Document Type : Original Article
Authors
1 Technical Institute of Babylon, Al-Furat Al-Awsat Technical University, Babylon, Iraq
2 Chemistry Dep., College of Science, University of Babylon, Babil, Iraq
Abstract
Three of new azo-Schiff base ligands were prepared in this work via the preparation of Schiff base ((E)-2-(((2,5-dichlorophenyl)imino)methyl)phenol DCSS) produced from the condensation reaction between salicyaldehyde and (2,5-dichloroaniline), this Schiff base was regarded as a coupling component in azo reactions to form the new azo-Schiff ligands [(2-((E)-((2,5-dichlorophenyl)imino)methyl)-4-((E)-(2,4-difluorophenyl)diazenyl)phenol) L1, (4-((E)-(4-chloro-2-fluorophenyl)diazenyl)-2-((E)-((2,5-dichlorophenyl)imino)methyl)phenol) L2, and (4-((E)-(4-bromo-2-fluorophenyl)diazenyl)-2-((E)-((2,5-dichlorophenyl) imino)methyl) phenol) L3], respectively, via the reaction of the diazonium salts of the (2,5-difluoroaniline, 4-chloro-2-fluoroaniline and 4-bromo-2-fluoroaniline) with the previous Schiff base (DCSS). These new Azo-Schiff ligands were characterized with many spectroscopic techniques such as (FT-IR, 1H-NMR, Mass spectra, UV-Visible, and elementary analysis) to ensure their preparation. Five divalent transition ions (Co, Ni, Cu, Pd, and Pt) complexes were prepared for each ligand and characterized (some of them) with the previous techniques, the mole ratio of the cobalt and copper complexes was (1:2), while the rest (Ni, Pd, and Pt) complexes have a (1:1) ratios. In addition, complementary techniques, such as atomic absorption, molar electrical conductivity, and magnetic susceptibility were performed to deduce their geometries and form the collected data. The square planer geometry was examined. The biological study includes the antibacterial screening against two types of bacterial species gram-negative (E.coli) and gram-positive (S. aureus) using tetracycline as a reference antibiotic. Antioxidant assay of some of the prepared ligands and their complexes as well as an MC7 cell line breast cancer assay for two prepared complexes (PdL1 and PtL2) and as compared, they noticed that the platinum azo-Schiff complex has an IC50 = 56.78 a more active anticancer function than the palladium complex with IC50 = 74.64.
Graphical Abstract
Keywords
Introduction
Metal complexes and organic molecules have received increasing attention as they have been used as pharmacophores in the medical and pharmaceutical fields [1]. Basically, the presence of multiple functional groups in molecules improves their properties [2] when directed in essential fields such as medical [3] and industrial [4]. Schiff base (azomethine) group compounds [5] are one the compounds that have had major significance over the past decades until now [6, 7]. An assortment of ligand type Schiff-base and their metal complexes have been separated, these compounds have very malleable and various structures. Therefore, their properties have been studied [8]. Schiff bases were prepared for the first time in 1964 by the German scientist Hugo-Schiff [9] from the condensation of aldehydes, aliphatic, or aromatic ketones with aliphatic or aromatic primary amines. Schiff bases prepared from condensation of aromatic amines with aromatic aldehydes are more stable due to the occurrence of resonance phenomenon [10].
In homocyclic and heterocyclic forms, azo compounds (-N=N-) play a fundamental role in the dye and pigment industries [11, 12]. It extensively entered the analytical chemistry fields [13, 14], actually into the industrialization of drugs and other medications [15], and LCD color filters [16].
Azo-Schiff base compounds can be synthesized according to the compound’s characteristics and reaction conditions in two ways. The first way is by synthesizing the azo-moiety, and then Schiff base reaction [17]. The second way is the Schiff base is synthesized, and then coupled to form azo compounds [18]. These compounds and their complexes have been of specific and large importance to present multiple aspects of biological [19] and industrial applications such as textile dye and photo electronic [20].
Platinum compounds (cisplatin, carboplatin, or oxaliplatin) are among the most common compounds used as a drug against cancer. All three of these drugs are square planar platinum (II) complexes, bearing two inert ‘non-leaving group’ ligands, and two labile ‘leaving group ligands [21]. Newly, Pd-aryl azo complexes have found application in C–C coupling reactions [22] interacting highly with CT DNA [23] and exhibiting photochromic activity [24].
Finally, in this article, it is aimed to report the synthesis and reactivates of new azo-Schiff ligands and their metal ion complexes via the preparation of Schiff base ((E)-2-(((2,5-dichlorophenyl)imino)methyl)phenol DCSS) produced from the condensation reaction between salicylaldehyde and (2,5-dichloroaniline) as an initial step, this Schiff base was regard as a coupling component in azo reactions to form the new azo-Schiff ligands L1, L2, and L3, by coupling the diazonium salt of three aniline diahalogens with the coupling component (DCSS) as a second step and characterizing them by the above-mentioned spectroscopic methods to prepare a series of divalent metal complexes and characterize them by the previously mentioned spectroscopic methods (some of it) in addition to study the physical and chemical properties of the prepared compounds and studying the biological and toxicological tests for some of these compounds on human cells and the possibility of using them as an antioxidant, antimicrobial, and anticancer agents.
Materials and Methods
All chemicals and solvents utilized in this research are analytical grades. The amines were supplied from florochem chemical Company. For another chemical from (Merck and Sigma) companies, the stoichiometric analysis (CHN) of the ligands and their complexes was performed using ECS 4010 CHNOS Elemental analyzer. Metal contents were estimated on flame atomic absorption (Shimadzu AA 6800), and IR spectra were recorded using Bruker FT-IR Spectrophotometer and KBr pellets.
The UV-Visible spectra were recorded in the absolute ethanol as a solvent using Shimadzu 1650PC. HNMR data were collected using Varian 500MHz frequency and was used as a DMSO as a solvent. Mass spectra were recorded using Agilent technology (HP) and an ion source of 70ev. Info Lab Terminal recorded the conductivity values for the new complexes, and the magnetic susceptibility data were collected using the Sherwood instrument for magnetic measurements.
Schiff base (DCSS) synthesis [25]
In a 250 mL round bottom flask, 0.01 mol (1.22 g) of salicylaldehyde dissolved in 50 mL of absolute ethyl alcohol was refluxed with 0.01 mol (1.62 gm) 2,5-dichloroaniline dissolved in 50 mL of absolute ethanol with the addition of few drops of glacial acetic acid as a catalyst for the reaction, the reflux was performed within 3 hours. With TLC (ethyl acetate: hexane3:1) monitoring, the Schiff base precipitant was developed after cooling, they were filtered, dried, and recrystallized from hot ethyl alcohol, and also the m.p and yield were determined.
Azo-Schiff base synthesis
A 0.01 mole of the previous three di-halogenated amines were dissolved in 18% HCl and cooled within 5 °C, and then 0.01mole of NaNO2 dissolved in 10 mL DW was added slowly with stirring and cooling at the same degree until the diazonium salt formed within 10 minutes.
A 0.01 mole of the coupling component (DCSS) was dissolved in 10% alkaline alcoholic solution, and to this component, the diazonium salt solutions were gently added with stirring, keeping the temperature not raised more than 5 °C, they observed the color developing and the azo derivatives compounds were precipitated, when the addition was completed, and for complete precipitation, the whole solutions should be neutralized, and they filtered, washed twice with DW, and dried by air, the yields of the new azo derivatives compounds were assumed and their m.ps were recorded as presented in Table 1. Scheme 1 shows the preparation of azo-Schiff base ligands.
Synthesis of complexes
The solid complexes of the five divalent (Co, Ni, Cu, Pd, and Pt) ions were prepared, after the mole ratio (M:L) determination (1:1 for all ions except Cobalt and copper 1:2), by the reaction of (2 mmol) alcoholic ligands solutions with (1 mmol) of the cobalt and copper chlorides, and (1 mmol) of the same ligands with (1 mmol) of the other three chloride ions solutions, by refluxing them for 3 hours, and monitoring them by TLC techniques (ethanol: chloroform 2:1), when the reaction complete, the solution was reduced to a minimum and cooling with an ice bath, the colored solid complexes were developed, and they were filtered and dried in an oven (50 °C), and their yields and m.p were determined, as listed in Table 1.
Biological study
Antibacterial activity
The synthesized ligands and their complexes were screened for their biological activity against two strains of bacteria Staphylococcus aureus and Escherichia coli by the wells method [26]. Mueller-Hinton agar was used as a culture medium for bacterial growth. All compounds were dissolved in DMSO. Tetracycline was used as a reference antibiotic and DMSO as a control. The zones of inhibition were determined at the end of the incubation period of 24 hours at 35 °C.
Antioxidant
The electron-donating ability of samples and standard tannic acid were determined from bleaching of purple-colored methanol solution of DPPH. Free radical scavenging activity of test samples was measured according to Brand-Williams et al. [27, 28]. DPPH (5 mg/100 mL) was prepared as a normal solution in MeOH. Methanol with DPPH was used as a control. Different concentrations (25, 50, 75 μg /mL) of samples were taken in separate test tubes and volumes were made up to 20 μl using methanol. Next, 200 μl of DPPH solution was added to each test tube and these solutions were kept in dark for thirty minutes. The same procedure was followed for tannic acid as well. All the samples were tested in triplicate. Later optical density was recorded at 517 nm using a spectrophotometer. The decrease in absorbance indicates increased radically scavenging activity, which was determined by the following formula [29].
Cell line assay
MCF-7 human breast cancer cell line was obtained from the American Type Culture Collection (ATCC, Manassas, VA, USA). Cells were grown and maintained in Dulbecco’s Modified Eagle Medium (DMEM; Gibson, Life Technologies, Waltham, MA, USA) supplemented with 10% fetal bovine serum (FBS; Bio West SAS, Nubile, France) and 1% PSF (antibiotic antimitotic solution, Sigma-Aldrich®, St. Louis, MO, USA) in a humidified incubator with 5% CO2 in air at 37 °C. After reaching ~75% confluence, cells were detached using 0.25% trypsin (Gibson, Invitrogen, Waltham, MA, USA) and 0.1% ethylenediaminetetraacetic acid (Merck, Darmstadt, Germany) in phosphate-buffered saline (PBS) at 37 °C. Cells were then re-suspended in DMEM with 10% FBS and 1% PSF. The cells were seeded onto the 96-well plates at a density of 5000 cells per well and incubated for 24 hours prior to the experiments. The cells were washed with PBS (phosphate-buffered saline, pH 7.4) and incubated in a fresh medium containing different concentrations of samples (1000, 500, 250, 125, 62.5, 31.25, 0 µg/ml) for 72 hours. The cell viability assay was measured using the 3-(4, 5dimethylthiazol-2-yl)-2, 5-diphenyltetrazolium bromide (MTT) dye reduction assay. MTT was performed to determine the cytotoxic effect of the samples at various concentrations. After 72 hours of incubation (37 °C, 5% CO2 in a humid atmosphere), MTT (5 mg/mL in PBS) was added to each well, and the plate was incubated for a further 4 hours at 37 °C. The resulting formazan was dissolved in 100 µl of DMSO with gentle shaking at 37 °C, and absorbance was measured at 570 nm with an ELISA reader. The results were given as the mean of three independent experiments. Concentrations of samples showing a 50% reduction in cell viability (i.e. IC50 values) were then calculated.
Results and Discussion
Shibata method for azo compounds synthesis was followed for the synthesis of these new ligands after the preparation of the Schiff base (DCSS) coupling component. They were thermal stable solids, no hygroscopic, and soluble in the most organic solvents were insoluble in water.
Spectroscopic results
A huge number of techniques were used for organic compounds characterization, monocrystalline X-Ray technique is still the unique more precise one for the identification, but when it is abstinence, the researchers resort to use the least accurate one such as the FT-IR techniques, that can gives the agreed information about the nature and type for the functional groups in organic compounds [30].
The IR data are provided in Table 2, and the mean functional groups appeared.
In the Schiff base (DCSS) spectra, the hydroxyl group appeared at 3219 cm-1, the azo-methine group (C=N) [31] at 1613cm-1, as displayed in Figure 1. While in the new azo-Schiff ligands spectra, we noticed the new azo group (N=N) appeared within 1470 cm-1 regions [32], with these mentioned in the Schiff base spectra, these illustrated in Figures 2, 3, and 4, and Table 2.
For the new azo Schiff ligands complexes spectra, found clear changes in intensities and cites of some of their functional groups (shifting somewhat) due to the coordination [33] between the lone pair of the azomethine nitrogen atom and the hydroxyl oxygen atom in the vacant orbitals of the metallic ions forming bidentate chelation, while the azo group still in their regions i.e. non-sharing in the coordination, indeed to the appearance of the M-N and M-O frequencies [34] that indicate the coordination, and the water of coordination frequencies [35] was present in the complexes spectra's, all these can be illustrated in Figures 5, 6, and 7, and Table 2.
Mass spectrometry
Molecular species (organic, inorganic, and polymers) [36] can be precisely detected using an adequate technique such the mass spectrometry, that able to give successive degradation of the target compounds, the molecular masses can easily be informed, the stability of the fragments can be expressed by their intensities.
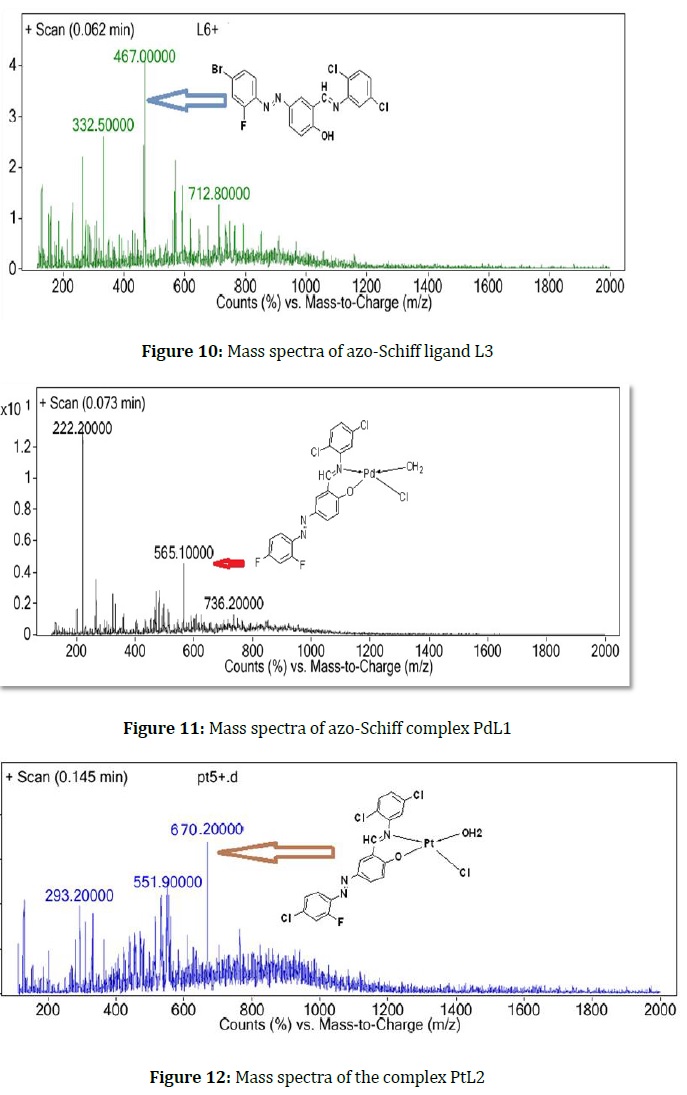
The studied ligands and some of their complexes were measured by this technique with a high degree of precision, that L1, L2, and L3 give the mother ion of their masses at m/z values equal to M = 406, 422, and 467, respectively, as in following Figures 8, 9, and 10.
Some ligand complexes' masses were determined via ESI mass techniques, and they give frequents equal to their masses with m/z values (565, 670, and 578), respectively, for the PdL1, PtL2, and NiL3 complexes as shown in Figures 11, 12, and 13.





1H-NMR spectroscopy
In solutions, the structure of the organic compounds can be deduced via the utilizing of the magnetic resonance techniques [37], when the IR spectra reveal the types of functional groups present in a molecule, 1H-NMR gives information about the number of magnetically distinct atoms of the hydrogen nuclei and the nature of the immediate environment of each type [36].
The free ligands spectra showed the mean signals at δ=12.74, 12.87, and 13.02 related to the hydroxyl phenolic protons [38], δ (1H) azomethine (HC=N-) protons at δ=9.05, 9.07, and 9.17, and a multiple signals related to the aromatic rings protons at the ranges δ 7.47-7.39 ppm, 7.66-7.51 ppm, and 7.68-7.66 ppm for each of L1, L2, and L3, respectively, indeed to the appearance of a signal at ς 3.3 that may be related to the humidity. These can clearly be observed in Figures 14, 15, and 16.
While for these ligands complexes, the coordination effects were clearly visible in their spectra's via some changes as the disappearance of the phenolic hydroxyl protons (due deprotonated and confirm the bonding between the oxygen and ions) [39], with the somewhat shifting in the original ligands signals, this can be shown in Figures 17, 18, and 19.
UV-Visible spectroscopy
Azo ligands can act as a chromogenic reagent for a huge number of different ions [40] and, indeed, to their ability to detect trace amounts of these ions in different samples [41].



In this study, the free ligands (L1, L2, and L3) gave two principle bands, as mentioned in Table 2 and observed in Figures 20, 21, and 22, that related to the electronic transitions (n→π^* and π→π^*), respectively, while a red shifting in their complexes transitions was taken due to the coordination [42] between the ligands nitrogen and oxygen lone pairs to the vacant orbitals of the selected ions, indeed, to the color change of these complexes comparing to its ligands and these can also be observed in the same Table 2, and Figures 20, 21, and 22.
Conductivity and magnetic susceptibility
To decduce complexes formula, the researcher needs some information such (as molar conductivity and magnetic susceptibility), that enhanced them to geometries suggestion.
Molar conductivity [43] is proportional to the charged particles (ions or molecules) in their solutions (solvent) and the ionic characteristics of complexes are determined, by measuring the conductivities in a low dielectric constant solvent, from the results in Table 3 for the conductivity measurements in two solvents (DMF and DMSO), the depression in their conductivity values indicate the non-ionic characteristic of the study complexes.

The magnetic susceptibility of the complexes plays an important role in the suggestion of the complex geometries [44] via the determination of the number of odd electrons count and the oxidation state of the metal ions.
Based on the results, cobalt complexes gave values that were equivalents to the presence of three odd electrons in the 3d orbitals that were compatible with the octahedral cobalt complexes [45].
Copper complexes gave values agreed with the presence of a single electron, which may suggest an octahedral copper complex [46], while the d8 ions (Nickle, Palladium, and Platinum) [47, 48] complexes did not give a reliable value due to their diamagnetic properties, as provided in Table 3.

Biological evaluations
Antibacterial screening
The bacterial assay was tested for azo-Schiff ligand and their metal complexes using two types of bacterial gram-negative (E.coli) and gram-positive (S.ureus), compared with tetracycline as a reference antibiotic, the results are observed in Table 4 and Figure 23.
Based on the collected data, it is clear that the formation of complexes boosts the antimicrobial activity. Such increased activity of complexes may be related to the concept of overtone and the chelation theory.
According to the concept of overtone [49], the presence of a lipid membrane around the cell wall allows lipid and soluble substances to pass easily and this enhances the antimicrobial activity of the bacteria. Regarding the chelation [49, 50]. The orbital overlap of the ligand resulting from the partial participation of the metal ion with the donor group leads to a decrease in the polarity of the metal ion and an increase in the spread of electrons on all chelating rings and, in turn, enhances the fatty alpha of the complexes and breaks the cell permeability barrier, and thus delays the normal cellular processes.
Antioxidant assay
According to the results, it is clear that all the synthesized compounds showed excellent inhibiting activity compared with the standard because all synthesized compounds contain electron donor groups such as nitrogen and oxygen, which have the ability to scavenge free radicals, and thus increase the antioxidant activity of ligands and their metal complexes [51]. In addition, there have been some reports in the literature of metallic complexes with antioxidant activity in ligand. It is believed that the metal moiety will enhance that activity [52-54] because the addition of the metallic moiety improved the ligand's proton donor capacity. The antioxidant activity of ligands and their metal complexes was increased, as depicted in Figure 24.
MC7 cell line breast cancer assay
Two of azo-Schiff base complexes (PdL1 and PtL2) were examined for breast cancer MC7 cell line assay to estimate their abilities for breast cancer inhibition, the results are observed with their IC50 values, in Table 5 and Figures 25, 26, and 27.
In this study, the platinum azo-Schiff complex (PtL2) IC50's value (the minimum concentration required for 50% inhibition in vitro) was 56.78 μg/mL and a cytotoxicity percent of 56.23%. In this regard, a moderate value due to the presence of the platinum ion (Pt+2) as these presents in cis- Platin and its analogs drugs [55, 56] and the substituted fluoride ions, as in fluorinated anticancer agents [57].



The other azo-Schiff complex (PdL1) IC50's value was 74.64 μg/mL and a cytotoxicity percent of 50.25 %, showing less value than platinum complexes, and these types of halogenated compounds can develop a sensible anticancer ability, indeed, to the previously mentioned.
Conclusion
Three new halogenated azo-Schiff ligands were synthesized via the coupling of the diazonium salt of di-substituted halogen anilines with aniline derivatives which was previously mentioned (DCSS) as a coupling component to form these new ligands (L1, L2, and L3) and their transition metal divalent ion complexes for (Co, Ni, Cu, Pd, and Pt) using various spectroscopic techniques for improving their structures as discussed above. According to data, the suggested geometries for the cobalt and copper ions complex have an octahedral complex, and a square planer geometry for d8 (Ni, Pd, and Pt) ion complexes, the ligands serve as a bidentate chelating ligand via the azomethine nitrogen atom and the monobasic oxygen atom of the salicylaldehyde. The magnetic study of the complexes reveals a high spin (cobalt and copper) ion complexes compared with the low spin (Nickel, Palladium, and Platinum) ion complexes, the complexes are more thermally stable than the ligands themselves. The results are agreed with the compositions of the ligands and their complexes as determined via the elemental analysis.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Authors' contributions
All authors contributed to data analysis, drafting, and revising of the paper and agreed to be responsible for all the aspects of this work.
Conflict of Interest
The author declared that they have no conflict of interest.
HOW TO CITE THIS ARTICLE
Dhekra. J. Hashim, Saad.M.Mahdi. Preparation, Characterization, and Biological Study of New Halogenated Azo-Schiff Base Ligands and Their Complexes. J. Med. Chem. Sci., 2023, 6(7) 1555-1576


