Document Type : Original Article
Authors
- Mohamed J. Saadh 1, 2
- Faris Anad Muhammad 3
- Romina Mahdavi 4
- Yasaman Nazariyan Parizi 4
- Ebrahim Balali 5
- Juan Carlos Cotrina-Aliaga 6
- Mohaned Adil 7
- Salam Ahjel 8
- Meryelem Tania Churampi Arellano 9
- Yanina Gallardo-Lolandes 10
- Renato R. Maaliw III 11
- Mahmoud Mirzaei 12
- Kun Harismah 13
1 Faculty of Pharmacy, Middle East University, Amman, 11831, Jordan
2 Applied Science Research Center. Applied Science Private University, Amman, Jordan
3 Department of Pharmacy, Al-Noor University College, Nineveh, Iraq
4 Department of Pharmaceutical Chemistry, Faculty of Pharmaceutical Chemistry, Tehran Medical Sciences, Islamic Azad University, Tehran, Iran
5 Department of Organic Chemistry, Faculty of Pharmaceutical Chemistry, Tehran Medical Sciences, Islamic Azad University, Tehran, Iran
6 Faculty of Engineering, Universidad Peruana los Andes, Huancayo, Peru
7 Pharmacy College, Al-Farahidi University, Baghdad, Iraq
8 Department of Pharmacy, Al-Zahrawi University College, Karbala, Iraq
9 Department of Industrial Engineering, Universidad de Lima, Lima, Peru
10 Faculty of Nursing, Universidad Privada San Juan Bautista, Lima, Peru
11 College of Engineering, Southern Luzon State University, Lucban, Quezon, Philippines
12 Laboratory of Molecular Computations (LMC), Department of Natural and Mathematical Sciences, Faculty of Engineering, Tarsus University, Tarsus, Turkiye
13 Department of Chemical Engineering, Faculty of Engineering, Universitas Muhammadiyah Surakarta, Surakarta, Indonesia
Abstract
Density functional theory (DFT) calculations were performed to investigate the features of vitamin B3 (Vit-B3) in oil and water environments. Two up and down structural conformations were found based on the orientation of hydrogen atom of attached carboxylic acid group to pyridine scaffold, in which the up-conformation was found more suitable than the down-conformation. The models were stabilized in gas phases and 1-octanol and water solvents environments to explore the partition coefficient (LogP) for each conformation. In addition, the electronic features were investigated based on frontier molecular orbital levels. The results of this work indicated a higher suitability of formation for the up-conformation in all three environments and the highest suitability of formation of both up and down conformations in water medium. Accordingly, the LogP value was found smaller than one indicating watery tendency for the models. As a final remark, the structural and electronic features of Vit-B3 indicated insights into its development for further applications.
Graphical Abstract
Keywords
Main Subjects
compounds are very important to be considered [5-8]. Indeed, varieties of diseases, infections, and accidents always make the topic of such biomedical explorations very important regarding the maintenance of human health system [9-12]. Among those topics, learning details of such compounds could help to provide new insights into the development of new compounds and their prospective applications [13-16]. Vitamin B3 (Vit-B3) is among those compounds with significant biological activities such as an antilipemic drug, a vasodilator agent, and an antidote [17, 18]. Vit-B3 or nicotinic acid or niacin with the formula C6H5NO2 (Figure 1) is a carboxylic acid derivative of pyridine heterocyclic scaffold, in which it could be available as a single standing structure or in combination with other structures [19]. Furthermore, it could work as an inhibitor against the nicotinamidase enzyme [20]. Vit-B3 could be also involved in DNA repairing process by working as a precursor for nicotinamide adenine dinucleotide (NAD) and nicotinamide adenine dinucleotide phosphate (NADP) [21]. Vit-B3 is indeed a form of vitamin B3 with dominant roles for maintain the human health system in lowering low density lipoprotein (LDL) cholesterol and raising high density lipoprotein (HDL) cholesterol [22-24]. Naturally, it could be biosynthesized from the tryptophan amino acid in plants and animals used as a dietary supplement [25]. Deficiency of Vit-B3 could lead to appearance of severe effects in human and supplying additional sources in daily dietary could overcome on this issue [26-28]. Vit-B3 has been prescribed as a medication for years. However, it still shows adverse side effects such as hepatotoxicity and an increased risk of diabetes type-II [29]. Accordingly, several efforts have been dedicated to improve the efficacy and safety of Vit-B3 consumption [30-32]. In this regard, analyzing the features of drug substance in oil and water solvent environments could reveal insights for developing its further applications [33]. It should be noted that availability of all vitamin contents in human body is very important and learning their features could help to make synthetic forms of vitamins for further complimentary applications and treatments [34-36].
The term of partition coefficient (LogP) meaning the solubility of a substance in oil and water solvent environments is very important for recognizing crucial features of the pharmaceutical agents [37-39]. However, several efforts are still required to develop efficient drug substances and treatments protocols [40-43]. To this aim, the current research work was done to investigate Vit-B3 in 1-octanol as representing the oil medium and water solvent environments to provide insights into the Vit-B3features. Density functional theory (DFT) calculations were performed to obtain the required results of structural and electronic features at molecular and atomic scales [44-48]. Several methodologies have been developed to investigate various aspects of chemical and biological related systems during recent decades [49-52]. Not only the experimental methods, but also several other computational or theoretical methods and protocols have been developed up to now [53-56]. However, the obtained successes are not certain yet as new diseases or the older ones will become wild time by time [57-60]. In this regard, the modelling procedures and computer-based evaluated descriptors could help to recognize the characteristic features of molecular systems for approaching their further applications especially in the field of computer-aided drug design [61-64]. Accordingly, Vit-B3 was carefully examined in this work for leaning new details based on the impacts of environmental issues on its original features. Earlier works indicated benefits of employing DFT calculations to explore details of such small heterocyclic molecular models for learning their further featured details [65-68]. Not only the small molecules, but also complexes and larger molecules are suitable to be investigated by employing such types of DFT calculations [69-72]. Especially in the case of exploring impacts of environments on molecular properties, such DFT evaluated features could help to learn more details [73-75]. The materials, graphical representations, and obtained results of this work were exhibited in Figures 1 and 2 and Tables 1 and 2 to be discussed for assessing Vit-B3 in 1-octanol and water solvent environments.

Materials and Methods
The 3D structure of Vit-B3 was obtained from the ChemSpider structural bank with ID: 913 [76]. As it could be found by two up and down orientations of hydrogen atom of carboxylic group (Figure 2), two up and down conformations were considered for Vit-B3 in all calculations. Both conformational models were optimized in gas, 1-octanol, and water solvent environments at the B3LYP/6-31+G* level of DFT calculations using the Gaussian program [77].

Next, the models were re-calculated to evaluate their characteristic features based on the same employed solvent environments and method. As a result, the stabilized structures and their electronic features were evaluated for assessing Vit-B3 in different solvent environments.
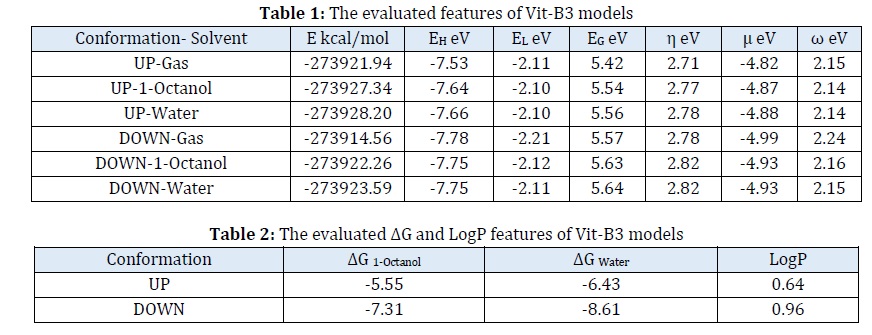
The stabilized structures and their electrostatic potential (ESP) surfaces were depicted in Figure 2. The calculated values of total energy (E) and molecular orbitals features including energy levels of the highest occupied and the lowest unoccupied molecular orbitals; HOMO and LUMO, (EH and EL), energy gap (EG), chemical hardness (η), chemical potential (μ), and electrophilicity index (ω) using Equations 1-4 were summarized in Table 1 [78].

The values of calculated Gibbs’ free energy (G) of molecules in gas phase and each of 1-octanol and water solvent environments were converted into ΔGSolvent using Equation 5. Next, the values of LogP were obtained using Equation 6 at the room temperature (T) 298.15 K and the general gas constant (R) 0.00199 kcal/Kmol [79]. The results of ΔG and LogP were summarized in Table 2.

Results and Discussion
This work was done for providing insights into a form of vitamin B3; nicotinic acid (NA), in oil and water environments. It was earlier mentioned that explorations of biological related systems are very important due to several reasons from appearing the new diseases or the wildness of already known ones [80-83]. Not only the direct treatments of patients, but also several other issues are needed for dealing with the biological systems for keeping the human health systems and levels [84-87]. Naturally originated structures are further very interesting to be investigated for their further developments [88-90]. In this regard, the current work was done to explore such important topic for the case of Vit-B3 in different media. The heterocyclic structures are very important to be investigated because of their unique structural features especially for initiating drug design and development processes. Accordingly, their features in different environment and detecting the effects of such environments are very important to be learned. As known, Vit-B3 is a carboxylic acid derivative of pyridine (Figure 1) as a single standing compound or it could be connected to other chemical substances and structures. The 3D structure of Vit-B3 was obtained and geometry optimization was done based on DFT calculations. Interestingly, two up and down conformations were found by the orientation of attached hydrogen atom of the carboxylic acid group, in which the stabilized conformations were demonstrated in Figure 2. This achievement showed that the small hydrogen atom and its connection to the oxygen atom could be very important for determining unique properties for structural models. The first optimization calculation of Vit-B3 was done in the gas phase and the second and third optimization calculations were done in 1-octanol and water solvent environments resembling the oil-water system. The geometries of Vit-B3 in different solvent environments were not significantly changed and each of C=O, C–O, and O–H bond distances were found ~ 1.22, 1.35, and 0.98 Å, respectively. In addition, two C–N bond distances were found ~ 1.34 Å and C–C and C–H bond distances were found ~ 1.39 and 1.09 Å. Consequently, the geometries showed small changes in both up and down conformations and among the embedded gas phase, 1-octanol, and water environments. However, comparing the values of evaluated total energy (E) in Table 1 indicated a higher stability for up-conformation than down-conformation in all three environments.
In addition to the obtained impacts of conformations on stability, the highest stability of each conformation was found in water medium and the lowest one was found in the gas phase showing the impacts of embedded medium for obtaining a desired structure. In this regard, the Vit-B3 structure was supposed to be better stabilized in water medium rather that the oil medium. Further analyses of the results of Table 1 for the frontier molecular orbitals features indicated the changes of HOMO and LUMO levels and their related features between up and down conformations and among the embedded environments. As the results showed, the energy levels of HOMO and LUMO (EH and EL) moved to a farther distance to each other from the up-conformation to the down-conformation in all three environments. Accordingly, the values of EG, η, µ, and ω detected such effects of conformational changes and embedded environments. Examining the ESP surfaces could also show effects of conformational changes on the electronic features, as the semi-blue head of carboxylic acid region in the up-conformation was converted to the semi-red head into the down-conformation. The red-green-blue colors of ESP mean electrostatic-based negative-neutral-positive regions, and yellow is further a semi-negative region. This is indeed important to mention that exploring such detailed features of structures are the highlighted advantages of performing computer-based investigations to provide insights into further developments of materials applications [91].
As explained above, the molecular models of this work were investigated in oil-water solvent environments to explore their features for providing insights into a form of vitamin B3; nicotinic acid (NA). The results indicated slight changes of geometries, but more significant changes of structural stabilities and electronic features in different embedded environments. To identify these features of medium-dependency of NA, the models were analyzed based on their thermochemistry ΔG values and LogP. As mentioned earlier, LogP could represent the tendency of a molecule for existing in oil with values larger than 1 and existing in water with values smaller than 1. Accordingly, the models were recognized using Equations 5 and 6 to evaluate the values of ΔGSolvent and LogP; the results were listed in Table 2. As could be found by the obtained values, the water medium was more suitable than the oil medium for both up and down conformations. From the obtained values of E of Table 1, the up-conformation was more stable than the down-conformation, accordingly smaller changes of ΔGSolvent were found for up-conformation compared with down-conformation. In other words, it could be mentioned that lower changes of solvent effects were found for up-conformation compared with higher changes of solvent effects for down-conformation. In accordance with the values of ΔG1-Octanol compared with ΔGWater, a value of < 1 was found for LogP of both up and down conformations. Furthermore, the up-conformation showed a smaller value (LogP = 0.64) meaning a better suitability of existing in the water medium even than the down-conformation (LogP = 0.96). However, it should be noted that positive values of LogP are still representing an oily tendency of Vit-B3, but not so much as the watery tendency. As a result, the conformational changes of Vit-B3 showed considerable impacts on both electronic and thermochemistry features, and also the stabilities and LogP of the conformations showed such structural effects. In addition, the solvent environments showed their significant effects on each conformational structure of Vit-B3 with a higher significance for down-conformation than up-conformation. However, it was obvious that the solvents had their significance on defining new structural features for a molecule. It should be reminded here that molecular models are mainly dependent on their characteristic features. In this case, knowing their features could help to improve them not only for the innovating new chemical compounds, but also for developing further applications. Especially in the case of biological related issues, the informative models are very important for employing them in various purposes. On the other hand, any need of their further modifications could be also found helpful to bring new structures and compounds for an already specified target. In the case of current study of Vit-B3, it was found that only the slight changes have their significance on the properties of compounds in both structural and electronic features. As it was shown that models were only different with their conformations, they were also found different with their specifications. As a consequence, both structural and electronic descriptors of molecular models are needed for determining their next properties and features. The solvents effects have been as very important issues to be learned, as they were easy to be achieved by performing DFT calculations and evaluating the related specifications. Indeed, customizing the chemical structures for various purposes and applications are essential in terms of developing novel materials especially for the cases of biological related and life science applications and they were achievable for running further assessments and determinations. The representative Vit-B3 of this work showed the advantage of exploring all internal and surrounding specifications of a targeted bio-active chemical system.
Conclusion
The main goal of this work was focused on the DFT investigation of Vit-B3 in oil and water solvent environments to provide insights into the features of a form of vitamin B3. In accordance with the obtained results, highlighted remarks were concluded. Based on the obtained values of total energy, the formation of up-conformation of Vit-B3 was more suitable than that of down-conformation. In both cases of up and down conformations, the highest stability was found for the models in water medium and stabilities in oil medium and gas phase were in the next orders. Accordingly, the obtained values of LogP for both conformations were found below than one indicating the Vit-B3existence in a watery medium rather that an oily medium. Moreover, the LogP value of up-conformation showed waterier tendency than that of down-conformation. In this regard, the models were compared based on their structural and electronic features to show characteristics of each conformation of Vit-B3 for developing further applications.
Disclosure Statement
No potential conflict of interest was reported by the authors.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Authors' Contributions
All authors contributed to data analysis, drafting, and revising of the paper and agreed to be responsible for all the aspects of this work.
Orcid
Mohamed J. Saadh
https://www.orcid.org/0000-0002-5701-4900
Faris Anad Muhammad
https://www.orcid.org/0009-0003-3886-5536
Romina Mahdavi
https://www.orcid.org/0009-0006-1955-702X
Yasaman Nazariyan Parizi
https://www.orcid.org/0009-0006-2919-6993
Ebrahim Balali
https://www.orcid.org/0000-0002-2653-9149
Juan Carlos Cotrina-Aliaga
https://www.orcid.org/0000-0003-0293-0394
Mohaned Adil
https://www.orcid.org/00009-0004-9980-307X
Salam Ahjel
https://www.orcid.org/0009-0007-5880-2822
Meryelem Tania Churampi Arellano
https://www.orcid.org/0000-0002-7973-1863
Yenina Gallardo-Lolandes
https://www.orcid.org/0009-0008-1024-8639
Renato R. Maaliw III
https://www.orcid.org/0000-0002-7310-2708
Mahmoud Mirzaei
https://www.orcid.org/0000-0001-9346-4901
Kun Harismah
https://www.orcid.org/0000-0002-8231-8164
HOW TO CITE THIS ARTICLE
Mohamed J. Saadh, Faris Anad Muhammad, Romina Mahdavi, Yasaman Nazariyan Parizi, Ebrahim Balali, Juan Carlos Cotrina-Aliaga, Mohaned Adil, Salam Ahjel, Meryelem Tania Churampi Arellano, Yanina Gallardo-Lolandes, Renato R. Maaliw III, Mahmoud Mirzaei, Kun Harismah. Exploring Impacts of Oil and Water Environments on Structural and Electronic Features of Vitamin B3 along with DFT Calculations. J. Med. Chem. Sci., 2023, 6(10) 2419-2431
DOI: https://doi.org/10.26655/JMCHEMSCI.2023.10.17
URL: https://www.jmchemsci.com/article_171907.html


