Document Type : Case Report
Authors
1 Skull Base Research Center, Loghman Hakim Hospital, Shahid Beheshti University of Medical Sciences, Tehran, Iran
2 Department of Radiation Oncology, Shohadaye Haftome Tir Hospital, Iran University of Medical Sciences, Tehran, Iran
3 Stem Cell and Regenerative Medicine Research Center, Iran University of Medical Sciences, Tehran, Iran
Abstract
The main aim of this study was to find out some new findings about Sinus Approach for Cushing's Disease in Iranian Hospitals with t-test in this way. Although pituitary adenoma (PA) is one of the three most common brain tumors besides glioma and meningioma. Its prevalence is even higher in autopsy series and screening radiologic studies in the normal population. PAs can be either functional (tumor secreting) or nonfunctional. Proportion of different kinds of PAs is reported differently in studies. Up to half of all PAs may be nonfunctional (including FSH/LH secreting tumors). In this research study, we have assessed 84 patients with Cushing disease, retrospectively. The mean age was 49.4 years. There were 49 female 35 male patients. They are classified as usual cases of macroadenomas and microadenomas and a third group of intricate Cushing’s disease (ICD). ICD comprises reoperation cases, MRI- negative patients, and cases with multiple microadenomas. All patients have undergone trans-sphenoid endoscopic surgery. ICD patients are operated either by conventional transsphenoidal endoscopic surgery or by TPTICS modification. Demographic information and postsurgical outcome are compared among these groups. An informed consent was filled and signed by all patients. Statistical analysis was performed using SPSS 20. P values lower than 0.05 are considered to be significant. Chi Square and student t tests were used to compare nominal and continuous variables, respectively, between study groups.
Graphical Abstract
Keywords
Main Subjects
Introduction
Among PAs that need surgical evacuation transsphenoidal microscopic or endoscopic approaches are used most commonly. Endoscopic pituitary surgery in recent years rapidly opens its way to replace the conventional microscopic approach. Recent advances in endoscopic instruments and techniques as well as developing surgical expertise may someday make it the standard surgical treatment of PA [1-4]. Although there is no firm evidence that prefers either of these approaches, better visualization of the lateral walls using different angled lenses and maneuvering the endoscope tip makes the endoscopic transsphenoidal surgery ideal for macroadenomas, invasive adenomas, and tumors adherent to the dural wall [5-7] (Figure 1).

Figure 1: Current therapeutic algorithm for management of Cushing disease [8]
Diminished difference of complete removal rate between microadenomas and macroadenomas in endoscopic surgery versus microscopic surgery may explain the above-mentioned benefits of endoscopic surgery [8-10].
Interestingly in one histological study, the rate of invasiveness in Cushing disease (not Nelson syndrome) was lower relative to the other pathological subtypes of the pituitary adenomas.2 It is obvious that certain types of Cushing’s disease are more prone to respond incompletely after surgery or recur later after a partial response [11-14]. This is mostly due to a remnant tumor [4-6]. It has been said that the best response in reoperation is achieved in circumstances that we have had a recurrent tumor in the same location of previously resected tumor with an initially acceptable response [15] (Figure 2).

Figure 2: Introduction: The Nature of Drugs and Drug Development and Regulation [9]
Cushing syndrome may be the result of a tiny tumor of the pituitary gland. Sometimes the tumor is so small that cannot be detected in imaging studies. In some cases, a blind operation is indicated to detect the tumor intraoperatively and then remove it. In fact, some non-responders may have multiple tumors of which some are not observable in imaging studies. Therefore, it is of crucial importance to not rely on imaging studies in Cushing surgery and perform a very meticulous intraoperative search for any extra tumoral tissue other than what is seen on imaging. In this study, we discuss technical nuances for so-called trans-planum trans-intercave nous sinus (TPTICS) approach, for a wider exposure of the pituitary gland that makes it easier to find any tiny tumoral tissue in the sella or suprasellar space. We suggest using this technique in three selected types of Cushing’s disease with a high probability of missing the remaining adenoma tissue including reoperation cases, MRI- negative patients, and cases with multiple microadenomas which are believed to be associated with lower remission rates. We call them intricate Cushing’s disease (ICD) [16] (Figure 3).

Figure 3: Research for the pharmaceutical and medical industries [12]
In this article Medical and Pharmacological Study of Sinus Approach for Cushing's Disease in Iranian Hospitals is investigated and Case Study is also, 84 Patients with Cushing Disease. We try to use t-test to find some new findings about it [17].
Material and Methods
A t-test is a type of inferential statistic used to determine if there is a significant difference between the means of two groups, which may be related in certain features. It is mostly used when the data sets, like the data set recorded as the outcome from flipping a coin 100 times, would follow a normal distribution and may have unknown variances. A t-test is used as a hypothesis testing tool, which allows testing of an assumption applicable to a population [18].
A t-test looks at the t-statistic, the t-distribution values, and the degrees of freedom to determine the statistical significance. To conduct a test with three or more means, one must use an analysis of variance. Essentially, a t-test allows us to compare the average values of the two data sets and determine if they came from the same population. In the above examples, if we were to take a sample of students from class A and another sample of students from class B, we would not expect them to have exactly the same mean and standard deviation. Similarly, samples taken from the placebo-fed control group and those taken from the drug prescribed group should have a slightly different mean and standard deviation.
Mathematically, the t-test takes a sample from each of the two sets and establishes the problem statement by assuming a null hypothesis that the two means are equal. Based on the applicable formulas, certain values are calculated and compared against the standard values, and the assumed null hypothesis is accepted or rejected accordingly [19].
If the null hypothesis qualifies to be rejected, it indicates that data readings are strong and are probably not due to chance. The t-test is just one of many tests used for this purpose. Statisticians must additionally use tests other than the t-test to examine more variables and tests with larger sample sizes. For a large sample size, statisticians use a z-test. Other testing options include the chi-square test and the f-test. There are three types of t-tests, and they are categorized as dependent and independent t-tests. Consider that a drug manufacturer wants to test a newly invented medicine. It follows the standard procedure of trying the drug on one group of patients and giving a placebo to another group, called the control group. The placebo given to the control group is a substance of no intended therapeutic value and serves as a benchmark to measure how the other group, which is given the actual drug, responds.
After the drug trial, the members of the placebo-fed control group reported an increase in average life expectancy of three years, while the members of the group who are prescribed the new drug report an increase in average life expectancy of four years. Instant observation may indicate that the drug is indeed working as the results are better for the group using the drug. However, it is also possible that the observation may be due to a chance occurrence, especially a surprising piece of luck. A t-test is useful to conclude if the results are actually correct and applicable to the entire population.
In a school, 100 students in class A scored an average of 85% with a standard deviation of 3%. Another 100 students belonging to class B scored an average of 87% with a standard deviation of 4%. While the average of class B is better than that of class A, it may not be correct to jump to the conclusion that the overall performance of students in class B is better than that of students in class A. This is because there is natural variability in the test scores in both classes, so the difference could be due to chance alone. A t-test can help to determine whether one class fared better than the other.
Sellar cavity surrounding pituitary gland is a bony pocket made in the sphenoid sinus which is open superiorly but is separated from the basilar surface of brain by a layer of arachnoid called diaphragma sella. Surgical access to the pituitary gland is limited superiorly, posteriorly, and laterally by optic apparatus and the Willis circle, brain stem and basilar artery, and cavernous sinuses, respectively [20]. So, the least dangerous way of access is a trans nasal trans-sphenoid transsellar route. Dura but not arachnoid layer covers the sellar cavity interiorly. So, pituitary gland is an intradural extra-arachnoid part of the brain. Intra-arachnoid space is located superior to diphraghma sellae through which the pituitary stalk passes toward the third ventricle’s floor.
Inter-cavernous sinuses are vascular channels that are commonly present between the two cavernous sinuses at the margins of diaphragm sellae. They have many variations in morphology. Inter-cavernous channels that are located anterior to the pituitary stalk, usually the largest group, are called anterior inter-cavernous sinuses [21-23].
It is not uncommon for ACTH-producing pituitary adenoma to involve dural lining of the sella7 and we have observed cases with the involvement of inferior or even superior surfaces of the diaphragma sella. Many patients with recurrent or remaining Cushing syndrome may have tumor remnants in these areas [24]. Conventional microscopic or endoscopic transsphenoidal approaches have shortcomings in gaining a good visual access to the mentioned areas (Figure 4).
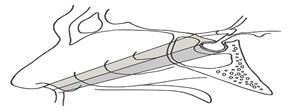
Figure 4: Upper shaded area is the extra visual angle achieved by TPTICS approach relative to the conventional ETSS (lower shaded area). A superior view of the pituitary gland and the sellar cavity as well as a good view to the pituitary stalk, diaphragma sella and optic apparatus are among advantages of this technique [25]
So, after recognition of a certain group of patients, we decided to use a modification and an extension to the conventional ETSS approach to see the pituitary gland and the potential locations of a small ACTH-producing adenoma in a different angle and a wider space [24-26].
In the so-called transplanum trans-intercavernus sinus (TPTICS) approach, we initially get access to the sellar face with conventional routes. We usually use a binostril three to four-handed approach. Middle turbinates are pushed aside bilaterally and if it is needed it is resected in one (usually right) side. Posterior part of the septum, as well as sphenoid rostrum, is resected. Neuronavigation co-registered to MRI and CT scan simultaneously helps stay in the midline and locate the sellar floor especially in reoperation cases [27-29] (Figure 5).

Figure 5. Nano based drug delivery systems: recent developments and future prospects [14]
We usually prepare a vascular-pediculated nasoseptal flap to be used in skull base reconstruction. Sphenoid face is resected with a high-speed drill. Then we remove the sellar floor and continue drilling to remove the planum of the sphenoid bone so that the dura mater of the posterior part of the anterior skull base is exposed [30-32]. The anterior intercave nous sinus is coagulated by bipolar cautery. Two left-to-right incisions are made one in dura that was overlying the anterior sellar face and the other in dura that was overlying the planum sphenoidale. Then the midpoints of these two incisions are connected by a third incision which passes through the coagulated intercave nous sinus [33-35]. Dural flaps are pushed aside. At this stage, we have a panoramic view of the pituitary gland and its stalk and diaphragma sella from an additive anterosuperior angle of view. So, all parts of the sellar cavity, two surfaces of the diaphragma sella, and pituitary stalk can be exposed and investigated accurately for any tumoral tissue. During the operation, all dural walls and both sides of the diaphragma sella are observed for tumor nests and the pituitary gland is mobilized to different direction to see all hidden aspects [36-38].
After tumor removal base of skull is reconstructed with fascia lata graft and is augmented by the nasoseptal flap. Nasal cavities are packed for two days. We do not use lumbar drain for uncomplicated cases. Post-operatively, antibiotics are continued as long as we remove nasal packing and the patient is usually discharged after 48 to 72 hours [39].
Results and Discussion
Of 759 patients with pituitary adenoma treated by endoscopic transsphenoidal surgery, 84 patients had the diagnostic criteria of Cushing’s disease/ Nelson syndrome. Twenty-seven patients had microadenomas with the criteria compatible with ICD. Of these patients, 15 patients underwent the conventional ETSS technique and in 13 of them, a biochemical remission (cortisol level <2 μg/dL and undetectable ACTH in serum samples) was achieved during an average of 18 months follow-up (18.1 +/- 5.2) [40]. Twelve patients underwent the TPTICS approach and a biochemical remission was achieved in all of them with an average follow-up period of 11 months (11.1 +/- 3.6). There was no statistically significant difference in follow-up times between two surgical groups (Pearson Chi-Square asymptotic two-sided significance: p=0.487). Transient diabetes insipidus occurred in 2 cases of ETSS and 4 cases of TPTICS group and all of them subsided with conservative measures. Fortunately, a clinically significant CSF leakage, meningitis, or any other complication did not occur. Although a trend toward a higher remission rate was seen in TPTICS, this was not statistically significant (Pearson Chi-Square asymptotic two-sided significance: p=0.189). According to the anesthesiologist records there was no difference in blood loss in either group, although duration of surgery was longer in TPTICS group (t-test, p=0.04). The mean duration of surgery was 37 min longer in TPTICS group.
Cushing’s disease is one of the most challenging pituitary adenomas as it is problematic in both detection and resection phases. A wide range of remission rates has been reported elsewhere from below 15 % to more than 90 %. These differences are mainly attributable to tumor type composition of series (microadenoma, macroadenoma, Nelson syndrome, and reoperation), application of other treatment modalities such as radiation, and time of follow-ups. A recent literature review has calculated the range of recurrence rate from 3 to 47 % in a range of 16 to 49 months. Remission rate is usually lower in macroadenomas and patients with Nelson Syndrome due to their more invasive nature of the tumor. While in macroadenomas the cavernous sinus invasion causes many treatment failures, remission rate in microadenomas is correlated with intraoperative detection of the tumor. It is not uncommon to see adenomas adherent to dura and even to the arachnoid membrane [4-6]. So, it is of crucial importance to evaluate all dural walls meticulously. Capability of a tiny MRI-negative adenoma to produce a full-blown Cushing’s syndrome and possibility of the presence of multiple microadenomas in the same patient add to the complexity of surgical management of CD.13 In one study, all tumor recurrences occurred at the original tumor place22 which may be due to incomplete removal of the tumor. In comparison, localization of the tumor by surgical exploration is much higher than other modalities such as CT, MRI, or inferior petrosal sinus sampling. So, recurrence of CS in an MRI-negative patient can be best managed by repeat transsphenoidal surgery [22,23].
Lonser et al. [12] evaluated 87 patients with Cushing’s disease and found some evidence of dural invasion in 34% of them. The major site of invasion was the lateral walls. They revealed susceptibility of MRI in the detection of dural invasion including cavernous sinus invasion. So inspection of all dural lining of sellar cavity and removal of any involved dura is very important to gain remission.7 In our personal observations, we encountered many reoperative cases with intraoperative signs of dural invasion. Dural or arachnoid microinvasions may underlie most cases of surgical failure in Cushing’s disease. So, we decided to use a more extensive approach in cases with ACTH-producing microadenoma.
Benefits: TPTICS approach gives us a more panoramic view of the sellar contents as well as the suprasellar part of the pituitary apparatus and diaphragma sella. In TPTICS we look at the pituitary apparatus from an anterosuperior rather than anteroinferior angle and we can move the gland and examine all dural lining for tumor remnants. This may underlie a higher remission rate in our TPTICS group, although it was not statistically significant.
Limitations: There are some limitations for applying TPTICS approach. A larger skull base defect in TPTICS theoretically increases the likelihood of complications such as CSF leak, meningoencephalocele, meningitis, etc., although we did not observe such an effect in this study. Moreover, it lengthens the operation time to provide more exposure as well as more time that is needed for skull base reconstruction. Obviously, it is a more invasive approach relative to conventional ETSS and it should be rationalized to be used for a specific group of patients. So, we applied it in patients with a higher chance of treatment failure which include reoperations, MRI-negative, and multiple microadenoma cases (i.e. intricate Cushing’s disease, ICD) [5-7, 14-16].
Conclusions
In the present work, we discussed a modification to the conventional endoscopic transsphenoidal resection of ACTH-producing pituitary adenoma (TPTICS approach) and we proposed the best patient population that may benefit from the application of this approach (i.e., ICD). We observed a very favorable postoperative outcome for particular patients with Cushing’s disease that have undergone a TPTICS approach without any increment in complication rate. TPTICS should be considered in patients with a high risk of surgical failure; although, it should be assessed further in larger groups with stronger study design and longer follow-up times.
Conflict of Interest
We have no conflicts of interest to disclose.
HOW TO CITE THIS ARTICLE
Guive Sharifi, Masoumeh Najafi, Amin Jahanbakhshi. Medical and Pharmacological Study of Sinus Approach for Cushing's Disease in Iranian Hospitals (Case Study: 84 Patients with Cushing Disease), J. Med. Chem. Sci., 2022, 5(2) 275-282
DOI: 10.26655/JMCHEMSCI.2022.2.15
- Lai C.C., Shih T.P., Ko W.C., Tang H.J., Hsueh P.R., J. Antimicrob., 2020, 55:105924 [Crossref], [Google Scholar], [Publisher]
- Bahadori F., Sahebazzamani Z., Zarei L., Valizadeh N., Tehran Univ Med J., 2018, 76:608 [Google Scholar], [Publisher]
- Tahamtan A., Ardebili A., Expert Rev. Mol. , 2020, 20:453 [Crossref], [Google Scholar], [Publisher]
- Gibson P.G., Qin L., Puah S.H., J. Aust., 2020, 213:54 [Google Scholar], [Publisher]
- Fu F., Lou J., Xi D., Bai Y., Ma G., Zhao B., Liu D., Bao G., Lei Z., Wang M., Radiol., 2020, 30:5489 [Crossref], [Google Scholar], [Publisher]
- Liu J., Yu H., Zhang S., J. Nucl. Med. Mol. Imaging., 2020, 47:1638 [Crossref], [Google Scholar], [Publisher]
- Poortahmasebi V., Zandi M., Soltani S., Jazayeri S.M., J. Emerg. Med., 2020, 4:e57 [Crossref], [Google Scholar], [Publisher]
- Fang Y., Zhang H., Xie J., Lin M., Ying L., Pang P., Pang P., Ji W., Radiology., 2020, 296:E115 [Crossref], [Google Scholar], [Publisher]
- Shi F., Wang J., Shi J., Wu Z., Wang Q., Tang Z., He K., Shi Y., Shen D., IEEE Rev. Biomed. Eng., 2020, 14:4 [Crossref], [Google Scholar], [Publisher]
- Mokhtare M., Alimoradzadeh R., Agah S., Mirmiranpour H., Khodabandehloo N., Middle East J. Dig. Dis., 2017, 9:228 [Google Scholar], [Publisher]
- Etemadi S., Mahmoodiyeh B., Rajabi S., Kamali A., Fard M.M., Romanian Soc. Cell Biol., 2021, 25:2417 [Google Scholar], [Publisher]
- Fard A.M.M., Fard M.M., Eurasian J. Sci. Tech., 2021, 1:284 [Crossref], [Google Scholar], [Publisher]
- Motaharian E.S., Mahmoodiyeh B., Lorestani S., Sadri M.S., Fard M.M., Fard A.M.M., Amini A., Chem. Rev., 2021, 3:171 [Crossref], [Google Scholar], [Publisher]
- Alimoradzadeh R., Mokhtare M., Agah S., J. Age., 2017, 12:78 [Google Scholar], [Publisher]
- Alimoradzadeh R., Mirmiranpour H., Hashemi P., Pezeshki S., Salehi S.S., Neurology Neurophys., 2019, 10:1 [Google Scholar], [Publisher]
- Rahmati J., Fathi H., Sultanova N., Davudov M.M., Danesh H.A., J. Otorhinolaryngol. Head Neck. Surg., 2020, 9:86 [Crossref], [Google Scholar], [Publisher]
- Rakei S., Rad H.I., Arbabisarjou A., Danesh H.A., Drug Invent. Today, 2019, 11:3123 [Google Scholar], [Publisher]
- Rakei S., Rad H.I., Irandegani F., Danesh H.A., Drug Invent. Today, 2019, 12:2809 [Google Scholar], [Publisher]
- Danesh H.A., Focus Med. Sci. J., 2018, 4 [Crossref], [Google Scholar], [Publisher]
- Danesh H.A., Saboury M., Sabzi A., Saboury M., Jafary M., Saboury S., J. Islam. Repub. Iran, 2015, 29:172 [Crossref], [Google Scholar], [Publisher]
- Hashemi S.M., Hashemi M., Bahari G., Khaledi A., Danesh H., Allahyari A., Asian Pacific J. cancer prevention: APJCP, 2020, 21:2479 [Crossref], [Google Scholar], [Publisher]
- Abdolrazaghnejad A., Banaie M., Safdari M., J. Emerg. Med., 2018, 2:e7 [Crossref], [Google Scholar], [Publisher]
- Akhlaghi N., Payandemehr P., Yaseri M., Akhlaghi A.A., Abdolrazaghnejad A., Emerg. Med., 2019, 73:462 [Crossref], [Google Scholar], [Publisher]
- Abdolrazaghnejad A., Banaie M., J. Pharma, 2017, 12:180 [Crossref], [Google Scholar], [Publisher]
- Pakniyat A., Qaribi M., Hezaveh D.R., Abdolrazaghnejad A., Acute Dis., 2018, 7:241 [Crossref], [Google Scholar], [Publisher]


