Document Type : Original Article (Special Issue)
Authors
1 Department of Pharmaceutics, College of Pharmacy, University of Mosul, Mosul, Iraq
2 Department of Pharmaceutical Chemistry, College of Pharmacy, University of Mosul, Mosul, Iraq
Abstract
Citrullus lanatus (Citrullus L.) co-products have been shown in multiple studies to have biological attributes, attempting to make them a naturally-derived source for medicinal products. The pharmacological relevance of these co-products is linked to the quality and quantity of important phytoconstituents, such as saponins, alkaloids, fatty acids, phenolics, citrulline, lycopene, coumarins, minerals, and other natural co-products. In this study, the effects of varying heating temperatures (40, 50, 60, and 70 °C) on the phytoconstituents’ qualities and bioactivities of a Citrullus L. seed aqueous extract were investigated. This work involved abstracting the seeds from the Citrullus L. fruit, crushing the acquired seeds, and defatting the obtained powdered materials. The crude was extracted with water for 3 hours under several defined heating temperatures. The resultant four extracts were inspected for the availability of some important essential and non-essential phytochemicals. Also, the in vitro biomedicinal activities of these extracts were evaluated and correlated to the found chemicals. The investigated activities included the cytotoxic effect (CE) against many lines abstracted from various cancer phenotypes, the harmless effect (HE) on line of normal cells, free-radical neutralizing effect (FE), and dual 5-LOX/COX inhibitory effects (DE). The findings demonstrated that the variation in the heating temperature has a significant influence on the quality of the abstracted phytoconstituents and their biomedicinal effects. This conclusion may shed light on the use of the seed aqueous extract to manage many disorders, including cancer, inflammation, and those linked to oxidative overload.
Graphical Abstract
Keywords
Main Subjects
Introduction
Various chronic diseases have emerged as a result of substantial changes in human lifestyle and food habits during the previous century. Chronic diseases like cardiovascular disease, diabetes, cancer, inflammation, and neurologic disorders are worldwide health issues that claim the lives of millions of people. Fruits, vegetables, and grains have been shown to have a preventive impact on the development of several of these chronic diseases [1,2].
Since immemorial times, plants and their products have been used as folk medicine in several parts of the world. The earliest record documenting the use of about 1000 plant-derived remedies in Mesopotamia dates back to 2600 BC. It is thought that around 400,000 equatorial flowering plant types have therapeutic properties [3].
In this regard, phytochemical molecules that encompass a diverse range of chemical structures, including phenolic compounds, peptides, pigments, vitamins, minerals, phytosterols, amino acids, fatty acids, and others, can be found in varying amounts in vegetables, fruits, and cereals [4].
The development of novel pharmaceuticals from natural sources is still a difficult undertaking that requires a lot of effort. The collection, extraction, isolation, purification, and characterization of the natural product usually precedes the identification of its pharmacological and toxicological effect [5]. Despite these challenges, natural products continue to provide a large source of chemical molecules with unique molecular frameworks and mechanisms of action [6].
Citrullus L. is the largest cultivated species of the Cucurbitaceae family, with significant economic importance and widespread consumption worldwide [7]. It accounts for roughly 7% of global vegetable output, with China accounting for 67% of the total. Bioactive chemicals with different chemical structures include xanthophylls, carotenoids, phenolic compounds, citrulline, and unsaturated fatty acids [8]. When comparing the beneficial effects of these chemicals on human health, the high quantities of total polyphenols, vitamin C, citrulline, and lycopene (about 40% more than fresh tomatoes) suggest the importance of this fruit compared with other crops [9].

Figure 1: The visual appearance of Citrullus L. that illustrates its seeds
The aim of this study was to investigate how different heating temperatures (40, 50, 60, 70°C) affected the phytoconstituents' qualities and bioactivities of a Citrullus L. seed aqueous extract. For each defined temperature, two concentrations were prepared (5% and 10%) to investigate the influence of the concentration variation on the examined biomedicinal effects. The in vitro investigation of these effects included the CE tested versus five common lines abstracted from various tumors and HE tested on one line of normal cells. The FE was tested by specifying the capacity of the obtaining extracts to neutralize two types of free radicals and detecting the extracts’ total reducing power. Finally, the DE was tested by quantifying the ability of the acquired extracts to inhibit three inflammation-mediated enzymes, including COX-1, COX-2, and 5-LOX.
Materials and methods
All solvents, reagents, and cancerous cell-lines and their cultures hired in this study were obtained from Tokyo Chemical Industry and Sigma-Aldrich, and they were of analytical grade. The fruits were bought from a native market and their botany specified by a proficient taxonomist from Mosul University's College of Agriculture and Forestry. The analytical grade of the laboratory water-chamber provided with a mechanical vibrator and thermostat was employed in the maceration achievement.
Algorithm of extracts acquiring
Each seasoned Citrullus L. fruit from the tested batches was sanitized by hand with water from the tap and afterwards decanted into pure water before being individually chopped into 8 wedges with a serrated blade meat cleaver. The seeds were crushed in a medicinal herb mixer and mesh sieved to produce a crystalline dust after being shade-dried for 21 days at room temperature in darkness. This dust was kept in tightly sealed emerald vessels and put in the fridge till prepared to be used in the advanced phase [10].
Analysis of phytochemical compounds
To identify a variety of phytochemical compounds, successful methods of analysis have been used to conduct phytochemical screening of Citrullus L. seed aqueous extracts [11-14]. The inspected phytochemical compounds were classified into two groups: essential and non-essential fruit-derived chemicals. The formal group consists of sugars, steroids, proteins, fatty acids, and amino acids while the latter group is constructed from coumarins, flavonoids, alkaloids, emodins, anthraquinones, saponins, anthocyanins, betacyanins, phenolics, tannins, and terpenoids.
Evaluating procedures of the biomedicinal effects
Assessment of the FE
Maceration-derived aqueous extracts (5 and 10% w/v percent) were tested for their ability to neutralize the reactive moieties of DPPH (1,1-diphenyl-2-picrylhydrazyl) and hydroxyl, as well as make a significant electron contribution in an electron-dependent interaction. The neutralizing percentage (N%) results of three independent replicates for each extract phenotype were computed at a certain colored wavelength, using the standard mathematical law:
The absorption readings of the control and investigated maceration-derived aqueous extract were designated by the symbols Abscontrol and Abssample, correspondingly [15].
DPPH-free radical neutralization assessment
The maceration-acquired aqueous extract (1.5 ml, 5 or 10% w/v percent) was blended with an ethanolic solution of DPPH (0.5 ml, 0.1 mM). The covered system was kept at 25°C for 30 minutes after the mixed solution was layered with aluminum platelets to shield it from sunlight. At 517 nm, the extract's capability to obliterate the DPPH purple color was measured via a visible photometer. The control solution for this assessment consisted of 1.5 ml of distilled water and 0.5 ml of ethanolic DPPH solution [16].
Hydroxyl-free radical neutralization assessment
The maceration-acquired aqueous extract (1.5 ml, 5 or 10% w/v percent) was mixed with 2.4 ml of potassium phosphate-buffered saline (0.2 M, pH 7.8). To this mixture, 60 μl of 0.001 M ferric chloride, 90 μl of 0.001 M pyridino[3,2-h]quinoline, and 150 μl of 0.17 M dihydrogen dioxide were added in that sequence. The resulting combination was examined via a visible photometer at 560 nm after being maintained at 25°C for approximately 5 minutes. The control solution for this assessment had all of the above ingredients except the aqueous extract, which was substituted with distilled water [17].
Assessment of total reducing power capacity (TRP)
The maceration-acquired aqueous extract (1 ml, 5 or 10% w/v percent) was treated with 2 ml of potassium phosphate-buffered saline (0.2 M, pH 6.6) and 2 ml of 1% aqueous potassium ferricyanide solution. For 20 minutes, the combination was kept at 50°C in a thermo-scientific digital water bath, and subsequently treated with 2 ml of 10% aqueous trichloroacetate solution to slow down the reaction. The resultant blend was then centrifuged at 2000 rpm for 10 minutes. A 2 mL supernatant was added to a combination consisting of 2 ml of distilled water and 0.4 ml of 0.01% aqueous iron perchloride solution. The resultant combination was examined via a visible photometer at 700 nm after being kept at 25°C for 10 minutes. The control solution for this assessment was prepared in a similar method as the test mixture, but the aqueous extract was substituted by distilled water [18].
Assessment of the CE
The MTT-color probe methodology was used to detect the viability of tumor/normal cells when treated with our aqueous extracts. The specific tumor/normal cells (10000 cells) were placed to each well of a 96-well microplate and treated separately after 24 hours with a specified maceration-acquired aqueous extract. The next 72 hours of treatment were spent separating the medium, providing the MTT-color probe (27 μl, 3.28 mM), and incubating the treated cells for 1.5 hours at 37°C. A microplate photometer adjusted to 492 nm was used to measure the readings values of the treated (Wt) and untreated (Wu) wells absorbances. The following formula was used to compute the percent of viability suppression (VS%) of three separate trials [19,20].
Assessment of the DE
5-Lipoxygenase (5-LOX) inhibitory assessment
The capacity of our aqueous extracts to inhibit the catalytic activity of 5-LOX enzyme was tested using the human recombinant 5-LOX estimate model, coded 437996, which was bought from Sigma-Aldrich. In summary, 100 μl of a defined aqueous extract, 90 μl of 5-LOX solution, 100 μl of standard chromogen solution, and 10 μl of standard substrate (arachidonic acid, 0.1 mM) constitute the test combination. The values of absorbance were determined via a visible photometer at 490 nm after a 10-minute incubation period and in comparison to the blank [21].
Cyclooxygenase (COX-1 and COX-2) inhibitory assessment
The ability of our aqueous extract to inhibit the catalytic capacities of COX-1 and Cox-2 enzymes was assessed using the ovine and human COX blocker estimation models, respectively. These kit models were acquired from Cayman and designated 560131. In summary, 100 μl of a defined aqueous extract, 0.96 ml of Tris-hydrochloric acid buffered solution (0.1 M), 10 μl of COX-1/COX-2 enzyme solution, and 10 µl of the standard substrate (arachidonic acid, 0.1 mM) constructed the test blend that was treated with 50 μl of Ellman's reagent (1 μM) after a 10-minute incubation at 37°C. The readings of color absorbance were specified at 410 nm via a visible photometer and compared to the blank [22].
Results and discussion
Many ancient societies have embraced the use of herbal products due to their safety and feasibility, allowing them to gain benefits from their biological activity [23]. More specifically, aqueous extracts derived from various sections of a variety of plants are commonly used in traditional home treatments [24-27].
Bibliography of phytochemical products
Our aqueous extracts were analyzed to specify the quality of their contained phytochemical products using standard screening protocols. The results indicated the bibliographical catalogs of these extracts were as follows: YN1 and YN2: Proteins, amino acids, carbohydrates, glycosides, and steroid; YN3 and YN4: Proteins, amino acids, carbohydrates, glycosides, steroid, and tannins; YN5 and YN6: Proteins, amino acids, carbohydrates, glycosides, steroid, tannins, terpenoids, and emodins; YN7 and YN8: Proteins, amino acids, carbohydrates, glycosides, steroid, tannins, terpenoids, and anthocyanin, phenolics, and flavonoids.
From these bibliographic catalogs, the authors concluded that the increase in the extracting temperature resulted in improving the quality of the extracted phytochemical products [28-30].
Citrullus L. seed aqueous extracts as antioxidant crudes
Current studies on free radicals support the importance of antioxidant-rich fruits in the prophylaxis and management of various oxidative overload-related diseases involving cancer and cardiovascular diseases. This medicinal property may contribute to the presence of several qualified co-products that accumulate in different areas of these fruits and lend pharmacological significance to them [31].
The results of comparing the ability of our aqueous extracts to act as antioxidant crudes to the standard antioxidant were reported in Table 1 and pictorially displayed in Figure 2.
Table 1: The findings obtained from examining the FE of our aqueous extracts
|
Symbol of examined solution |
DPPH-free radical neutralization |
Hydroxyl-free radical neutralization |
TRP |
|
LA1 |
12.45±1.02 |
11.24±0.78 |
11.08±1.08 |
|
LA2 |
17.01±0.98 |
13.62±0.82 |
12.87±1.03 |
|
YN1 |
2.17±1.14 |
1.36±0.91 |
1.02±0.99 |
|
YN2 |
2.89±0.92 |
1.57±0.97 |
1.25±0.92 |
|
YN3 |
2.34±1.16 |
2.02±1.01 |
1.56±1.12 |
|
YN4 |
3.16±0.87 |
2.49±0.98 |
1.64±1.05 |
|
YN5 |
4.04±1.02 |
3.09±0.85 |
2.68±1.04 |
|
YN6 |
4.92±0.89 |
3.42±0.96 |
3.04±1.02 |
|
YN7 |
6.54±0.96 |
6.12±0.90 |
6.36±0.98 |
|
YN8 |
7.68±1.15 |
7.62±0.83 |
6.91±1.01 |
L-ascorbic acid (0.5%, 25°C), L-ascorbic acid (1%, 25°C), maceration-acquired aqueous extract (5%, 40°C), maceration-acquired aqueous extract (10%, 40°C), maceration-acquired aqueous extract (5%, 50°C), maceration-acquired aqueous extract (10%, 50°C), maceration-acquired aqueous extract (5%, 60°C), maceration-acquired aqueous extract (10%, 60°C), maceration-acquired aqueous extract (5%, 70°C), maceration-acquired aqueous extract (10%, 70°C), and standard deviation of three independent experiments are symbolized here as LA1, LA2, YN1, YN2, YN3, YN4, YN5, YN6, YN7, YN8, and SD. The results are estimated as mean L% or TRP±SD.

Figure 2: Schematic illustration of the findings from assessing our aqueous extracts as antioxidant crudes
The results in Table 1 enabled the authors to sketch a variety of conclusions. The first is that, under the conditions of this investigation, the FE of the maceration-acquired aqueous extracts was encouraging and satisfactory, but it was still lower than that of the reference L-ascorbic acid at the two concentrations used. Second, in decreasing FE order, they are: YN8, YN7, YN6, YN5, YN4, YN3, YN2, and YN1. This order indicates that as concentration and temperature rise, the FE will rise as well. This also suggests that as the quality of the extracted phytochemical products in our aqueous extracts improves, so will the FE. Finally, the presence of phenolics and flavonoids in the extracts YN7 and YN8 may be responsible for their improved FE, according to these findings.
Citrullus L. seed aqueous extracts as cytotoxic crudes
Our aqueous extracts were evaluated for their CE versus six common lines abstracted from various tumors, including KYSE-30, SK-OV-3, AMN3, SKG, HeLa, and MCF-7. The full names of these cell lines are Human Asian Esophageal Squamous Cell Carcinoma, Caucasian Ovary Adenocarcinoma, Murine Mammary Adenocarcinoma, Human Papillomavirus, Related Cervical Squamous Cell Carcinoma, Epithelioid Cervix Carcinoma, and Caucasian Breast Adenocarcinoma, respectively. These tumor lines' global regulations are 94072011, 91091004, CVCL-M395, C27676, 93021013, and 86012803 [32-34]. This assessment was carried out using MTT-color probe relying methodology with two different concentrations of 5-Fluorouracil (FU) as favorable standards and methanol as a deleterious standard.
The gained results are reported in Table 2 and pictorially displayed in Figure 3. Investigating the research results in this table revealed a number of noteworthy points. First, our aqueous extracts have a high CE versus the cancerous cell lines tested [35]. Second, the declining CE was in the correct sequence: YN8, YN7, YN6, YN5, YN4, YN3, YN2, YN1. This order implies that the CE was increased by increasing the phytochemical products in the extracts. Furthermore, as the temperature goes up, the CE would then improve. Third, this CE mode is significant compared with the FE of our aqueous extracts [36]. As a result, the authors believe that the CE of our aqueous extracts is due to their ability to satiate harmful free radicals. Fourth, the greatest effect was observed for the extracts YN7 and YN8, particularly when compared with the MCF7 cell-line, which could be related to the presence of phenolics and flavonoids phytoconstituents not found in other aqueous extracts [37]. Finally, concerning HE, our aqueous extracts showed a significant and harmless effect on normal cells compared with the aqueous FU solutions employed [38].
Table 2: The findings obtained from examining the CE and HE of our aqueous extracts
|
Symbol |
HeLa |
MCF7 |
SKOV3 |
KYSE30 |
AMN3 |
RWPE1 |
|
FU1 |
16.43±1.07 |
16.68±1.02 |
11.46±0.90 |
8.44±0.92 |
11.02±0.96 |
19.06±1.05 |
|
FU2 |
18.11±0.96 |
18.34±1.08 |
12.67±0.89 |
8.89±1.02 |
11.56±0.81 |
21.26±1.04 |
|
YN1 |
1.96±0.87 |
1.76±0.93 |
1.02±0.86 |
0.83±0.78 |
1.14±1.02 |
0.57±0.92 |
|
YN2 |
2.08±0.92 |
1.95±1.03 |
1.04±0.92 |
0.84±0.87 |
1.18±1.04 |
0.58±1.02 |
|
YN3 |
2.06±1.12 |
1.98±1.00 |
1.06±1.03 |
0.98±0.95 |
1.16±0.94 |
0.51±0.98 |
|
YN4 |
2.57±1.03 |
2.26±0.88 |
1.12±0.90 |
1.08±0.96 |
1.21±0.96 |
0.55±1.03 |
|
YN5 |
2.46±1.11 |
2.23±0.92 |
1.43±0.97 |
1.37±0.92 |
2.59±0.88 |
0.61±0.87 |
|
YN6 |
3.02±0.98 |
2.86±0.90 |
1.79±1.03 |
1.56±1.05 |
2.87±0.91 |
0.63±0.94 |
|
YN7 |
5.41±1.03 |
6.72±0.79 |
5.36±1.02 |
3.12±0.93 |
4.46±1.05 |
0.63±0.97 |
|
YN8 |
5.56±0.87 |
7.12±0.94 |
6.01±1.10 |
3.40±0.79 |
4.93±0.91 |
0.62±1.12 |
The results are reported as mean VS%±SD. The symbols FU1 and FU2 represent the aqueous solution of FU with a concentration of 0.5% and 1%, respectively
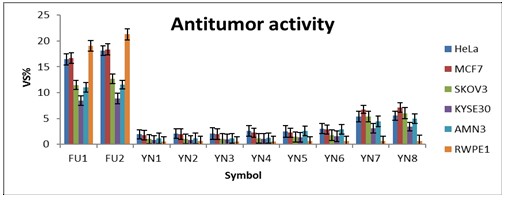
Figure 3: Schematic illustration of the findings from assessing our aqueous extracts as cytotoxic crudes
Citrullus L. seed aqueous extracts as anti-inflammatory crudes
The pro-inflammatory enzymes, including COX-1, COX-2, and 5-LOX, are important enzymes in the arachidonic acid metabolism and required for the physiological synthesis of inflammatory eicosanoids, such as leukotrienes, prostaglandins, and thromboxane. So, the kits of these inflammatory-related enzymes are employed to test and evaluate anti-inflammatory candidates because they represent the rate-limiting steps in the synthesis of the abovementioned eicosanoids [39].
The values reflected the DE activities of our aqueous extracts and aspirin at 0.25% and 0.5% concentrations as controls, as shown in Table 3 and graphically illustrated in Figure 4. In addition, this table includes the selectivity marker (SM) values, which were calculated for each investigated solution by the following formula: COX-1 blocking percent/ COX-2 blocking percent [40]. This matrix, as shown in Figure 5, can help to predict COX-2 selectivity of the employed solutions (COX-2 selectivity is increased in solution with a higher SM value) [41].
Table 3: The findings obtained from examining the DE of our aqueous extracts
|
Symbol |
COX-1 |
COX-2 |
SM |
5-LOX |
|
AN1 |
18.95±1.02 |
2.65±0.89 |
7.15 |
0.92±0.86 |
|
AN2 |
20.03±0.85 |
2.81±0.96 |
7.13 |
0.98±0.89 |
|
YN1 |
1.34±0.93 |
0.84±0.78 |
1.60 |
1.12±1.04 |
|
YN2 |
1.35±0.88 |
0.87±0.91 |
1.55 |
1.17±1.01 |
|
YN3 |
1.42±1.03 |
0.92±1.03 |
1.54 |
1.22±0.89 |
|
YN4 |
1.54±0.89 |
0.93±1.11 |
1.66 |
1.29±0.90 |
|
YN5 |
1.43±1.12 |
1.01±1.02 |
1.42 |
1.41±1.01 |
|
YN6 |
1.61±0.91 |
1.09±0.89 |
1.48 |
1.49±0.88 |
|
YN7 |
1.54±1.02 |
1.25±0.90 |
1.23 |
1.98±0.79 |
|
YN8 |
1.74±0.85 |
1.33±1.02 |
1.31 |
2.32±1.06 |
The results are recorded as mean EB% (enzyme blocking percent) ±SD. The symbols AN1 and AN2 represent the aqueous solution of aspirin with a concentration of 0.25% and 0.5%, respectively.

Figure 4: Schematic illustration of the findings from assessing our aqueous extracts as anti-inflammatory crudes

Figure 5: Schematic illustration of the SM values concerning the reference solutions and our aqueous extracts
The analysis of the results in Table 3 yielded four major conclusions. To begin with, the effect of the maceration-acquired aqueous extracts on 5-LOX enzyme was very enticing and greater than that acquired from the reference control by its two concentrations, with the extract YN8 showing the best effect compared with the other extracts. The phenolics and flavonoids found in this aqueous extract have been linked to its powerful impact against the 5-LOX enzyme, according to the authors. Second, the three inflammatory-related enzymes are blocked in parallel and in a concentration-dependent manner [42]. Third, maceration-acquired aqueous extracts with a concentration of 5% showed greater selectivity for COX-2 over COX-1 than extracts with a concentration of 10%. Finally, the DE is inextricably linked to the FE and CE.
Conclusion
Based on the outcomes of this research study, the authors concluded that the quality of the Citrullus L. seed aqueous extract improves as the extracting temperature increases within the applied heating range. This improvement was manifested by the appearance of certain phytochemical products involving phenolics and flavonoids. Also, the outcomes indicated that our aqueous extracts have good antioxidant, cytotoxic, and dual 5-LOX/COX inhibitory effects. Moreover, the antioxidant capacity of these extracts is in the same direction as their cytotoxicity and anti-inflammation abilities. The authors attributed the last two effects to the capacity of the investigated aqueous extracts to neutralize the damaging free radicals. Finally, and based on the cellular safety profiles of our aqueous extracts, the authors recommend that these extracts can represent promising crudes for managing various types of cancer, inflammatory conditions, and disorders related to oxidative overload.
Acknowledgment
The authors gratefully thank the University of Mosul/College of Pharmacy for providing facilities that improved the quality of this work. They are also grateful to Dr. Sara Firas Jasim, Dr. Sarah Ahmed Waheed, Dr. Rahma Mowaffaq Jebir, and Dr. Reem Nadher Ismael for their efforts to improve this work's quality.
Funding
This research did not receive any specific grant from fundig agencies in the public, commercial, or not-for-profit sectors.
Authors' contributions
All authors contributed to data analysis, drafting, and revising of the paper and agreed to be responsible for all the aspects of this work.
Conflict of Interest
There are no conflicts of interest in this study.
ORCID:
Noora Thamer Abdulaziz
https://www.orcid.org/0000-0001-8330-7777
Yasser Fakri Mustafa
https://www.orcid.org/0000-0002-0926-7428
HOW TO CITE THIS ARTICLE
Noora Thamer Abdulaziz, Yasser Fakri Mustafa. The Effect of Heat Variable on the Chemical Composition and Bioactivities of a Citrullus lanatus Seed Aqueous Extracts. J. Med. Chem. Sci., 2022, 5(7) 1166-1176


