Document Type : Case Report
Authors
1 Resident, Department of Dermatology and Venereology, Faculty of Medicine, Universitas Airlangga, Dr. Soetomo General Academic Teaching Hospital, Surabaya, Indonesia
2 Dermatovenerologist, Department of Dermatology and Venereology, Faculty of Medicine, Universitas Airlangga, Dr. Soetomo General Academic Teaching Hospital, Surabaya, Indonesia
Abstract
Introduction: Bullous pemphigoid (BP), an autoimmune condition that mostly affects the elderly and is marked by subepidermal blisters. Although the pathophysiology of this condition is still not entirely known, it is thought to have a close relationship with a number of medications. The treatment effectiveness is dependent on the severity of the patient's ailment and comorbidities.
Case: A 76-year-old man with many blisters and bodily erosions. The patient expresses discomfort and itching. Upon physical examination, it is seen that the upper and lower extremities have a large number of tense-walled bullae with numerous erosions and crusts. Subepidermal blistering with eosinophilic and lymphocytic infiltrate is visible on skin biopsy. Desoximetasone was applied topically to him, and oral methylprednisolone (MP) was administered. MP is gradually reduced in accordance with the patient's development. Treatment for hypertension was also suspended till the condition became better. The patient's condition improved as a result, and no new blisters appeared.
Discussion: The best methods for ruling out other blistering diseases when diagnosing bullous pemphigoid, besides clinical symptoms, continue to be direct immunofluorescence and skin biopsy. Other than a mix of oral and topical glucocorticoids for the treatment of drug-induced BP, stopping some medications will have an immediate effect and is typically more responsive. Reducing the dosage and overall duration of oral glucocorticoids is crucial since systemic glucocorticoids can have some negative effects, particularly in the elderly.
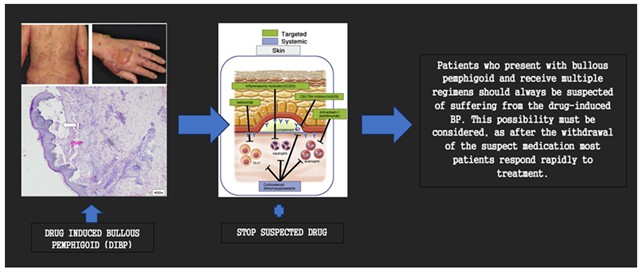
Conclusion: Bullous pemphigoid patients should constantly be wary of drug-induced bullous pemphigoid when receiving several therapies. Because most patients respond quickly to treatment after the suspected substance is stopped, this possibility should be taken into account.
Graphical Abstract
Keywords
Introduction
Bullous pemphigoid (BP) is the most common autoimmune disease among the elderly [1], primarily affecting those above 60, with a peak in the 70s. It can also occur in young adults, children, and drug-induced cases. BP is not linked to specific races, ethnicities, or genders [2-4]. About 20% of cases occur [5]. BP is characterized by skin and mucosa inflammation, caused by autoantibodies targeting BP180 and BP230 proteins, leading to skin blisters [6, 7]. It should be considered as a differential diagnosis in older patients with pruritus [1]. The BP pathophysiology is influenced by genetic factors, skin changes due to aging, and specific triggers, including drugs. Drug-induced BP (DIBP) is a similar condition caused by specific medicines [7]. Particularly in the elderly [5] autoimmune bullous disease greatly contributes to illness and mortality. A recent research of 502 patients in France found that BP had a mortality rate of 38% after one year, making it a potentially lethal condition [8]. The mortality rate from BP is mostly influenced by the age of onset and the systemic medication used, rather than by the severity and scope of PB [5]. Therefore, in this situation, effective treatment is crucial. The severity of condition and the patient's comorbidities are two aspects that affect how to treat BP [4]. The primary line of defense against bullous autoimmune disorders is corticosteroids. The specific bullous condition and the severity of it are taken into account while treating with corticosteroids. Osteoporosis, diabetes, and immunosuppression are more prevalent and potentially fatal side effects of systemic glucocorticoid therapy in the elderly.
Therefore, every effort should be made to minimize the total dose and duration of oral corticosteroids [9]. Drugs can also induce BP, although they have a better prognosis and offer great therapeutic results after the offending medication is stopped [1]. Here, we present the case of an older patient who developed bullous pemphigoid due to a pharmacological interaction and was successfully treated with oral corticosteroids before the offending medication was discontinued.
Case report
Clinical presentation
The emergency room received a visit from a 76-year-old man with a history of hypertension who had a tight blister eruption all over his body.
Two weeks before to admission, blisters on the hands migrated to other parts of the body and got worse and more irritating.
He has never before taken medication or seen a doctor. The itching persisted, so the patient visited the doctor. The doctor gave the patient an injectable prescription (unknown medicine name), which temporarily stopped the irritation before it returned and was followed by blisters. Prednisone 2×2, interesting 3×1, and co-amoxiclav 3×1 were prescribed when the patient visited Haji Hospital a week later, but there was no improvement. They experience agony in the injured skin as the blisters grow and some of them break open into sores. Furthermore, the patient reported having a temperature and ongoing itching. Lips and genitalia are free of lesions. The patient is able to eat and drink without feeling queasy or throwing up. The patient had been using candesartan, amlodipine, Concor, spironolactone, and nitrocaf regularly for ten years due to his hypertension. Malignancy or tumor denial and history of the same illness. The family made no other such concerns.
Body weight was 80 kg, blood pressure was 150/90 mmHg, pulse rate was 115 beats per minute, respiration rate was 20 beats per minute, and body temperature was 39 ᵒC. These measurements were taken during a physical examination of general health. There were several erosions in the face area that were partially covered by crusting, but there was no conjunctival hyperemia or hemorrhagic crust on the lips (Figure 1a). There were numerous bullae with tight walls that were challenging to break, filled with clear liquid, and measuring 2-3 cm in diameter in the region of the thorax, abdomen, and extremities (Figures 1b, 1e, and 1f). Numerous erosions were present in the antebrachii sinistra et dekstra and the region of the brachialis (Figures 1c, 1d). There was no mousy odor, a negative Nikolsky sign, and a positive Asboe-Hansen sign. The genital region showed no signs of deterioration.
Diagnosis
Direct immunofluorescence analysis (DIF) and standard microscopy were both done on a punch biopsy. Eosinophils, lymphocytes, and hyperkeratosis were seen in the material, which showed subepidermal bulla development (Figure 2a). IgG and C3 were not linearly detected by direct immunofluorescence along the dermal-epidermal interface (Figure 2b). A blood culture did not reveal any signs of bacterial development. The lesion pus culture included the Gram-positive cocci Staphylococcus aureus. Bullous pemphigoid with secondary infection and under control hypertension was determined to be the working diagnosis based on the history, physical examination, and laboratory examination.

Figure 1: The clinical appearance revealed many types of erosion in the facial region (A), multiple erosions and crust on the superior and inferior extremities, and numerous bullae with walls that were tense and transparent liquid in some areas (B, C, D, E, and F).

Figure 2: Skin pathology and DIF revealed subepidermal blisters with eosinophil and lymphocyte infiltration in the dermis layer (A) and DIF failed to reveal bullae with IgG and IgA deposition (B)
Treatment approach
Based on the patient's body weight (0.75 mg/BW prednisone), 48 mg of methylprednisolone (MP) were administered orally to the patient for two weeks before tapering off once blister formation had stopped. In addition, the patient received topical steroid desoximetasone twice daily, oral clindamycin 300 mg, cetirizine 10 mg, and 500 mg of paracetamol if necessary. The course of treatment also included atrial fusidate cream for the erosion and wound care with NaCl compresses. In addition, until the patient's condition became better, the hypertension medication was temporarily withdrawn. The back and chest had only erythematous lesions one month after starting treatment, and the clinical look had completely disappeared (Figure 3).

Figure 3: The complete resolution of bullous pemphigoid on the face, upper extremities, and trunk were observed
Results and Discussion
This case presents a 76-year-old male patient who exhibited widespread blister formation. Bullous pemphigoid (BP) predominantly affects the elderly population, with the majority of patients experiencing the onset of the disease during their eighth and ninth decades of life. The average age at which BP is diagnosed often falls between 75 and 80 years [10, 11]. A retrospective study was conducted in two regions of Germany to examine the incidence of BP based on age and sex. The findings revealed that the likelihood of developing BP increased significantly in individuals aged 60 years and above. Moreover, patients aged over 90 years exhibited a substantially higher risk, estimated to be approximately 300 times greater, compared to those aged 60 years or younger. This study provides additional evidence to support the assertion that BP should not be classified as an uncommon condition among older individuals [12].
Bullous pemphigoid (BP) is an autoimmune blistering disease characterized by tight subepithelial blisters, often appearing on erythematous or normal-looking skin [13]. It may involve mucosal membranes in about 10-30% of patients [14]. The onset of blisters is preceded by a prodromal phase with non-bullous lesions, lasting weeks or even months, making clinical diagnosis challenging. Pruritus severity varies and can occur without cutaneous symptoms. Excoriation, eczema, urticaria, and hemorrhagic crusts may appear during this phase. In the bullous stage, severe itching and multiple tight blisters and vesicles occur on normal or reddened skin [15]. The intensity of itch can precede blister formation over weeks, months, or even years [16]. In individuals over 70 with persistent pruritus and no identifiable systemic cause, considering BP in the differential diagnosis is essential [1]. In one case, a patient developed blisters on the hands that spread, leading to increasing pain and pruritus. The patient initially experienced generalized pruritus for three months and sought medical attention a month ago, receiving an undisclosed injectable pharmaceutical that temporarily relieved itching but was followed by blister formation. Mucosal involvement is observed in 10-35% of cases, most frequently affecting the buccal mucosa with localized blisters or erosions that typically do not scar [5]. The patient in this case did not exhibit blistering or erosion in the conjunctiva, oral cavity, or vaginal region. Many cases of BP lack a clear cause, but a detailed medical history examination can help identify contributing factors, including concurrent medical issues and prescribed drugs [7]. Systemic therapy is often associated with BP, with over 50 medicines potentially linked to its onset [12]. Understanding the exact cause of drug-induced BP, especially in older patients taking multiple medications, is challenging [3]. Drugs containing sulfhydryl groups, phenol ring compounds, angiotensin-converting enzyme inhibitors, non-steroidal anti-inflammatory drugs, immunomodulators, and TNF-a inhibitors are frequently implicated in drug-induced bullous pemphigoid [4]. Genetic predisposition may play a role in drug-induced BP, as individuals with genetic susceptibility exposed to certain drugs can disrupt their immune system, leading to autoantibody production [1]. The ''two-step'' concept suggests that exposure to two medications with similar molecular structures initiates and amplifies the immunological response, ultimately leading to BP development [5].
The patient had a history of hypertension and was taking candesartan, amlodipine, concor, spironolactone, and nitro car. ). A number of case-control studies have suggested a robust correlation with aldosterone antagonists. The results of the multivariate analysis conducted in these studies indicate a substantial association between the long-term use of spironolactone and BP, with odds ratios ranging from 2.3 to 3.1. However, a retrospective study did not find a significant association when adjusting for factors like age and co-morbidities [15].
Diagnosis of BP employs clinical, histological, and immunopathological methods using direct or indirect immunofluorescence. Prolonged treatment can reduce the frequency of positive immunofluorescence results [9, 16].
Figure 4: List of reported drugs to induce bullous pemphigoid [7]
|
Likely associationa |
Probable associationb |
Uncertain associationc |
|
Alogliptin Anagliptin Aspirin Biostim® D-Penicillamine Enalapril Erlotinib Etanercept Everolimus Furosemide Ibuprofen Levofloxacin Linagliptin Nivolumab Phenacetin Psoralens with UVA Rifampicin Serratiopeptidase Sirolimus Sitagliptin Teneligliptin Tetanus toxoid Tiobutarit Vildagliptin
|
Actinomycin-D Adalimumab Amoxicillin Ampicillin Arsenic Atezolizumab Bumetanide Celecoxib Cephalexin Chloroquine Ciprofloxacin Diclofenac Dorzolamide Durvalumab Fluoxetine Gabapentin Griseofulvin Hepatitis B vaccine Hexavalent combined vaccine Hydrochlorothiazide Infliximab Ipilimumab Lisinopril Losartan Mefenamic acid Metamizole Metronidazole Penicillin Rosuvastatin Spironolactone Sulfasalazine Terbinafine Ustekinumab |
Aldesleukin (IL-2) Amantadine Amlodipine Anthralin (dithranol) Azapropazone Captopril Coal tar Complementary medicines Dabrafenib Doxepin Enoxaparin Escitalopram Fluorouracil Flupenthixol Herpes zoster vaccine Influenza vaccine Iodide Levetiracetam Mesalazine Nadolol Nifedipine Novoscabin (benzyl benzoate) Omeprazole Placental extracts Photodynamic therapy Risperidone Rotavirus vaccine Sulfonamide Swine flu vaccine Timolol Valsartan |
aRecurrence or exacerbation with rechallenge supports association with medication, or recognised by extensive body of evidence (e.g. gliptins); bTemporal relationship with initiation of medications in younger individuals, or spontaneous resolution with the cessation of the offending medication; cTemporal relationship with the initiation of medication in older individuals. Colour indicate presence of functional groups: thiol (red), phenol (blue), non-thiol non-phenol (black).
Initial bullae histopathology shows subepidermal blisters with eosinophils, neutrophils, and inflammatory infiltrate, primarily consisting of eosinophils and neutrophils, which lacks specificity in the nonbullous phase [9, 17, 18]. A skin biopsy on a recent blister reveals a diagnostic pattern, with fine, linear IgG and/or C3 deposition at the dermal-epidermal interface, occasionally resembling IgA and IgE patterns [9, 17, 18]. Primary BP antigens are bullous pemphigoid antigen 1 (BP230) and bullous pemphigoid antigen 2 (BP180) [19]. In this case, a punch biopsy was performed, revealing subepidermal bullae with eosinophils, lymphocyte hyperkeratosis, and mild acanthosis. However, direct immunofluorescence analysis did not detect linear IgG and C3 deposition along the dermal-epidermal contact. Another challenge lies in categorizing older adults with generalized pruritus, with or without skin lesions, displaying circulating autoantibodies targeting the epidermal basement membrane zone (BMZ) and reactivity with BP180 and/or BP230. Conventional immunofluorescence microscopy may not yield affirmative results in these cases, cautioning against sole reliance on circulating autoantibodies as a diagnostic criterion for bullous pemphigoid (BP). Some individuals with initially negative direct immunofluorescence microscopy results may later develop BP, falling under the classification of incipient pemphigoid [12].
Despite extensive research on bullous diseases, the precise identification of distinct antigens associated with drug-induced bullous pemphigoid (DIBP) remains elusive. There is a current hypothesis suggesting that DIBP antigens may bear similarity to those observed in naturally occurring bullous pemphigoid, as documented in prior studies [19].
"Drug-induced Bullous Pemphigoid (DIBP)" refers to cases resembling the idiopathic form, triggered by specific medications, mostly orally but occasionally topically. When assessing older patients with medication changes, consider potential drug-induced effects. DIBP cases vary in clinical presentation compared to idiopathic BP, often complicating diagnosis. Histological features in drug-induced BP (DiBP) differ from idiopathic BP (IBP), but specific antigens are not recognized, suggesting partial overlap. Immunofluorescence patterns resemble those in IBP. The natural history of DABP is currently under investigation, with ongoing efforts to define its trajectory. As demonstrated in Figure 5, two distinct trajectories have been identified. One is an acute, self-limited manifestation with decisive remission upon medication cessation, resembling a true drug reaction. The other is a chronic form akin to idiopathic BP, persisting after the causative factor's removal and necessitating extended treatment measures for control [7, 19].
The main goal in BP treatment is effectively managing clinical manifestations. Treatment choice depends on factors like disease extent and the patient's overall health. BP can be categorized as mild, moderate, or severe. Mild cases can use superpotent topical corticosteroids like clobetasol propionate cream. A daily dosage of 20-30 grams is suggested for general involvement. Topical therapy can be applied directly to the affected area in limited cases. In severe instances, oral corticosteroids, while historically used, have risks, especially for older patients. Some studies favour topical corticosteroids over oral steroids. Adherence may be challenging, particularly in older patients.
Table 2. Differences between drug-associated bullous pemphigoid and idiopathic bullous pemphigoid [7]
|
|
Drug-associated bullous pemphigoid |
Idiopathic bullous pemphigoid |
|
History |
Patient is often subject to polypharmacy. Recently introduced drug therapy |
Patient may be subject to polypharmacy No recently introduced drug therapy |
|
Clinical findings
|
Tendency toward younger age groups Lesions on normal appearing skin May resemble other entities such as erythema multiforme, or pemphigus Mucosal involvement may be present May have involvement of palms and soles Nikolsky signa may be positive |
Tendency towards older age groups Lesion on erythematous or urticarial base Often classic features of pruritus followed by development of large tense bullae Mucosal involvement is very rare Frequent involvement of the extremities Nikolsky signa is negative |
|
Investigations
|
Prominent eosinophilic infiltrate May be evidence of necrotic keratinocytes May be evidence of intraepidermal vesicles Formation of thrombi may be apparent. Marked eosinophilia present in serum |
Eosinophilic infiltrate may be apparent Rarely evidence of necrotic keratinocytes No evidence of intraepidermal vesicles Formation of thrombi is seldomly observed Eosinophilia present in serum |
|
Course |
Responds promptly to treatment with topical, or systemic corticosteroids May improve with cessation of inciting agent Relapses may occur (i.e. drug-triggered) May rarely lead to scarring |
May exhibit a protracted course despite systemic corticosteroid therapy No provocative agent is identified Relapses often occur Scarring not a prominent feature |
aNikolsky sign has been of diagnostic importance in pemphigus, although concern has arisen regarding the sensitivity and specificity of the technique in particular in terms of the operator dependence and interpretation
Consider systemic steroids like oral prednisone at 0.5-1 mg/kg/day as necessary. Oral corticosteroids, while widely used, carry risks like hypertension, diabetes, and osteoporosis. Adjuvant therapies like azathioprine and mycophenolate mofetil are options for refractory BP, but they can be expensive [4, 18, 21-23]. The elderly patient with extensive BP and hypertension received methylprednisolone at 48 mg/day, gradually tapered as lesions healed. The treatment included desoximetasone, clindamycin, cetirizine, and paracetamol. Intermittent urticated lesions were acceptable [12]. Systemic corticosteroid treatment at 0.5-1 mg/kg/day is recommended for severe BP cases [17, 24]. Studies vary in treatment protocols. Prednisolone and prednisone are common choices. The dose often ranges from 40-80 mg/day, but lower initial doses (20-40 mg/day) have been suggested, with severity somewhat linked to corticosteroid quantity [16].
Treatment for classic bullous pemphigoid relies on uncontrolled research using oral and topical corticosteroids along with adjuvant medications [16]. Managing drug-induced BP includes promptly stopping the causative medication. Patients with DIBP respond well to low-dose oral corticosteroids (prednisone at 0.5 mg/kg/day) and potent topical corticosteroids, with temporary suspension of hypertension therapy [19]. Gradual corticosteroid dosage reduction involves decreasing by 10 mg/day, tapering at slower paces for lower doses, and ultimately reducing to 1 mg/week when under 10 mg/day. Studies show systemic glucocorticoids control moderate or severe BP in 95% and 91% of cases after a 21-day treatment [20]. In this scenario, blisters cease within a week, and erosions heal after eight days. The methylprednisolone dosage is reduced from 48 mg/day to 12 mg/day upon discharge [16].
Bullous pemphigoid is often characterized by a self-limiting nature, although its clinical progression might extend over several months to years, marked by periods of remission and relapse [25]. Furthermore, it should be noted that BP has the potential to result in considerable morbidity, such as the development of eroding lesions and impetiginized wounds [18]. The death rate associated with BP is mostly influenced by the age at which the disease manifests and the systemic treatment provided, rather than the severity of BP condition [5]. The study presented findings on the 1-year mortality rate of bullous pemphigoid, which was reported to range from 20% to 40% [26]. A retrospective study conducted in the United Kingdom revealed that people diagnosed with BP have a death rate that is twice as high as that observed in a control group consisting of healthy persons. Several studies have documented a 1-year death rate of 19% among patients diagnosed with BP in both the United States and the United Kingdom [4]. Hypertension is a chronic condition that necessitates ongoing monitoring and therapy until patients achieve full remission and discontinue all therapeutic interventions. It is imperative to conduct regular evaluations to ascertain that patients are not being prescribed excessive doses of topical or systemic medication beyond what is required to adequately manage their condition. The presence of sporadic urticated lesions or blisters is deemed acceptable and serves as an indication that the patient is not being excessively treated. It is recommended that a gradual decrease in medication be considered every 1-2 months for patients who are stable. This adjustment should be based on clinical judgment rather than relying solely on inflammatory markers. The patient underwent a follow-up visit at our outpatient clinic. The patient exhibited favorable recovery following the administered medication, as evidenced by the lesions amelioration and the absence of any new blister formation.
Bullous pemphigoid is a potentially life-threatening condition that has a significant prevalence among individuals. Its curability is contingent upon the administration of appropriate care; however, this is not consistently achieved. The objective of this case study is to enhance knowledge on the adverse health effects associated with the illness, emphasize the various treatment options that are currently accessible, and promote the importance of seeking consultation with a dermatologist, considering the possible seriousness of the condition. There is yet no identified biomarker that is distinct to drug-induced BP changes. Furthermore, the clinical presentation and immunopathological findings of drug-induced BP changes may be indistinguishable from those of the classic variety. The disease's commencement at a younger age and typically within a span of around three months following the administration of a novel medication, accompanied by swift disease management and a reduced frequency or absence of subsequent occurrences upon discontinuation of the implicated drug, may indicate its potential as a precipitating factor [1].
Conclusion
The identification of pharmaceuticals linked to BP enables medical practitioners to promptly recognize probable instances of drug-induced BP (DIBP) and discontinue the administration of the implicated medications. The objective of treatment for all individuals with BP should encompass the restoration of skin barrier function. The aforementioned approach has demonstrated notable advantages in the geriatric population, characterized by a higher prevalence of concurrent medical conditions that restrict the range of available therapeutic interventions.
Acknowledgements
I would like to acknowledge and give my warmest thanks to my supervisor Dr. Linda Astari, Sp.DVE(K) who made this work possible. Her guidance and advice carried me through all the stages of writing my report.
Disclosure Statement
No potential conflict of interest was reported by the authors.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Authors' Contributions
All authors contributed to data analysis, drafting, and revising of the paper and agreed to be responsible for all the aspects of this work.
ORCID
Linda Astari
https://orcid.org/0000-0002-2939-1493
Erik Rizka Yulianggoro
https://orcid.org/0009-0001-6022-7421
HOW TO CITE THIS ARTICLE
Erik Rizka Yulianggoro, Linda Astari*. Drug-Induced Bullous Pemphigoid: Rapid Resolution with Corticosteroid Therapy. J. Med. Chem. Sci., 2024, 7(2) 300-310.