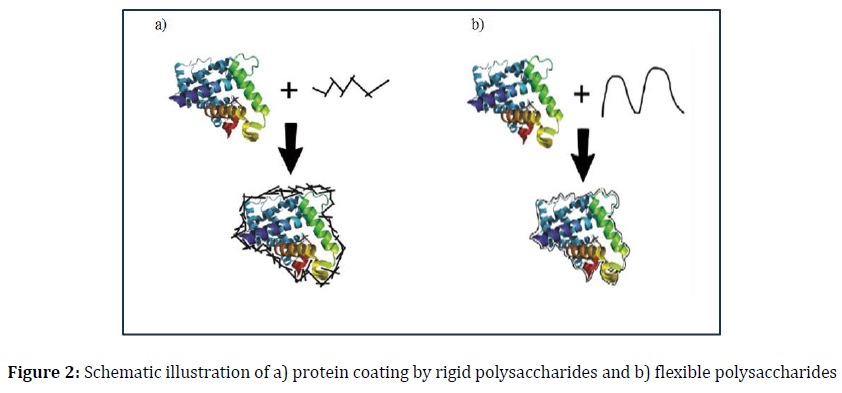
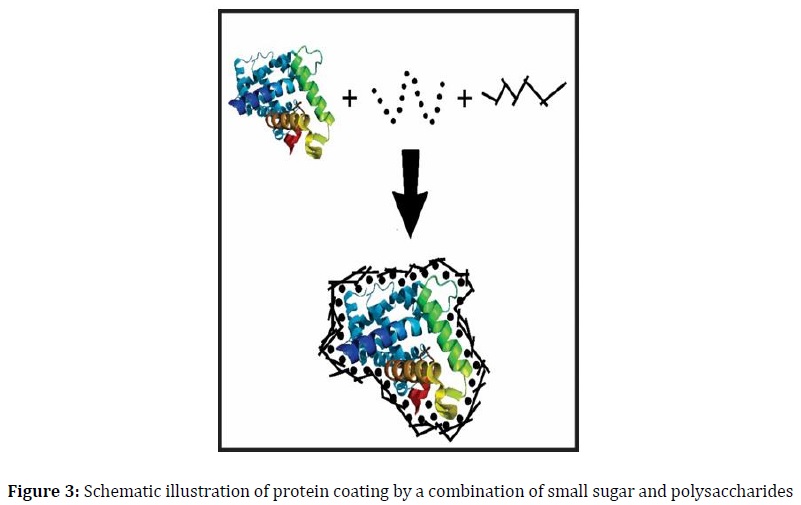
Several theories, including vitrification/shielding, particle isolation, water replacement, and refinements, have been proposed to elucidate how sugar preserves protein integrity [5, 14]. The vitrification theory illustrates the formation of proteins within a tightly structured coating of a sugar glass matrix, effectively reducing protein molecular mobility and degradation rates [15]. This sugar glass formation can be quantified by measuring its glass transition temperature (Tg). However, it is important to note that a simple sugar coating may not be universally effective in stabilizing all types of proteins. In high relative humidity (RH) conditions, while small sugars can create a tight coating (Figure 1), they may exhibit limited physical durability due to their low Tg. Conversely, the use of rigid oligo- or polysaccharides can result in a non-tight covering (Figure 2a), offering superior physical stability. These findings suggest that optimal protein stability may be attained by combining flexible, molecularly structured oligo/polysaccharides (Figure 2b) with disaccharides and additional oligo/polysaccharides (Figure 3). This approach is grounded in the fundamental mechanism through which proteins are fortified with sugar.
It has been observed that a sugar's ability to stabilize a protein is enhanced when its glass transition temperature (Tg) exceeds a certain threshold [16].
This signifies a higher propensity for kinetic immobilization. Previous studies have shown that the Tg of sugar glasses rises with increasing Molecular Weight (MW) of the stabilizer. Furthermore, it has been suggested that the protein becomes immobilized when the Tg surpasses the storage temperature [17]. The Fox-Flory equation has been found to be particularly applicable for a specific type of oligo- or polysaccharide at different molecular weights. This equation provides valuable insights into the relationship between molecular weight and the glass transition temperature, offering further guidance in optimizing sugar-stabilized protein formulations. Proteins have the potential to aggregate or engage in intermolecular reactions. This propensity underscores the significance of theories like the particle isolation theory, which posits that a sugar matrix acts as a physical partition between particles. This partition effectively prevents their aggregation or reaction with one another. Sugar molecules in this case should have minimal translational mobility or be in a glassy form [15]. According to the water replacement theory, the hydrogen bonds between protein and water molecules in the protein layer are replaced with sugar to achieve structural protection and stabilization based on the thermodynamic law [15, 17]. Over several decades, the vitrification and water replacement theories have been widely used to explain the stabilization mechanism of sugar in the dried state.
The molecular flexibility of sugar, including density and interaction, as well as the miscibility between protein and sugar, has all been explored in the context of refined stabilization hypotheses. Mobility types should be understood because they often determine the routes of degradation and the protection strategies to be employed. Local and global mobilities are associated with chemical deterioration and physical degradation, respectively [5, 18]. The vitrification theory states that the local mobility (β relaxation) of certain protein groups may exhibit greater predictability than global mobility (α relaxation) [5, 19]. While plasticizers enhance both global and local mobility, the use of anti-plasticizers inhibits local mobility [17, 20]. Several studies have analyzed the ability of different sugars to replace water in stabilizing proteins [21, 15]. Small oligosaccharides were reported to possess a greater ability to stabilize protein during storage after lyophilization compared to large oligosaccharides. This appears to be due to the hydrogen bonds between small oligosaccharides, which are more molecularly flexible with protein during freeze-drying [15].
The incorporation of sugar is an absolute requirement for stabilizing protein molecules [22]. The ability of a protein to blend with sugar will decrease with the increasing size of sugar in the IgG protein model, leading to an elevation in protein aggregation. The probability of a protein being present on the particle surface reduces when sugar and surfactants are added [23].
The impact of various sugar types on protein stability in formulations
In most cases, when sugar is added to a protein, it is to immobilize protein molecules in sugar glass matrices. Several applications of a single or a combination of sugar in protein stabilization have been reported.
Single sugar
Single sugars refer to the use of only one type of sugar for stabilization, whereas blends involve combinations of sugars with other molecules. Small sugars, with lower molecular weight, can pose a challenge to protein stabilization. Their low glass transition temperature (Tg) renders them susceptible to environmental factors, potentially compromising the stability of therapeutic proteins. Monosaccharides such as glucose and fructose, classified as simple sugars, have been utilized to stabilize proteins through the formation of hydrogen bonds and other interactions. However, their effectiveness may be limited by their low Tg, particularly in high humidity condition. Disaccharides, such as sucrose, maltose, and trehalose, which are composed of two simple sugar units, have been incorporated into dry protein formulations as excipients. These sugars provide a hydrophilic environment that aids in preventing aggregation and denaturation. Trisaccharides, such as raffinose, contribute to maintaining protein integrity and preventing denaturation under various stress conditions by physically protecting proteins and potentially forming hydrogen bonds for structural stabilization.
Polysaccharides, such as inulin, dextran, and pullulan, composed of long chains of monosaccharides, enhance protein stability. Inulin has demonstrated the ability to reduce protein aggregation and stabilize proteins during freeze-drying [22].
Monosaccharides: Glucose and fructose
Monosaccharides such as glucose, fructose, and galactose, are simple or single sugar, meaning they cannot be broken down into smaller sugar molecules. These have been used to stabilize protein by forming hydrogen bonds and other interactions with the protein molecules.
Glucose
The first application of sugar to stabilize lysozyme was reported by Allison et al. in 1999 [24]. Allison et al. compared several saccharides including glucose as a monosaccharide to apply in a protein model lysozyme. The FTIR observation shows that the presence of glucose weakens the hydrogen bond in the protein's carboxyl group, causing the loss of a carboxyl bond at a wavelength of 1580 cm-1 due to the lack of water in the dry form. Protein aggregation and denaturation can be prevented by monosaccharides' presence, but they are not adequately protected by glucose during lyophilization which causes their structure to unfold. Furlan et al. [25] tried to obtain stable protein formulations with monosaccharide protection through a glassy state mechanism. In a study using bovine plasma protein as a model, glucose was employed to stabilize the protein produced through the freeze-drying method. From the DSC profile, the Tg value was to be influenced by the saccharide's MW, and a higher Tg generally indicated a greater stabilization ability of the saccharide. The MW of a saccharide is known to affect its thermal stability, and in the case of freeze-drying, Tg is considered as a key factor in dried protein stability. This highlights the importance of considering the MW and other physical characteristics of the protein when choosing a saccharide for stabilization purposes.
Bashir et al. [26] further compared several monosaccharides and disaccharides, including glucose for stabilization of α-lactalbumin (α-LA). The concentrations of glucose at 1 M or higher resulted in a notable reduction in scattering at 400 nm, indicating a significant inhibition of aggregation. The prevention of α-LA aggregation by sugar osmolytes is attributed to the formation of hydrogen bonds between these osmolytes, as evidenced by the findings of molecular docking studies.
Fructose
Fructose has been found to stabilize the protein. Its low freezing point and high solubility lead to being an ideal excipient for use in freeze-drying processes to improve protein stability. This stabilization mechanism is believed to be due to the formation of hydrogen bonds between fructose and protein molecules, which helps to prevent denaturation and aggregation [6]. Fructose can act as an osmoprotectant, helping to prevent protein denaturation due to changes in osmotic pressure during the freeze-drying process. However, it is not a suitable protein stabilizer due to having a low Tg value (20-25 °C or <40 °C) that varies based on factors such as fructose concentration, the presence of other substances, and the method used to determine Tg [15]. Bashir et al. [26] also employed fructose and a combination of glucose and fructose to inhibit α-LA aggregation. Transmission electron microscopy (TEM) analysis demonstrated that a blend of glucose and fructose was the most effective inhibitor of α-LA fibril formation. This interaction facilitated by hydrogen bonding plays a pivotal role in preventing α-LA aggregation in the presence of sugar osmolytes.
The use of monosaccharides is often avoided because they have a very low Tg. This is specifically exacerbated by the presence of strong residual moisture promoting Tg decrease. Furthermore, the Tg value is often below the storage temperature, which negatively affects its stability. Another factor to consider is that monosaccharides cause Maillard browning due to reducing sugar.
Disaccharides: Sucrose, trehalose, and maltose
Disaccharides are sugar composed of two simple sugar units or monosaccharides. These have been used as excipients in dry protein formulations to enhance stability by providing a hydrophilic environment that helps to prevent aggregation and denaturation. In addition, they can form hydrogen bonds with a protein to ensure conformational stability.
Sucrose
Wang et al. examined the relationship between protein and sucrose in dry formulations using water sorption isothermal data and FTIR spectra of the protein’s secondary structure. In this study, freeze-dried formulations of IgG and a cytokine fusion protein were tested with varying ratios of sucrose. Beyond a ratio of 1:1, adding more sucrose does not significantly increase sugar-protein bonds, due to its required bonding with water [27]. Furlan et al. also confirmed sucrose as a protein stabilizer [25] by comparing inulin, sucrose, and glucose as stabilizers for bovine plasma freeze-dried formulation. The best stability of this formulation was obtained with inulin due to the MW effect, while sucrose was more effective than glucose. This was confirmed by the higher Tg obtained from the bovine plasma-based sucrose formulation compared to the glucose-based. Furthermore, the sucrose concentration affected the Tg value of the formulation, as both parameters increase proportionally.
Trehalose
Another disaccharide, trehalose, has been discovered to exhibit a protective effect on protein by preventing denaturation and aggregation under conditions of stress, such as freeze-drying and exposure to extreme heat. Allison et al. compared the effects of trehalose and sucrose on the lysozyme protein (egg white lysozyme) stability in a model system. The results obtained showed that without a disaccharide, a carboxyl bond at 1580cm-1 disappeared, indicating a reduction of hydrogen bonds in the protein's carboxyl group due to water loss in the dry state. Elevation in the starting concentration of sucrose/trehalose led to an increase in the carboxylic band. However, sucrose was found to better preserve the dry lysozyme's ability to maintain protein structure and form hydrogen bonds, compared to trehalose [24].
Moreover, Amaro et al. developed a salmon calcitonin protein to produce nanoporous Microparticles (NPMPs) with the addition of trehalose as a stabilizer. The results of the morphology test conducted using SEM showed spherical particles with non-porous surface characteristics. Crystallinity analysis using XRD showed that the particles generated were amorphous. Thermal characteristics tested with DSC found high Tg values of about 124 °C, representing a good physical stability indicator for trehalose-based formulations [28].
Maltose
Leblanc et al. (2016) assessed the impact of temperature, formulation, and concentration on sugar reactivity in protein preparation. Furthermore, immunoglobulin was used as a model protein to compare its stabilization by three different sugar, including sucrose, trehalose, and maltose. The results showed that non-reducing sugar, such as sucrose and trehalose, effectively prevented glycation and had low reactivity even at high temperatures [29]. Tonnis et al (2015) found trehalose as the most effective in improving protein stability compared to other formulations [21].
Trisaccharides: Raffinose
A trisaccharide can help to maintain the integrity of the protein structure and prevent denaturation during various stress conditions such as drying, high temperatures, and high humidity. For example, raffinose is capable of physically protecting protein, retaining their hydration shell, and potentially forming hydrogen bonds with the molecules for structural stabilization. Amaro et al. [28] reported the raffinose addition in a salmon calcitonin protein formulation to produce NPMPs. Spray-drying formulations with the addition of raffinose also yielded the same morphological and crystallinity characteristics as the trehalose-based NPMPs, which are spherical particles with a non-hollow surface and amorphous nature. The difference in the thermal characteristics stemmed from the fact that formulations with the addition of raffinose were not affected by the difference in spray-drying conditions, while the trehalose-based types were affected by the drying conditions. The Tg of the NPMP formulations incorporated with raffinose was slightly lower (120 °C), but still considered high and showed good physical stability. Experiments on storage stability revealed that composite systems containing raffinose exhibited greater stability compared to those containing trehalose. Combining salmon calcitonin in NPMPs at sugar:protein ratio of 4:1 indicated a potential biological activity, hence, tends to deliver protein through the pulmonary route.
An exposure of molecules such as trehalose to higher RH (>45%), which often occurs during spray drying, can cause the powder to become very sticky. On the other hand, supposing spray drying is carried out at very low RH, the powder becomes highly electrostatically charged, making it more difficult to handle. Therefore, using sugar with higher MWs may be an alternative solution, as they have a much higher Tg.
Poly saccharides: Inulin, dextran, and pullulan
Polysaccharides are complex carbohydrates made up of long chains of monosaccharides. These include Inulin, dextran, and pullulan which are capable of enhancing protein stability.
Inulin
Muntu et al. (2023) analyzed the physicochemical characteristics and stability of spray-freeze-dried (SFD) insulin powder. Inulin is one of the excipients used in this formulation, resulting in the lowest insulin content compared to formulations containing trehalose and their combination. These findings are attributed to inulin being a polysaccharide with a rigid structure, potentially limiting its ability to provide maximum protection to insulin. However, its physical characteristics are generally superior due to its high glass transition temperature (Tg) and the lowest moisture content among the formulations tested. These attributes may contribute to optimal physical quality, aligning with the desired characteristics of dry powder for intranasal administration [30]. Grasmeijer et al. (2013) also investigated the use of inulin to stabilize alkaline phosphatase as a model. The results indicated that protein can be stabilized in sugar glasses through the mechanism of vitrification and water substitution. Complete loss of protein activity was observed in samples with Tg below 50-60 °C, while the reduction of protein activity was significantly smaller in samples with higher Tg [17].
Furlan et al. (2011) compared the effectiveness of inulin to other saccharides in stabilizing a model of bovine plasma protein, using the freeze-drying process. The results showed that inulin's stabilizing properties were more effective at greater storage temperatures and the dried sample provided better stabilization than monosaccharides and disaccharides [25].
Dextran
Allison et al. (1999) were the first researchers to analyze the impact of dextran on the unfolding of protein structures during lyophilization. The results obtained showed that dextran was unable to prevent the opening of protein structures due to its incapacity to form strong hydrogen bonds with dehydrated protein. At a concentration of 100 mg/ml, dextran had a minor effect on the opening of protein, which was attributed to its lack of efficacy in forming the necessary hydrogen bonds, as evidenced by a steric obstruction. These observations were supported by the intensity of the carboxylic band [24]. Mensink et al. (2015) used dextran to preserve the structure and activity of Lactate Dehydrogenase (LDH) from degradation during storage. The study was based on the formation of hydrogen bonds between sugar and protein. The results showed that the least stability was discovered in dextran formulations with the smallest hydrogen bonds. Smaller-sized sugar was found to be more flexible molecularly, thereby forming larger hydrogen bonds, which provided better stability and were less affected by steric obstruction [18].
Pullulan
Teekamp et al. (2017) used pullulan (a polysaccharide with an MW of 200-300 kDa) to stabilize the protein model, β-galactosidase. This sugar molecule was employed because it has a higher Tg value (261 °C) than other polysaccharides. The glass transition and crystallization of the spray freeze-dried mixture were analyzed with Differential Scanning Calorimetry (DSC). Under low humidity conditions, the ability of pullulan to encase the protein is constrained due to its bulky nature, leading to lower stability compared to formulations containing trehalose. Conversely, in higher humidity conditions (e.g. 56% RH); the maltodextrin formulation is superior and reflects good stability in daily storage [31].
High-molecular-weight sugars possess favourable physicochemical properties; however, they may not efficiently coat proteins, thereby disrupting vitrification and water replacement stabilization. To address this issue, blending small and long chain sugars can serve as a viable alternative.
Combination of small sugar with polysaccharides
Since 2014, the practice of protein stabilization has progressively shifted towards employing a combination of disaccharides and oligo/polysaccharides. This trend can be partly attributed to the limitations of using only one sugar type.
Pullulan and trehalose
Teekamp et al. [31] optimized protein stability by combining binary polysaccharides and disaccharides in a glassy state. Furthermore, the combination matrix of a polysaccharide and disaccharide was formed by selecting different compositions of pullulan and trehalose. For this study, β-galactosidase (βgal) was employed as a model protein and the freeze-drying method was used to stabilize the pullulan-trehalose glass matrix. All compositions of the pullulan/trehalose mixture were reported to have a single Tg value, indicating that the highest frozen fraction of the concentrate included a homogenous combination of two saccharides. The enzymatic activity of βgal in both formulations decreased after four weeks of storage. However, formulations containing sugar were more stable than those without sugar. The formula containing pullulan only showed an enzymatic activity of 83%, while that of trehalose had up to a value of 96%. This increase in inactivity was observed along with a reduction in the ratio of pullulan to trehalose. Based on these observations, the combination of small sugar (trehalose) and polysaccharide (pullulan) has great potential for solid protein stabilization.
Dextran and trehalose
Tonnis et al. reported that a combination of small sugar and polysaccharides provided remarkable improvement in protein stability. A combination of dextran 70 kDa and trehalose (1:1) was used to improve the stability of Insulin, HBsAg, LDH, and βgal as protein models. This combination improved the storage stability of protein models containing both sugars compared to the protein without sugar or protein with dextran only. The stability of protein was found to be affected by the MW of dextran, with higher MWs leading to greater improvements [21].
Inulin and trehalose
Muntu et al. (2021) analyzed the key parameters involved in the SFD process and determined the appropriate ratio of inulin or trehalose to be used in the insulin microparticle formulation. The critical process parameters that were identified for producing the desired insulin microparticles included ensuring uninterrupted continuity during the SFD process and running the FD process for a minimum of 50 hours without interruption. Seven insulin formulations were compared based on certain sugar components, including inulin or trehalose alone, as well as five variations of their combinations, each with ratios of 5:1, 2:1, 1:1, 1:2, and 1:5. The most optimal formulation was the inulin-trehalose ratio of 1:1. This was confirmed by the lowest moisture content of 2.62% and the highest encapsulation efficiency value of 99.68% [32].
Excipients blending of sugar-stabilized therapeutic protein
Saccharides were previously mentioned to be capable of enhancing protein stability. Other pharmaceutical excipients can also carry out this role through several mechanisms. They can be used alone or in combination, depending on the specific requirements of the protein involved and the formulations. Excipients such as silica and PVA adsorb onto protein surfaces, preventing aggregation and maintaining stability. Some also provide osmotic protection to protein by creating a high-viscosity environment that prevents denaturation during drying and storage. Several polysaccharides such as inulin and dextran, due to their ability to create a high-viscosity environment, have been proven to provide osmotic protection to proteins. This prevents protein denaturation and preserves their biological activity [33]. PVP can help to maintain the hydration of protein, reduce their interactions, and prevent denaturation.
Buffers such as sodium citrate and sodium bicarbonate maintain a constant pH during storage, preventing changes in protein conformation that could lead to degradation. PEG can modify the protein surfaces, decrease their interactions, and maintain stability [34]. The stabilization of protein formulations with sugar can be significantly influenced by the addition of excipients as shown in Figure 4.
The mechanism behind excipients' stabilizing effect is often related to the formation of a protective network around protein, which can reduce their aggregation rate, denaturation, and precipitation. This network is created through physical entrapment or by forming hydrogen bonds. The effect on protein stability will depend on the MW value, charge, and chemical structure, as well as the specific conditions of storage and use [35]. This section discussed the effects of blended excipients on sugar-stabilized therapeutic protein. Gagarskaia et al. (2017) [36] developed a formulation of actin-loaded glassy microparticles of polysaccharides using additional polymers. Dextran was incorporated with various MWs of PEG (PEG 2000, 4000, 6000, and 8000) to stabilize actin via inhibition unfolding reaction. PEG, dextran, and protein were all dissolved in water, and then freeze-dried to obtain an encapsulated protein. During the freezing stage, phase separation takes place, resulting in the formation of an emulsion with a dextran-rich phase and a PEG-rich phase. Subsequently, after freeze-drying, PEG is extracted. The results showed that encapsulated MGB & BSA have a higher resistance to solvents, shear forces, centrifugation process, and temperature compared to the non-encapsulated counterparts. Protein activity decreased slightly when formulated in polysaccharide microparticles. Hence, this method of freezing-induced phase separation provides a gentle and efficient approach to encapsulate proteins within hazard-resistant polysaccharide glassy particles. This ensures their stability during subsequent formulating processes, leading to the development of a polymer-based sustained-release system.
As mentioned earlier, the initial study conducted by Allison et al. (1999) also examined the effects of glucose, PEG, and a combination of sugar-PEG in enhancing lysozyme stability. PEG’s addition to sucrose and trehalose maintained lysozyme structure during freezing and drying. Likewise, a reduction in carboxylic band absorption was found in the Sugar-PEG formulation compared to the counterpart without PEG. In the presence of PEG, there was less sugar available to interact with protein, leading to decreased, hydrogen bonds due to the splitting of some sugar molecules with PEG in a compact and dehydrated state [24].
Effects of hydroxypropyl-beta-cyclodextrin (CD)/PVP blending on sucrose-stabilized protein
Haeuser et al. developed a sucrose-based Monoclonal Antibody (mAb) formulation with several excipient combinations to optimize storage stability at room temperature and shorten the cycle duration of the FD process. Normally, a freeze dry cycle is quite time consuming which makes the process quite expensive. Therefore, shortening the process would make production more cost effective. This might be accomplished by using blends of excipients. This was achieved by analyzing a mixture of excipients including 2-hydroxypropyl-beta-cyclodextrin (CD), PVP, and dextran 40 kDa (Dex). All formulations with 10 mg/mL mAb had shorter reconstitution time compared to the formulations with 50 mg/mL. Sucrose-based mAb formulations combined with CD or both CD and PVP can possess all the good attributes mentioned. These combinations also allow storage at room temperature for at least nine months and indicate excellent stability at 40 °C. The PVP inclusion as a bulking agent and CD was found to be crucial for increasing physical stability and Tg, which also improved mAb stability.
Utilizing CD or a combination of CD and sucrose could reduce production costs compared to formulations with the PVP addition by shortening the FD process duration and enhancing mAb stability at room temperature and 40 °C. These findings align with a previous study that achieved a 50% reduction in the FD cycle duration for mAb formulation. Moreover, formulations containing only CD or the CD-sucrose combination exhibited all the desirable quality attributes, including specific surface area, residual moisture, Tg and collapse temperature (Tc). The incorporation of CD or both CD and PVP into the sucrose-based mAb formulation resulted in good storage stability at 40 °C for three months. However, blending with either sucrose or PVP did not demonstrate added value [37].
Blending of amino acids in sugar-stabilized therapeutic protein formulations
Previous studies established that the presence of amino acids during lyophilization maintains protein conformation and increases their stability [38]. Various mechanisms have been identified for how amino acids optimize the stabilization of protein and sugar-stabilized therapeutic protein formulations [39]. Different types of amino acids, such as glycine, arginine, phenylalanine, serine, and histidine have been employed in the formulation of therapeutic proteins [38].
Glycine
Mirfakhraei et al. used several variations of amino acids in the formulation of trehalose and IgG, one of which is glycine. The obtained results showed that the natural structure of IgG remained intact in the presence of amino acids. The glycine-based powder sample exhibited a slight endothermic peak at approximately 45 °C. Among the amino acids, glycine demonstrated the least effectiveness. The reduced stabilization capability of glycine was attributed to its crystallization. Also, using trehalose alone was found to be more effective in stabilizing IgG than amino acids [40]. In another study, Zhang et al. (2023) incorporated glycine into sugar-stabilized mAb formulations. The optimized excipients system in this study comprised sucrose-glycine, along with polysorbate 80. In a formulation using only sucrose as excipient, the activity recovery of mAb after freeze drying, is around 80% and did not vary with different sucrose concentrations. In a sucrose-glycine system that was used, the use of the two involved excipients at different ratios indicated that the glycine crystals hindered activity recovery, leading to a decrease in recovery despite sufficient sucrose being present to protect the protein. Upon heating the sucrose-glycine formulations, there was a small but substantial reduction in activity recovery compared to the unheated controls, while no such effect was observed from using only sucrose as excipients [41]. Kraan et al. studied the development of Inactivated Polio Vaccine (IPV) using a drying method with excipients such as sucrose, d-sorbitol, d-trehalose dihydrate, mannitol, glycine, and myo-inositol. The dried formulations using blend of sucrose/trehalose with glycine produced led to minimal loss of potency during drying and storage compared to the liquid ones, indicating that the IPV was stable and can be stored at room temperature [42].
Emami et al. also used a combination of trehalose and several variations of amino acids including glycine, which obtained optimal stability in the IgG formulation. This result can be proven by the aggregate amount which was significantly lower than the excipients-free formulation. Nevertheless, formulations with trehalose alone also demonstrated the absence of IgG fragmentation both after SFD and during storage for two months. However, the formation of multimers after SFD was significantly lower in formulations containing a blend of trehalose and glycine. Furthermore, during storage at 40 °C and 75% RH for two months showed good stability with aggregates <2.2% and monomer peak >93% for formulations containing glycine [43].
Arginine
The stability of a SFD trehalose-based IgG formulation was assessed. The formulation comprising arginine had better stability than the excipients-free counterpart. Both IgG formulations with trehalose alone and formulations with a mixture of trehalose and arginine showed no fragmentation. However, formulations of IgG with a blend of trehalose and arginine exhibited the formation of multimers at 29.7-34.3%, which was significantly higher compared to the multimers in formulations of IgG with trehalose alone. In addition, it induced IgG aggregation and fragmentation during the SFD process attributed to the hydrophilic nature of arginine, which contains a positively charged guanidium group. The overall results suggested that stable IgG formulations can be produced from trehalose combinations with uncharged-nonpolar amino acids including leucine, phenylalanine, or glycine, through the SFD method. In contrast, hydrophilic amino acids, e.g., arginine, often exhibit IgG aggregation and fragmentation [43].
To develop adalimumab microparticles for inhalation, Emami et al. used arginine as a stabilizer. As discussed earlier in the glycine subchapter, this study showed that the use of amino acids provided good stability at 40 °C for three months. The aggregation level of this formulation is relatively low, below 4%. This result differs from findings in other studies that also used arginine with IgG, which tended to induce aggregation. In this case, the isoelectric point (pI) of the protein becomes the determining factor. Adalimumab and arginine have a smaller difference in pI values or both tend to be basic, thus the use of arginine in adalimumab formulations results in better stabilization effects with a low aggregation level [43, 44].
In sucrose/trehalose-based protein formulations produced through the SFD method, several combinations with polyol and L-arginine were compared for their effect on protein characteristics and stability. The results showed that the best value of moisture content was obtained from formulations containing a blend of trehalose and mannitol. In addition, the physicochemical characteristics of the protein produced are non-linearly correlated with the ratio of trehalose and mannitol. The relationship between stress and stress relaxation concerning sugar content exhibits a non-linear pattern across three excipient formulations containing sucrose and trehalose. Moreover, the texture of freeze-dried cakes does not consistently change with increasing sugar concentration [45].
Hackl et al. compared and analyzed several sucrose/trehalose and mannitol ratios in the L-arginine-based formulation for their effect on BSA stability. The ratio of arginine to excipient sugars and mannitol used in this formulation was standardized at a ratio of 1:4. In addition, the ratio variations of sugars to mannitol were in the range of 20:80 to 80:20 when subjected to high humidity and temperature during storage, formulations with relatively high failed to decrease protein aggregation. To effectively include l-arginine into a protein formulation while ensuring a mannitol-to-trehalose ratio above 1, it was necessary to decrease the concentration of trehalose and increase the concentration of mannitol, while the arginine content remains the same [45].
In another study, Startzel et al. used arginine in a sucrose-based Monoclonal Antibody (mAb) formulation. Furthermore, the effect of variations in excipients weight ratio on Monoclonal Antibody stability during the FD process and storage was analyzed. The stability of all the formulations was maintained during FD and varied among the different preparations and counter ions during storage. The sucrose-arginine-based mAb formulation showed significantly different degradation rates with varying counter ions during storage at 40 °C. Monoclonal antibody stability was greatly impacted by the type of counter ion used. The lowest degradation rate of mAb was observed in the formulation with chloride ions, followed by succinate, citrate, and phosphate ions. L-arginine-based formulations with added chloride ions were found to be the most effective in stabilizing a Monoclonal antibody, even at low temperatures, thereby preventing the structure from collapsing [46].
Startzel et al. further analyzed the L-arginine chloride-based formulation. The FD process parameters were then optimized and the concentration of excipients was varied, with and without the addition of BSA. The best results, characterized by minimal cake defects and optimal physical properties, were obtained once the ratio of protein to excipients was high. The impact of replacing BSA with a Monoclonal antibody was also evaluated and found that the protein's physical stability was improved under stressful storage conditions (i.e. 40 °C) in a sucrose-based formulation containing low L-arginine concentrations. Increasing the amount of L-arginine led to an elevation in cake defects. Proper optimization of the added L-arginine chloride concentration has great potential in extending shelf-life and improving the stability of protein formulated through FD [47].
Startzel et al. created a protein formulation using L-arginine and either mannitol or sucrose as an additive. Even at the highest L-arginine level employed in this investigation (mannitol:L-arginine of 2:1), partially crystalline solids were produced. Elegant cakes were produced by all formulations that comprised L-arginine. Only the L-arginine chloride and succinate-containing cakes had a surface "crust" created by phase separation. According to the X-ray powder diffraction, the ability of L-arginine to prevent mannitol from crystallizing differed depending on the type of counter ion present. Higher quantities of mannitol hemihydrate were obtained at high doses of L-arginine chloride because the counter ion impacted mannitol polymorphism [48].
Phenylalanine
It has been previously demonstrated that the incorporation of amino acids, such as phenylalanine, can assist in maintaining the integrity of IgG. The formulations examined found phenylalanine to be less effective than serine but more effective than glycine. Furthermore, trehalose was more successful in stabilizing IgG compared to amino acids [40]. The effect of combining trehalose and several types of amino acids, including phenylalanine, glycine, leucine, arginine, and cysteine, was analyzed by Emami et al. Similar to glycine and leucine, the use of phenylalanine, a hydrophobic amino acid, provides optimal stability of the IgG formulation. The addition of phenylalanine in the trehalose-based IgG formulation led to the best stability, followed by leucine and glycine [43].
Emami et al. also utilized phenylalanine in the development of an adalimumab aerosol formulation through the SFD method in another study. Phenylalanine promoted high stability, as demonstrated by the formulation's protection during the SFD process and storage. This result was also confirmed by the biological activity displayed. The aerodynamic testing conducted indicated the formulation containing phenylalanine to have the best aerodynamic characteristics, which correlates with the high FPF value obtained. Therefore, the phenylalanine addition in the formulation is suitable for anti-TNF-α therapy by pulmonary aerosol delivery [43].
Serine
Serine was also one of the amino acids included in Mirfakhraei et al.'s study. The powders containing serine were completely amorphous when examined at different temperatures. Out of the amino acids tested, serine was found to be the most effective, followed by phenylalanine and glycine, respectively. Trehalose was more effective in stabilizing IgG compared to amino acids [40].
Leucine
Emami et al. used leucine as one of the stabilizers in the IgG formulation produced through the SFD method. The reported results showed significantly better stability than the formulation without excipients. The formulation added with leucine had the second-best stability after phenylalanine. The use of these hydrophobic amino acids was suitable for IgG formulation, evidenced by the good stability provided for two months at 40 °C and 75% RH [43]. Emami et al. further used leucine in the microsphere formulation for adalimumab aerosol. The addition of various amino acids including leucine secured the stability of adalimumab during both the SFD process and storage. This optimal result was indicated by the high value of biological activity obtained in L-929 cells. The use of leucine in the formulation demonstrated other optimal results in the aerodynamic aspect, which were supported by the high FPF value of >60%. Therefore, this formulation shows great potential for inhalation delivery through the pulmonary system [43].
Sou et al. developed a Spray-Dried (SD) formulation of influenza antigen based on trehalose and leucine for aerosol powder vaccines. The results showed a glassy amorphous matrix and spherical particles. The amount of protein raises the water content and lowers the Tg value of the formulation, which are both drawbacks. SD vaccine does not cause lung parenchyma inflammation following pulmonary and subcutaneous immunization but produces high systemic and mucosal immunity comparable to the liquid vaccine. This study showed the potential efficacy of using an SD carrier as a system for delivering the influenza vaccine into the lungs [49].
Cysteine
The formulation comprising arginine, which is a polar sulfur-containing amino acid, exhibited aggregation and fragmentation. However, the presence of a free thiol group in cysteine caused it to act as a reducing agent, breaking apart the disulfide bonds between the heavy and light chains of IgG. The overall results showed that a mixture of trehalose and hydrophobic amino acids can be used to produce stable IgG formulations through the SFD method. The addition of hydrophilic and polar sulfur-containing amino acids in sugar-based formulations promotes IgG aggregation and fragmentation. The trehalose-based IgG formulation with the addition of cysteine is the only formulation with amino acid addition that resulted in fragment formation. The addition of other amino acids such as leucine, phenylalanine, arginine, and glycine only exhibited multimers but did not show any fragments after freeze-drying and storage. This result indicates that the addition of cysteine is not suitable for trehalose-based IgG formulations [43].
Histidine
Al-Hussein and Gieseler developed a histidine-based LDH formulation, which was compared with conventional formulations containing phosphate and citrate buffers to assess their impact on LDH stability. The formulation containing only histidine is more stable than other types, while stability of the component amino acids is affected by the concentration level. However, stability of the protein, including crystallized histidine, was affected by pH during FD preparation. The histidine integration into LDH formulation appeared to be a hopeful strategy for enhancing stability during the FD process. The use of histidine in addition to mannitol or sucrose, or even as the only excipients in the formulations, has shown promise in stabilizing protein during the drying process [50].
Blending of surfactants in sugar-stabilized therapeutic protein formulations
Blending of surfactants in sugar-stabilized therapeutic protein formulations is a common technique to improve stability. The surfactants addition can reduce protein aggregation, protect against their degradation, and prevent the formation of ice crystals during the FD process. The optimal blending ratio of surfactants and sugar depends on the type of surfactants, protein, and desired properties. The concentration of surfactants in formulations needs to be carefully controlled since their excessive amounts can negatively impact protein stability.
Polysorbate
Zhang et al. (2023) incorporated glycine and polysorbate into the sucrose-based mAb formulation. Adding 0.01% polysorbate 80 eliminated the crystallization effect of glycine. This led to improved stability of activity, irrespective of the sucrose and glycine proportion, as well as whether the formulation was annealed [41].
Pluronic
Ogawa et al. (2016) analyzed the effect of the drying method and addition of the pluronic F68 surfactants on the stability of a parainfluenza strain vaccine. The results indicated that the vaccine was most stable when surfactants were added and the foam drying method was employed, as shown by the tests conducted at 25 °C and 37 °C. In contrast, the lowest stability was observed in the surfactant-free formulation exposed to the SD method, while low stability was also detected in the FD preparation even with the addition of surfactants [51].
Eilts et al. (2023) further analyzed the effect of formulation on Orf viral vector stability. Results showed that the amount of sucrose in the formulation played a significant role in enhancing Orf viral vector stability. Moreover, the impact of drying method variations on stability depended on the formulation's components. Specifically, in the formulation with a 5:1 ratio of sucrose, the addition of pluronic F68 was found to reduce the rate constant value for physical aggregation. In addition, this formulation obtained the most optimal preserved infectivity [52].
Blending of polyol in sugar-stabilized therapeutic protein formulations
The blending of polyol, such as mannitol or trehalose, into protein formulation, has shown the potential to improve stability during storage and processing. The presence of polyol tends to prevent aggregation and degradation by creating a more stable environment and reducing water activity. In addition, the incorporation of polyol can help to maintain the protein's native structure and functionality. The blend's effectiveness depends on various factors, including polyol type and concentration, the kind of protein being stabilized, and processing conditions [53].
Mannitol
As previously mentioned, the incorporation of mannitol in the sucrose/trehalose-based formulation of IPV improved its stability in comparison to conventional liquid formulations, evidenced by the minimal loss of potency. The improved stability means there is no need for refrigeration at room temperature [42,54]. Several combinations with polyol and arginine were further tested in trehalose-based protein formulations using the SFD method to observe how they affected the protein stability and properties. The best value for moisture content was found in formulations containing mannitol. Moreover, mannitol has been proven to have the greatest moisture resistance out of the four sugar/polyol excipients evaluated, and it can effectively offset the high moisture uptake by hygroscopic arginine. The ratio of trehalose and mannitol is nonlinearly associated with the physicochemical properties of the resultant protein [45]. In a subsequent investigation, trehalose and mannitol ratio adjustments in the arginine-based formulation were compared and examined for their impact on BSA stability. The ratio variations ranged from 30:70 to 80:20. It was discovered that when subjected to high humidity and temperature during pre-reconstitution storage, formulations with relatively high quantities of trehalose (30-40 wt%) failed to decrease protein aggregation. To create an effective protein formulation incorporating L-arginine, the concentration of trehalose must be decreased while the mannitol concentration should be increased. Likewise, the mannitol-to-trehalose ratio needs to be maintained above 1 [45].
Startzel et al. developed a protein formulation based on L-arginine with the addition of mannitol or sucrose. Partial crystal formation was achieved even with the highest concentration of L-arginine (at a mannitol to L-arginine ratio of 2:1). The X-ray powder diffraction analysis revealed that L-arginine was more effective in preventing mannitol crystallization compared to sucrose, and its effect varied based on the type of L-arginine counter ion present. The counter ion influenced the polymorphism of mannitol, and higher levels of mannitol hemihydrate were observed with higher levels of L-arginine chloride [48].
Al-Hussein and Gieseler also conducted a study which developed histidine-based LDH formulation. Sucrose or mannitol was used as a stabilizer. Formulations added with mannitol alone showed lower stability compared to only histidine, but the combination with histidine was more stable. The histidine-based LDH formulation containing mannitol exhibited protein stabilizer potential during the FD process [50].
Sorbitol
The use of a combination of several excipients including sorbitol, mannitol, and myo-inositol in a sucrose/trehalose-based formulation for IPV produced optimal results. This is evidenced by the minimal loss of potency despite the drying and storage at higher temperatures. The formulation has the potential to be stored without special conditions, similar to other cold-chain products [42].
Inositol
The use of polyol including myo-inositol in the sucrose/trehalose formulation showed better stability results compared to conventional formulations, meaning that refrigeration is not needed [42].
Conclusion
In conclusion, saccharides or sugars are widely used as protein additives or stabilizers in the pharmaceutical industry, and their ability to maintain the stability of therapeutic proteins during drying and storage has been demonstrated. Sugars are capable of creating a glass matrix, known as sugar glass that can coat proteins and protect them from degradation. The ideal preservation process for solid formulations needs to incorporate cryopreservation and desiccation techniques to ensure the triggering of protein structure unfolding. The utilization of sugars requires carefully considering various factors such as the number of reducing groups and the Tg value. Sugars containing a high number of reducing groups may indicate a Maillard reaction, and a low Tg value may reduce the protective effects. Protein stability is primarily controlled by the vitrification mechanism at a Tg below the storage temperature. When the Tg value of the sugar-protein system exceeds the storage temperature, the protein molecules’ mobility is lost, and their stabilization is predominantly governed by water replacement. Using the maximum amount of sugar capable of forming hydrogen bonds with proteins can lead to a higher level of stability. On the other hand, sugar flexibility contributes to the extent of stabilization, as demonstrated by the application of small sugars with low Tg values in previous studies to stabilize solid proteins. The combination of small sugars and polysaccharides presents an opportunity to further enhance solid proteins’ stability. The impact of excipients blending on sugar-stabilized therapeutic protein refers to the effect that results from combining different excipients to improve protein stability. Excipients are added to protein formulations to improve their preservation during storage and transportation, and the type, quantity, and combination of excipients can significantly affect the stability of a sugar-stabilized therapeutic protein. This can result in changes to the protein's conformation, aggregation, and degradation rate. Choosing the appropriate excipients and blending can play a critical role in determining the quality and shelf-life of protein products.
Careful selection and optimization of the right combination are crucial to ensure successful stabilization because not all excipients are suitable for every protein. The blending process involves mixing protein with one or more excipients, such as sugar or polyol, to prevent degradation during storage and handling, which stabilizes the protein's native structure and prevents denaturation, aggregation, or chemical degradation. In addition, blending can enhance protein solubility, bioavailability, and delivery. To develop more effective formulations for therapeutic use and optimize protein stabilization, future studies should explore other excipient blends and surfactants.
Acknowledgements
Special thanks are extended to Dr. W.L.J Hinrich from the Groningen Research Institute of Pharmacy, University of Groningen, the Netherlands for his invaluable contributions and insights.
Disclosure Statement
No potential conflict of interest was reported by the authors.
Funding
The authors express sincere gratitute to Universitas Indonesia for funding this study through the PUTI Grant with contract number NKB-5/UN2.RST/HKP.05.00/2020.
Authors' Contributions
All authors contributed to data analysis, drafting, and revising of the paper and agreed to be responsible for all the aspects of this work.
ORCID
Cynthia Marisca Muntu
https://orcid.org/0009-0002-2485-8223
Christina Avanti
https://orcid.org/0000-0002-1770-8684
Hayun Hayun
https://orcid.org/0000-0002-1495-6228
Silvia Surini
https://orcid.org/0000-0003-1211-9706
HOW TO CITE THIS ARTICLE
Cynthia Marisca Muntu, Christina Avanti, Hayun Hayun, Silvia Surini. Impact of Excipients Blending on Sugar-Stabilized Therapeutic Proteins. J. Med. Chem. Sci., 2024, 7(6) 795-817.