Document Type : Original Article
Authors
1 Center for Toxicology and Health Risk Studies, Faculty of Health Sciences, Universiti Kebangsaan Malaysia, Jalan Raja Muda Abdul Aziz, 50300, Kuala Lumpur, Malaysia
2 Environmental Health and Industrial Safety Programme, Faculty of Health Sciences, Jalan Raja Muda Abdul Aziz, 50300, Universiti Kebangsaan Malaysia, Kuala Lumpur, Malaysia
Abstract
Introduction: A chemotherapeutic agent known as the etoposide is used to treat acute lymphoblastic leukaemia (ALL). Over the years, the etoposide usage in treating ALL has yielded positive outcomes. Nevertheless, it has been discovered that a number of ALL patients experience side-effects and are susceptible to cancer cells. Accordingly, drug research for improved chemotherapy becomes necessary. The results of numerous investigations using compounds containing organotin (IV) dithiocarbamate were encouraging.
Aim: The objective of this study is to examine the genotoxicity effects on Jurkat E6.1 cells, derivatives of the aforementioned substances were chosen for this investigation.
Materials and Methods: The assessment of cell cycle arrest and genotoxic effects on Jurkat E6.1 cell lines was accomplished using the substances triphenyltin (IV) diisopropyldithiocarbamate (C1) and triphenyltin (IV) dialkyldithiocarbamate (C2). The cell cycle arrest was established using cell cycle analysis. The average DNA tail moment score was used to calculate the genotoxic effects of DNA damage. Inhibitory concentration (IC50) was used to conduct both analyses. C1 has an IC50 of 0.1 M, while C2 has an IC50 of 0.2 M. On the other hand, the etoposide, which served as a positive control, has an IC50 of 0.87 M.
Results: The research findings demonstrated that after four hours of treatment, both triphenyltin (IV) dithiocarbamate and etoposide compounds prompted cell cycle arrest, with a significant difference (p < 0.05) in the S phase. Following a four-hour etoposide treatment, the findings of genotoxicity assessment revealed substantial DNA damage (p < 0.05). Furthermore, the C1 and C2 treatments revealed a similar level of DNA damage. However, when a statistical analysis of the comet assay results was accomplished, no discernible change (p > 0.05) was observed.
Conclusion: In summary, DNA damage and cell cycle trapping were caused by etoposide, C1, and C2 in Jurkat E6.1 cells.
Graphical Abstract
Keywords
Introduction
The Global Cancer Observatory (2021) [1] has recorded 474,519 cases of cancer worldwide and 311,594 deaths from cancer. In Asia, the leukaemia incidence for both sexes is 230,650, and is the highest compared to other continents. In Malaysia, leukaemia ranks as the ninth most common type of cancer, with 1,905 cases. For ALL, the American Cancer Society (2021) [2] stated an incidence rate of 5,690 while the number of deaths was 1,580 cases. This type of cancer is more commonly reported in children than in adults. In Malaysia, the Malaysia National Cancer Registry 2012-2016 reported that the incidence rate of ALL is the highest among children aged 0-4 years old compared to the other age categories.
There is a variety of treatments for ALL. Tyrosine kinase inhibitors (TKIs) combined with cytotoxic chemotherapy appear to be the primary treatment for Philadelphia chromosome-positive (Ph-positive) ALL. According to Daver et al. [3], the use of TKI at the beginning of the cancer stage, as well as continuous treatment will give the best results. Imatinib is the first TKI drug used to treat ALL Ph-positive patients. However, treatment for ALL differs for various age categories. Moreover, another treatment for ALL involves four phases that span 2-3 years of implementation. The four phases are induction, consolidation, intensification, and long-term care. In addition, to prevent relapse of the central nervous system, treatment should continue indefinitely.
Therefore, further studies related to the treatment for ALL need to be conducted to overcome the issue of resistance that occurs in the current treatments. According to Kamaludin et al. [4], current treatments can lead to treatment failure and cancer progression.
Therefore, there is a need for new drugs that can act as effective chemotherapeutic agents and do not give side-effects on patients. According to Amir et al. [5] and Liu et al. [6], in vitro studies to find treatments that use synthetic metals are the best in discovering new anti-cancer agents.
Organotin (IV) dithiocarbamate is one of the most in-depth studied organotin (IV) metals [7]. Organotin (IV) dithiocarbamate compounds possess chemotherapeutic effects and exhibit good structural diversity, according to a number of prior investigations [8]. These compounds have cytotoxic effects on different types of cancer while also serving biological applications, such as anti-microbial, larvicidal agents, and anti-malaria uses [9]. Hence, the discovery of new alternatives for ALL has prompted the use of triphenyltin (IV) diisopropyldithiocarbamate (C1) and triphenyltin (IV) dialkyldithiocarbamate (C2) compounds. To achieve this aim, in vitro analysis was performed to assess the genotoxicity of these two compounds on Jurkat E6.1 cell lines through cell cycle analysis and comet assay.
Materials and methods
Preparation of stock compounds
The triphenyltin (IV) dithiocarbamate compounds were synthesised in the Faculty of Health Sciences, UKM Kuala Lumpur Campus. Figure 1 shows that triphenyltin (IV) diisopropyldithiocarbamate (C1) compounds were used, whereas Figure 2 illustrates the use of triphenyltin (IV) dialkyldithiocarbamate (C2) and the commercially available drug etoposide (Etpo) as a positive control. The preparation of stock compounds was conducted by separately dissolving 0.0053 g (C1), 0.0052 g (C2), and 0.0040 g (Etpo) into 1 mL of dimethyl sulfoxide (DMSO). The stock compounds were stored at -20 °C prior to the treatment. Figures 1 and 2 depict the chemical structures of triphenyltin (IV) dithiocarbamate compounds.
Cell lines and cell culture
The cell line used in this study was the Jurkat E6.1 cell line. Cell culture was carried out by removing the cell stock stored in a liquid nitrogen freezer at -80 °C and thawing them in a water bath without submerging the cryovial cap. The cryovials were then disinfected using 70% alcohol. The next steps were carried out aseptically in a biosafety cabinet. The Roswell Park Memorial Institute (RPMI) medium, which was supplemented with 1% penicillin-streptomycin and 10% of foetal bovine serum, was used to culture the cells and subsequently incubated under 5% CO2 at 37 °C. Subculturing of the cells was conducted every two days.
Cells seeding and treatment
The half-maximal inhibitory concentration (IC50) for C1 was 0.1 µM, 0.2 µM (C2), and 0.87 µM (Etpo). The Jurkat E6.1 cells were treated with 1000 µL of the compounds at their corresponding IC50. As for the negative control (VC), 1000 µL of 1×106 mL-1 cells were used. The treatments were conducted in 6-well plates.


Analysis of cell cycle arrest
Assessment of the disruption in the cell cycle caused by the compounds on Jurkat E6.1 cells was done via the cell cycle analysis. The treatments were conducted for two hours and four hours prior to harvest and fix cells. Then, the cells were stained with PI/RNase in the dark and analysed using BDFACSCanto II flow cytometry.
Alkaline comet assay
The alkaline comet assay was carried out to determine the genotoxicity of organotin (IV) dithiocarbamate compounds on Jurkat E6.1 cells. The procedure started with the preparation of several solutions to be used in this analysis, which were the lysis solution, electrophoresis solution, neutralisation buffer solution, and also ethidium bromide solution. The treatments require preparation of low melting agarose (LMA) and normal melting agarose (NMA) beforehand. The treatments were conducted for four hours before cell harvesting. The samples were stored in dark and damp conditions at 4 °C prior to a fluorescent microscope analysis. The formation of a comet-like pattern indicates DNA damage. The tail moment of comet was analysed using the Comet Score Software by Tritek Corp.
Statistical analysis
IBM SPSS (version 26.0) was used for the statistical analysis of data collection. The significance (p < 0.05) of the cell cycle arrest and DNA damage caused by the triphenyltin (IV) dithiocarbamate compounds was assessed using one-way ANOVA analysis.
Results and Discussion
For determination of the cell cycle arrest, both the C1 and C2 compounds and etoposide showed significant differences (p < 0.05) in the cell population percentage at the S phase compared to VC after four hours of treatment. The Kruskal-Wallis non-parametric test was used to determine the significance due to non-normal data distribution. For the percentage of cell population in both C1 and C2 treatments, a significant cell decrease (p < 0.05) could also be seen in the G0/G1 phase compared to the VC after four hours of treatment. Each phase of cell cycle represents an average cell population percentage for three replicates of treatments ± S.E.M. Figures 3, 4, and 5, as well as Tables 1, 2, and 3 demonstrate the cell population (%) in each phase of the cell cycle after treatments by etoposide, C1 and C2.




The results show that the percentage of cell population in the S phase was significantly higher (p < 0.05) after four hours of treatment with etoposide, C1, and C2 when compared to the VC, with 52.22 ± 1.83, 58.94 ± 4.42, and 57.26 ± 1.8, respectively. The cell population percentages for the VC were 47.2 ± 3.0 for the G0/G1 phase, 44.0 ± 3.82 for the S phase, and 8.83 ± 0.83 for the G2/M phase.
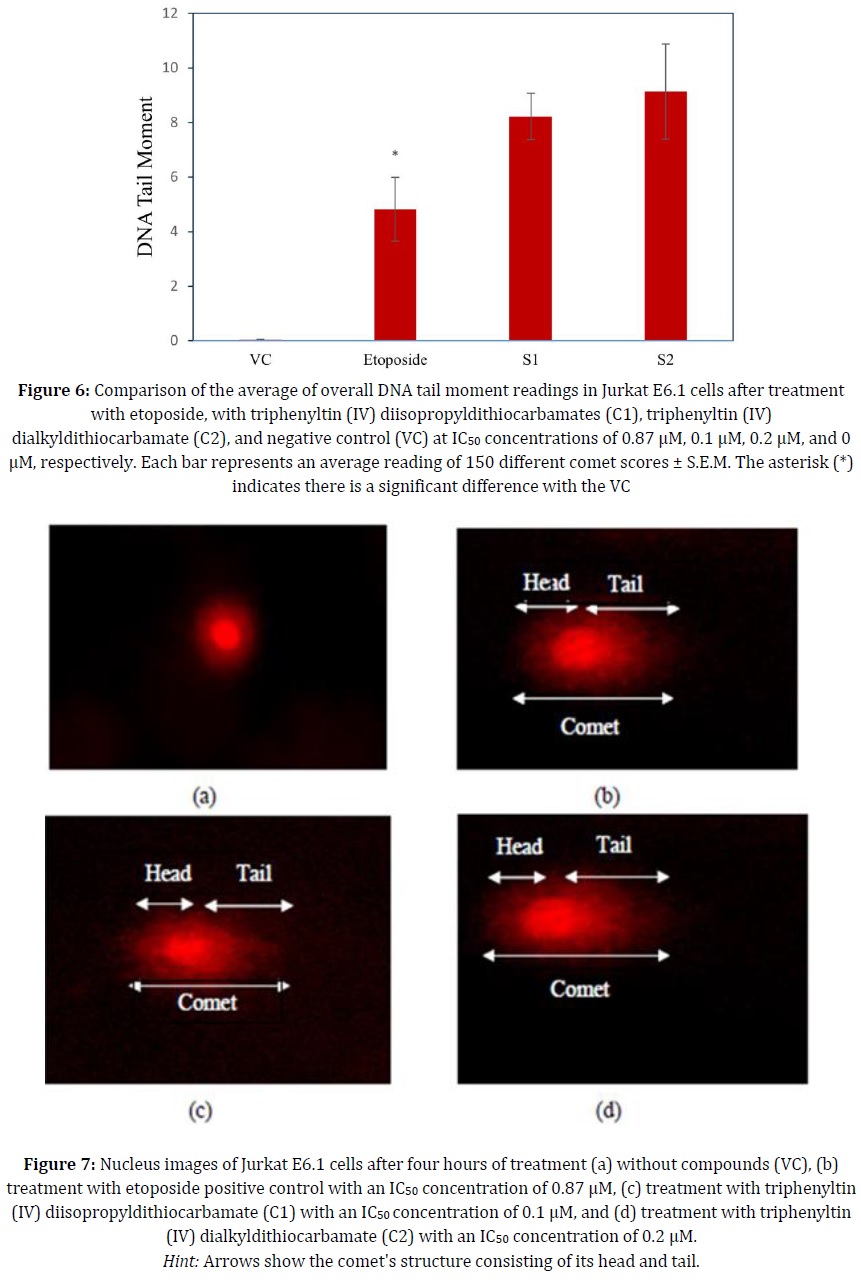
The DNA tail moment readings indicated a significant difference (p < 0.05) between the etoposide-induced DNA damage and that of the VC, according to the statistical analysis. However, statistical analysis did not show a significant difference (p > 0.05) in DNA damage between C1 and C2 treatments subsequent to four hours of treatment when compared to the VC. This analysis was carried out by taking an average of three replicates for three different experiments of etoposide treatment, C1, C2, and VC against Jurkat E6.1 cells. Comparisons were carried out by comparing the values of significant differences in C1, C2, and etoposide treatments against the VC. The mean DNA tail moment score for etoposide treatment was 4.82 ± 1.16. This indicates that there was significant (p < 0.05) DNA damage induced by etoposide. All three compounds underwent statistical analysis and were compared with the mean reading of the VC DNA tail moment score of 0.03 ± 0.017. Figure 6 demonstrates the average of overall DNA tail moment readings of Jurkat E6.1 cells after 4-hour treatments with etoposide, C1 and C2.
C1 and C2 treatments showed a significant difference (p < 0.05) in the DNA tail moment scores between them. Similarly, the mean DNA tail moment scores of both C1 and C2 compounds showed significant differences (p < 0.05) when compared to the etoposide, positive control. Figure 7 illustrates the DNA comet images after four hours of treatment with all three compounds including VC at their respective IC50 against Jurkat E6.1 cell lines.
Etoposide is an existing chemotherapeutic drug used to treat acute lymphoblastic leukaemia (ALL). Findings from Cancer Research UK (2022) [10] indicated that there are side-effects of etoposide use in cancer treatment, such as vomiting, decreased blood pressure, seizures, and inducing other leukaemias. Therefore, studies to obtain better chemotherapeutic compounds are needed.
The organotin (IV) compounds gained prominence subsequent to the discovery of their anti-tumour properties [11]. According to Awang et al. [12], these novel compounds showed high selectivity against a wide range of cancer continuous cells, regardless of ligand diversity. Triphenyltin (IV) compounds, which comprise three phenyl groups bound to tin (IV) atoms, exhibit greater cytotoxic effects when investigated on cancer continuous cells than diphenyltin (IV) compounds in most research, according to Syed Annuar et al. [13].
In the present study, there were significant differences (p < 0.05) in the Jurkat E6.1 cell population percentage in the S phase after four hours of treatment with etoposide, C1, and C2 when compared to the VC. This indicates the occurrence of cell cycle arrest during the treatment. Under normal circumstances, the S phase in the cell cycle is a phase of DNA replication and synthesis. If the treated cell population does not successfully pass the S phase checkpoint, cell death via apoptosis will occur. Consequently, arresting cell population in the S phase will result in a decline in cells proportion in the G2/M phase. This suggests that both C1 and C2 compounds have the potential to arrest the Jurkat E6.1 cell cycle in the S phase.
According to Wylie [14], there is an association between cell cycle arrest and apoptosis. The genes associated with the progression of cell cycle help to regulate apoptosis, where cell cycle arrest and the apoptosis induction will ensue due to the altered cellular content caused by any changes in the normal cell cycle [15]. The involvement of the p53 protein is important in the arrest of cell cycle and influences apoptosis during the occurrence of abnormal changes in the cell cycle and cellular content. However, the p53 protein does not function properly in most cancer incidents. This is because the p53 gene has mutated due to a sequence change in the gene. In fact, p53 mutants can cause the development of cancer cells.
A study by Yunlan et al. [16] showed that there is a cell cycle arrest that occurs when an organotin (IV) compound treatment was given. Moreover, the apoptosis occurrence was observed in the same study. Their findings are in alignment with that of the present study, in which cell cycle arrest, as well as apoptosis also occurred when Jurkat E6.1 cells were treated using triphenyltin (IV) compounds. Attanzio et al. [17] reported that the induction of cell death via apoptosis by the organotin compounds occur by binding to external phosphate groups of the DNA and disarraying the intracellular phospholipid metabolism. Subsequently, this alters the cellular content and leads to cell cycle arrest.
A study by Balas et al. [18] similarly showed cell cycle inhibition occurring in the S phase when leiomyosarcoma cells were treated with triphenyltin (IV) compounds. In addition, triphenyltin (IV) compounds have a greater toxic effect than diphenyltin (IV) compounds on human colorectal cancer, according to a study by Haezam et al. [19]. Adeyemi et al. [20] stated that the functional groups linked to tin atoms had in vitro toxic effects on the examined cells. Pallerito et al. [21] claimed that the toxicity of organotin (IV) compounds depends on their chemical structure. Organotin (IV) compounds cause DNA damage by intercalating bonding or groove binding with the DNA based on the coordination of the number of bonds and the type of functional group linked to the tin atom, according to Rehman et al. [22] and Yusof et al. [23].
Our cell cycle study results showed the occurrence of cell cycle arrest caused by the C1 and C2 compounds, which suggests that DNA damage was also occurring. However, comparisons of DNA tail moment scores of the comet assays for Jurkat E6.1 cells treated with C1 and C2 showed no significant differences (p > 0.05) when compared to the VC. This is in contrast to the etoposide positive control that showed the presence of significant DNA damage (p < 0.05) when compared with the VC. Etoposide-induced DNA damage occurs when etoposide reacts with topoisomerase II. This leads to the damage of the DNA strands [24]. Nevertheless, a comparison of DNA damage between C1 and C2 treatments showed that there was a significant difference (p <0.05) between them.
Conclusion
According to the results of the cell cycle analysis, both triphenyltin (IV) diisopropyldithiocarbamate (C1) and triphenyltin (IV) dialkyldithiocarbamate (C2), which are both organotin (IV) dithiocarbamate derivative compounds, can cause significant (p < 0.05) cell cycle arrest in Jurkat E6.1 cells in the S phase after four hours of treatment. Alkaline comet assays showed that both compounds exhibited genotoxicity effects on Jurkat E6.1 cell lines after four hours of treatment that resulted in DNA damage, as evidenced by the formation of comet-like structures. Moreover, a significant difference was observed in the DNA tail moment scores of cells treated with C1 and C2. Therefore, both compounds are proposed to undergo induction mechanism analysis such as the caspase analysis and reactive oxygen species assay so that their toxic effects can be more clearly understood. Consequently, this can assist in developing new and effective chemotherapeutic drugs for the ALL treatment
Acknowledgements
The authors would like to thank the Ministry of Higher Education (Malaysia) for funding this study through the FRGS/1/2018/STG01/UKM/02/20 grant. They would also like to thank the Faculty of Health Sciences, Universiti Kebangsaan Malaysia (UKM), and the Environmental Health and Industrial Safety Programme for providing the facilities to conduct this study. Special gratitude is given to the management and lab assistants of the Biocompatibility and Toxicology Laboratory of the Centre of Toxicology and Health Risk Studies of Universiti Kebangsaan Malaysia for the assistance provided throughout this research.
Funding
This research receives a grant from the Ministry of Higher Education (Malaysia) for funding this research.
Authors' Contributions
All authors contributed to data analysis, drafting, and revising of the article and agreed to be responsible for all aspects of this work.
Orcid
Normah Awang
https://orcid.org/0000-0002-3772-2088
Nurul Farahana Kamaludin
https://orcid.org/0000-0002-2202-5917
Sharifah Nadhira Syed Annuar
https://orcid.org/0000-0001-5872-988X
HOW TO CITE THIS ARTICLE
Normah Awang, Noor Hanim Mohd Nor, Nurul Farahana Kamaludin, Sharifah Nadhira Syed Annuar. Triphenyltin (IV) Dithiocarbamate Compounds Induce Cytotoxicity in Acute Lymphoblastic Leukemia (Jurkat E6.1) Primarily via DNA Fragmentation and Cell Cycle Arrest. J. Med. Chem. Sci., 2023, 6(10) 2397-2406


