Document Type : Original Article
Authors
- Bahaa Fadhil Hamzah 1
- Israa Taha 2
- Zainab Mohsen Najm 3
- Mustafa D Husseini 4
- Noor S. K. Al-Khafaji 5
1 Muistry of Education, Education Directorate Babylon, Babylon, Iraq
2 Department of Radiology Technique, Al-Nisour University College, Baghdad, Iraq
3 Anesthesia Techniques Department, Al-Mustaqbal University College, Babylon, Iraq
4 Babylon Sewage Directorate, Babylon 51001, Iraq
5 University of Babylon, College of Science, Biology Department, Babylon, Iraq
Abstract
In this work, new 1,3-oxazepine derivatives were synthesized from a Schiff base. The latter was prepared by reacting 4-amino antipyrine with 4-amino acetophenone, then the Schiff base, which has an amine group, reacted with maleic anhydride to produce the corresponding amic acid. The prepared amic acid compound has an azomethine group utilized in synthesizing new derivatives of 1,3-oxazepine through reacting the amic acid with different cyclic anhydrides (succinic anhydride, maleic anhydride, phthalic anhydride, tetrachlorophthalic anhydride, and citraconic anhydride). Different techniques have been used to confirm the structures of synthesized compounds in terms of physical properties and spectroscopic measurements, where proton nuclear magnetic resonance 1H-NMR and Fourier Transform Infrared FT-IR spectroscopies have been used to confirm the synthesized compounds. In addition, the antibacterial activity of oxazepinederivatives was examined against two types of bacteria, gram-negative and gram-positive.
Graphical Abstract
Keywords
Main Subjects
Introduction
Oxazepine is an organic molecule with a seven-membered, unsaturated, and non-homologous ring that contains two heteroatoms [1]; oxygen and nitrogen. Several isomers can be attributed to oxazepine based on the locations of the nitrogen and oxygen atoms in its structure. 1,3-oxazepine, for example, is an isomer of oxazepane with the oxygen atom at the first position and the nitrogen atom at third the position [2, 3].
The synthesized oxazepine derivatives have been subjected to many studies for decades. These compounds are very useful in medicine since they have diverse biological functions [4-7]. Nitrogen heterocyclic compounds are particularly important since they represent a large class of natural and synthetic compounds, many of which have beneficial biological properties.
The ability to synthesize heterocyclic compounds might lead to the creation of new drugs. In 1965, an oxazepine derivative was approved for the treatment of treating psychoneuroses characterized by anxiety and tension [8-11]. Oxazepine molecules can be synthesized using a variety of ways. On the other hand, the reaction of imine groups with cyclic anhydrides, is a convenient and efficient technique for generating oxazepinerings [12-16]. In this work, the new derivatives compounds of oxazipine were synthesized with antipyrine moiety by the reaction of new prepared molecules, which possess an imine group with five different cyclic anhydrides.
Materials and Methods
All the chemicals were acquired from Sigma and CDH suppliers, which were used without further purification. However, the Gallen Kamp capillary melting point apparatus was used to determine prepared compounds’ the melting points which were not corrected. Moreover, Shimadzu FT-IR-8400 of Fourier Transform Infrared spectrophotometer was utilized to record the FTIR spectra, whereas Bruker 400 MHz of nuclear magnetic resonance was used to record 1H-NMR spectra of the prepared compounds.
Synthesis of Schiff base (1)
Schiff base was synthesized from the reaction of 4-amino antipyrine and p-amino acetophenone, in which 0.01 mol of 4-amino acetophenone was placed in a round bottom flask after dissolving it in an absolute ethanol solvent (25 mL) and followed by adding three drops of glacial acetic acid, the solution was heated with stirring after adding 0.01 mol of 4-amino antipyrine dissolved in ethanol in batches with stirring, and the mixture refluxed for two hours. The formed precipitate was filtered and washed with cold ethanol [17].
Synthesis of amic acid (2)
Amic acid was synthesized by the reaction of (0.001 moles) amino Schiff base (1) and (0.001 moles) maleic anhydride dissolved in acetone and stirring the mixture for 1.5 hours; the formed solid product was filtered and washed with acetone. Then recrystallization by dissolving in bicarbonate solution then precipitating with hydrochloric acid. After that, the filtered formed solid was washed with deionized water and dried [7].
Synthesis of 1,3-oxazepine (3-7)
Derivatives of oxazepine were synthesized by reacting the prepared Schiff base (2) with different cyclic anhydrides, namely, maleic anhydride, succinic anhydride, phthalic anhydride, tetrachlorophthalic anhydride, and citraconic anhydride). (0.01 mole) from each compound were dissolved in 30 mL of tetrahydrofuran THF and refluxed for 14-16 hrs. After that, the solvent was evaporated under a vacuum, and the precipitate was collected and recrystallized from chloroform [12]. The structures of the synthesized compounds of oxazepine derivatives were illustrated in Scheme 1, whereas the physical properties of the synthesized compounds were listed in Table 1.
Antibacterial measurements
The antibacterial activity was assessed using the Mueller-Hinton agar well diffusion technique, with working zones in millimeters (mm) filled with 100 μL of test samples and incubated at 37 °C for one day. The diameter of the inhibitory zones was measured using a ruler after incubation. The inhibition regions were obvious and surrounded the injection sites of the samples with no bacterial growth.
Results and Discussion
Infrared spectroscopic analysis (FT-IR)
The absorption values of the major groups contained in the produced compounds were revealed by studying the infrared spectra of the compounds, where the imine group appeared clearly in compound (1) at 1587 cm-1, and the stretching frequencies at 1653 cm-1 are due to C=O groups of antipyrine ring, and the stretching vibrations of amine group appear at 3336 and 3217 cm-1 [18]. The reaction of compound (1) with maleic anhydride leads to the appearance of new frequencies in the chart at 2500-3203 cm-1, 1715 cm-1, and 1678 cm-1 due to O-H carboxylic, C=O amide and C=O carboxylic group, respectively, in addition to the frequencies at 1654 cm-1 due to C=O of antipyrine ring.
Table 1: The physical properties of prepared compounds


Scheme 1: Synthesis processes of oxazepine derivatives
For compounds (3-7), the stretching band of the imine group disappeared, and new (lactam/lactone) groups of oxazepine appeared at 1788-1699 cm-1, beside other bands at 1678-1650 and 1718-1670 cm-1 due to C=O of amide and carboxylic acid group, respectively [19]. The main absorption FT-IR bands of the main functional groups of the prepared compounds were listed in Table 2.
Nuclear magnetic resonance 1H-NMR spectroscopic analysis
The DMSO-d6 solvent was used to dissolve the prepared compounds to record the 1H-NMR spectra. Table 3 contains the result of 1H-NMR chemical shifting in ppm for some compounds and polymers prepared.
Table 2: Infrared spectroscopic absorption bands for the prepared compounds
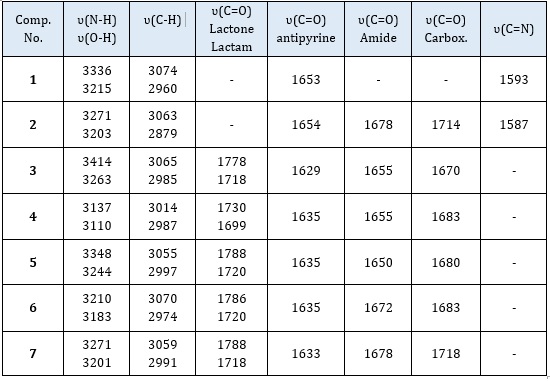
Table 3: 1H-NMR chemical shifting in (ppm)for the prepared compounds

Solubility of synthesized compounds
The prepared compounds (3-7) have good solubility in different organic solvents, as they dissolved in (ethanol, methanol, THF, DMSO and DMF); however, they were undissolved in (water, benzene, ether, and acetone).
Antibacterial activity of synthesized compounds
One of the wide spectra of applications that oxazepine compounds possess is the noticeable antibacterial activity. The antibacterial activity of synthesized oxazepane was tested by studying these activities against two types of bacteria: gram-negative (Escherichia coli) and gram-positive (Staphylococcus aureus), utilizing the well diffusion approach, which includes tracking the level of inhibition of these compounds by using the lowest inhibitor concentration. From the results in Table 4, the compounds (7) exhibited the best activity against gram-positive and gram-negative bacteria.
Table 4: Antibacterial activities for the prepared compounds (3-7)

Conclusion
New derivatives of 1,3-oxazepine compounds were successfully synthesized by many steps, including preparing a Schiff base from 4-aminoantipyrine and 4-amino acetophenone, which was used later to prepare the amic acid that possesses the imine group, which is necessary for synthesizing the targeted new compounds. The amic acid reacted with five different cyclic anhydrides to produce 1,3-oxazepine derivatives. The prepared compounds have been characterized using spectroscopic techniques such as FTIR and 1HNMR, whereas the physical properties have been determined for all the prepared compounds. While the prepared compounds have exhibited noticeable antibacterial activities, compound (7) showed the highest activity, which indicates promising biological applications.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Authors' contributions
All authors contributed to data analysis, drafting, and revising of the paper and agreed to be responsible for all the aspects of this work.
Conflict of Interest
There are no conflicts of interest in this study.
Orcid:
Mustafa D Husseini
https://www.orcid.org/0000-0002-1723-1855
HOW TO CITE THIS ARTICLE
Bahaa Fadhil Hamzah, Israa Taha, Zainab Mohsen Najm, Mustafa D Husseini, Noor S. K. Al-Khafaji. Synthesis, Characterization, and Antibacterial Activity of Some New Oxazepine Derivatives. J. Med. Chem. Sci., 2023, 6(6) 1239-1245
https://doi.org/10.26655/JMCHEMSCI.2023.6.4
URL: http://www.jmchemsci.com/article_160315.html


