Document Type : Original Article
Authors
1 Department of Soil &Water, College of Agriculture, Salahaddin University-Erbil, Kurdistan Region, Iraq
2 Department of Chemistry, College of Science, Alnahrain University –Baghdad, Iraq
Abstract
Heavy metals are one of environmental concerns in industrial wastewater effluents, and adsorption is one of the widely used processes in removing heavy metals from aqueous solutions. Concerning the importance of aforementioned topic, the main goal of the current study was to evaluate the efficiency of modified poly (styrene-co-maleic anhydride) [MP-SCMA]-based resins in the removal of chromium metal ions from aqueous solutions. To perform the present study, adsorption experiments were performed by mixing 10 mg of MP-SCMA resin with 10 ml of chromium solution in 100 ml Erlenmeyer flasks. In this study, parameters of pH (2-7), temperature (5-45 ᵒC), contact time (30-180 min), adsorbent dose (5-20 g/l) and initial chromium concentration (5-40 mg/L) was evaluated on the amount of chromium removal. Atomic absorption device made in Germany (model: Analytik Jena: NovAA 400 P) was used to determine the amount of chromium metal ions in wavelength of 357.9 nm. The findings of the present study showed that except for the variable "initial chromium concentration", other factors have a direct relationship with the removal percentage of chromium ions by MP-SCMA-based resins. In addition, the adsorption process of chromium metal ion by MP-SCMA was more consistent with the Langmuir isotherm model (R2=0.992) than the Freundlich isotherm model (R2=0.966). Based on the obtained results, it can be concluded that resin-based MP-SCMA adsorbent can be used as a high-efficiency adsorbent to remove metals from industrial wastewater.
Graphical Abstract
Keywords
Main Subjects
Introduction
Today, heavy metals are one of major concerns for environment and human health. Therefore, it is very important to investigate effective ways to remove heavy metal ions from industrial wastewater before discharging it into the environment or reusing it for different purposes [1]. The presence of heavy metals in concentrations exceeding the permitted standards of drinking water or food causes various complications, including poisoning, severe sensitivity, chromosomal lesions, mental retardation, Parkinson's disease, kidney stones, osteoporosis, skin itching, digestive discomfort, stomach ulcers, respiratory problems, weakening of the immune system, headaches and dizziness, and various types of cancer [2-7].
Several methods have been used to remove metal ions, such as precipitation, ion exchange, reduction, electrochemical purification, reverse osmosis, solution extraction, membrane filtration, etc. [8]. Most of these methods have a high cost and also cause the production of secondary pollutants. Among various methods, adsorption using low-cost adsorbents is an environmental and effective method for removing heavy metals from aqueous solutions . One of the most effective methods for removing metal cations is their adsorption on organic materials containing chelating functional groups. These polymers with suitable functional groups can have high efficiency in removing metal ions [9].
Synthetic chelating ion exchange resins have attracted great attention due to their application in various fields such as heavy metal removal, heterogeneous catalysis, solid electrolysis, ion exchange membranes, ion selective electrodes, and industrial wastewater treatment. Therefore, a lot of attention has been paid to the synthesis of chelating resins and a lot of research has been done on their adsorption behaviours for heavy metal ions from different matrices [10].
Chelating ion exchange resins, also known as complexing resins, are designed to achieve high specificity to an ion or a group of ions. This type of ion exchange resins adsorb metal ions by combining ionic and coordination interactions instead of simple electrostatic interactions in conventional anion or cation exchange. As a result, chelating resins show more selectivity than the conventional resins [11].
Concerning the importance of the above discussed subject, the main goal of the current study was evaluating the efficiency of modified poly (styrene-co-maleic anhydride) [MP-SCMA]-based resins in the removal of chromium metal ions from aqueous solutions. In addition, the effective variables were investigated on chromium ion adsorption by this adsorbent.
Martials and Methods
Chemicals and equipment used
To prepare chromium standard solutions, high purity chromium nitrate salt [Cr (NO3)3.9H2O] was purchased from Merck, Germany. Poly (styrene-co-maleic anhydride) was obtained from Sigma-Aldrich. Deionized distilled water was used in all stages of the experiment to prepare standard solutions and target solutions. Atomic absorption device made in Germany (model: Analytik Jena: NovAA 400 P) was used to determine the amount of chromium metal ions. In addition, a deuterium field surface absorption corrector with a wavelength of 357.9 nm and a line width of 0.5 nm was used. Analytic Jena model hollow cathode lamps were used as a source of light emission with a certain wavelength (Resonance lines) which has the highest intensity. Likewise, flames containing air-acetylene gases were used to measure metal ions.
The method of preparation of solutions and absorbents
Before use, all glass containers were washed with sulfochromic solution, and then with deionized distilled water. To prepare the initial standard solution with a concentration of 500 mg/liter, 0.982 grams of chromium nitrate salt was dissolved in a 250 ml volumetric flask containing some deionized distilled water. Next, after adding a few drops of concentrated nitric acid, it was deionized with distilled water to the considered volume [12].
Experimental methodology for investigation of effective variables
Surface adsorption experiments were performed by mixing 10 mg of MP-SCMA resin with 10 mL of chromium solution in 100 mL Erlenmeyer flasks. The desired samples of chromium were prepared by diluting the standard solution.
To investigate the pH effect on the test results, solutions of chromium with a constant concentration of 10 mg/L were prepared with different pH from 2 to 7. To investigate the effect of contact time, solutions of chromium with a constant concentration of 10 mg/L were prepared in the presence of 10 mg of MP-SCMA resin at pH=5 and then, it was mixed at room temperature for different periods of time from 10 to 180 minutes with a stirring speed of 150 rpm. To investigate the effect of the initial concentration of chromium metal ion on surface absorption, solutions with concentrations of 1, 5, 10, 15, 20, 25, 30, 35, and 40 mg/L of chrome were prepared and with a fixed amount of 10 mg of adsorbent in pH=5 and mixed at room temperature. After that, the action of surface adsorption was studied for 90 minutes with a stirring speed of 150 rpm. To evaluate the effect of the amount of adsorbent on the surface absorption of metal ion, first, different amounts of MP-SCMA resin (5, 10, 15, and 20 g/L) were prepared. Next, chromium solution with a constant concentration of 10 mg/L was added to each of the above-mentioned adsorbent amounts with pH=5. Finally, the solution consisting of adsorbate and metal ion was mixed at room temperature, for a contact time of 90 minutes and at a speed of 150 rpm. To investigate the effect of temperature on the surface adsorption of chromium ions, solutions with a constant concentration of 10 mg/L were prepared at different temperatures (10-45 °C) in the presence of 10 mg of MP-SCMA resin at pH=5. Then, it was mixed for 90 minutes with a stirring speed of 150 rpm and the results of surface absorption were analysed [12].
To investigate equilibrium studies, 10 mg of MP-SCMA resin was mixed with a solution containing different concentrations of metal ions (5-50 mg/L) for 90 minutes at pH=5. After filtering, the concentration of metal ions in the filtered solution was measured using atomic absorption spectroscopy.
Equation 1 was used to calculate the chromium ion removal percentage where, Co and Ce are, respectively, the initial concentration and the equilibrium concentration of chromium ion in mg/L [13].

The adsorption equilibrium is usually described by isothermal equations, whose parameters express the properties of the surface and the degree of adsorbent affinity. To determine the efficiency of an adsorbent, it is important that there is a precise equilibrium relationship between the concentrations of metal ions in the solid and liquid phases [14]. In the present study, the data obtained from the experiments were analysed with Langmuir and Freundlich models.
Langmuir isotherm was evaluated based on Equations 2 and 3. In this model, it is assumed that a surface contains a certain number of equivalent sites where absorbable species can be chemically or physically attached to that site. Physical adsorption includes Van der Waals interactions and chemical adsorption includes the formation of covalent bonds between the adsorbent and adsorbate [15].

Where, qe is the amount of metal adsorbed on the surface per unit weight of the adsorbent, which was calculated by Equation 4.

In Equation 4, Co is the initial concentration of the metal in the solution and Ce is the final concentration of the metal in the solution after equilibrium in terms of mg/L, the volume of the solution (V) is in litres and m is the mass of the adsorbent in grams. In the Langmuir equation, KL and qm are Langmuir constants that depend on the energy and capacities of adsorption [16]. To obtain the Langmuir constants, the graph of Ce vs. Ce/qe was used by Equation 3 and using the obtained data related to testing the effect of initial concentration of chromium.
Freundlich isotherm was evaluated based on Equation 5. The equation related to the Freundlich isotherm is an exponential equation. In other words, when the adsorbate concentration in the solution increases, its concentration in the solid phase (adsorbent surface) also increases. According to Equation 5, "KF" and "n" are the experimental constants of the Freundlich equation, which indicate the relative adsorption capacity of the adsorbent and intensity of adsorption, respectively. The magnitude of the "n" power is an indication of a favourable absorption. In general, if "n" values are in the range of 2 to 10, it indicates good adsorption, and if this value is in the range of 1 to 2, it means relatively difficult adsorption, and values less than 1 indicate weak adsorption [17, 18].
![]()
Equation 6 was used to determine the KF and n constants in the Freundlich equation. A straight line was obtained by plotting the logqe vs. logCe curve. Finally, the constants of the Freundlich equation can be calculated from the slope and width from the origin [19].
![]()
Results and Discussion
The results of the study revealed that with the increase in pH, the adsorption amount of chromium ions by the adsorbent increased and reached a constant state at a pH of approximately 5 (Figure 1). Therefore, to prevent the formation of chromium hydroxides and possible disturbances, pH=5 was chosen as the optimal pH. At low pH, the polymer is in the fully protonated state. Therefore, nitrogen atoms have a positive charge and cannot give electron density to metal cations. In addition, carboxylic acid groups are also protonated and will have less power in giving electron density. In other words, at low pH values, high concentration of H+ can react with carboxylate ions (COO-) and amine groups (NH+) to form the protonated form. The carboxyl groups of the prepared resins have a weak acid character. In addition, its protonation degree in acidic pH affects the ability of resin to bond with metal cations. The reason for the adsorption increase at alkaline pH can be attributed to the decrease in the concentration of hydrogen ions in that range. This reduces the competition of these ions with metal cations to bind to the active sites of the adsorbent, and therefore the possibility of more adsorption of these cations by the resin is provide [11, 12].

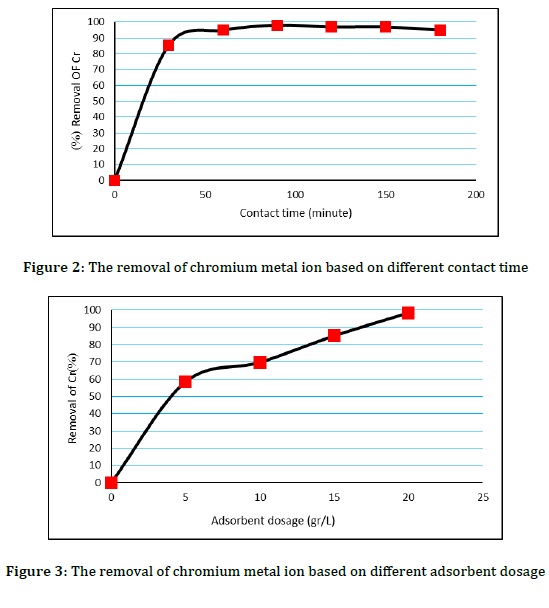
One of the other important parameters investigated in this study was contact time. The contact time plays an important role in the adsorption of metal ions on the solid surface. It is clear that the removal percentage until equilibrium is reached depends on the contact time. While, after reaching the equilibrium state, it is independent of the contact time, because the rate of surface adsorption and desorption is the same after reaching the equilibrium condition [20]. The results showed that the effect of contact time on the removal of chromium ions with MP-SCMA resin indicates fast adsorption in the first minutes, and after that, the rate of adsorption gradually decreases and reaches equilibrium in about 90 minutes (Figure 2). The reason for this could be that the amount of metal ion adsorption increases with increasing contact time. However, the polymer chains begin to aggregate over time and the number of available sites for adsorption decreases. Therefore, the penetration of metal ions to the adsorption sites becomes difficult and the adsorption process reaches equilibrium.
The variable effect of adsorbent amount on the removal percentage of chromium ions from aqueous solutions is highly important, because it determines the capacity of an adsorbent for initial concentration. To evaluate this variable, the adsorption capacity of chromium ions was studied as a function of the amount of adsorbent. The results of the present study showed that adsorption efficiency of chromium ions increases gradually with increasing amount of adsorbent (Figure 3). This may indicate the fact that increasing the amount of adsorbent provides a larger surface area or more adsorption sites for metal ions. Increasing the amount of adsorbent to remove chromium ions from 5 to 20 mg increases the percentage of removal from 58.5% to 98.5%, and then the amount of absorption is almost constant with the increase in the amount of adsorbent.
The results of the present study revealed that with increasing initial concentration of chromium, its removal percentage decreases (Figure 4). This decrease in removal efficiency at higher concentrations is due to the stability of the absorbent surface and consequently the stability of the active adsorption sites on the absorbent surface [20]. Higher concentrations of chromium ions saturate the active sites of MP-SCMA resin with chromium metal ions. The study of Moradi et al. (2014) about the removal of lead with Pumic adsorbent showed that as the concentration of lead increases, the efficiency of the adsorbent in removing that metal decreases [1]. In addition, the results of the present study, in terms of the influence of the initial metal concentration on the efficiency of the adsorbent, was consistent with the results of the studies by Sharma et al. [21], Karimi et al. [22] and Oliveira et al. [23].
Also, based on the results of the present study, it was found that with the increase in temperature, the adsorption rate of chromium metal ions increased on the surface of MP-SCMA (Figure 5). The reason for this could be that the adsorption method in the studied process is mainly chemical adsorption. In this type of adsorption, a chemical reaction takes place between the adsorbed component and the absorbent surface, and it is an irreversible process, because a lot of energy is needed to carry out the photoreaction. Unlike physical surface absorption, in this type of absorption, the rate of absorption or reaction increases with enhancing temperature. Therefore, it can be mentioned that with increasing temperature, the adsorption efficiency increases [24].
The results of the present study showed that the adsorption process of chromium metal ion by MP-SCMA is more consistent with the Langmuir isotherm model (R2=0.992) than the Freundlich isotherm model (R2=0.966) (Figures 6 and 7). The results of the present study were in agreement with the results of some studies and were not consistent with the results of others.


Rastgar et al. (2018) reported that in the process of removing Lead by Lignocellulose Nano-Fibers (LCNFS) and Chitin Nano-Fibres (CNFS) from aqueous solutions, the most consistent isotherm model was related to the Langmuir isotherm (R2=0.999) [25]. Kardam et al. (2014) used cellulose nanofibers to remove cadmium, nickel, and lead metals from aqueous solutions, and both Langmuir and Freundlich isotherm models were in high agreement with the data obtained from the mentioned adsorption process [26]. Nasiruddin Khan et al. (2015) used lignocellulose activated carbon to remove lead, and the Freundlich isotherm was more consistent with the obtained data [27]. Labidi et al. (2016) used chitin adsorbent to remove copper, and the resulting data were more consistent with the Langmuir isotherm [28]. Saman et al. (2017) showed that in the mercury removal process by lignocellulose waste, Langmuir and Temkin isotherms were more consistent with the obtained data [29]. Karthik and Meenaksi (2017) used chitin adsorbent for the removal of lead and copper, the data obtained from the study were more consistent with the Freundlich isotherm [30]. Different factors can be effective in the difference or similarity between the results of this study and the results of other studies, which include the type of adsorbate and adsorbent under investigation, contact time, type of aqueous solution, pH, adsorbate concentration, amount of adsorbent, temperature, and other similar factors [31–33].
Conclusion
Based on the results of the present study, it can be concluded that except for the variable "initial chromium concentration", the other factors had a direct relationship with the removal rate of chromium ions by modified poly (styrene-co-maleic anhydride) [MP-SCMA]-based resins. In addition, the adsorption process of chromium metal ion by MP-SCMA was more consistent with the Langmuir isotherm model (R2=0.992) than the Freundlich isotherm model (R2=0.966). Finally, it can be mentioned that resin-based MP-SCMA adsorbent can be used as a high-efficiency adsorbent to remove metals from industrial wastewater.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Authors' contributions
All authors contributed to data analysis, drafting, and revising of the paper and agreed to be responsible for all the aspects of this work.
Conflict of Interest
There are no conflicts of interest in this study.
Orcid
Lubna Salah Muhammed
https://orcid.org/0000-0003-2132-9131
Hamsa Thamer Sadeq
https://orcid.org/0000-0003-3088-6976
HOW TO CITE THIS ARTICLE
Mysara Rumman, Khaled Sabarna, Jessica Badawi, Israa Bahar, Mohammad Darawai. Using Modified Resins in Chromium Ion Removal from Aqueous Solutions. J. Med. Chem. Sci., 2023, 6(8) 1862-1871


