Document Type : Original Article
Authors
Department of Chemistry, College of Education for Pure Science Ibn -Al-Haithem, University of Baghdad, Baghdad, Iraq
Abstract
In the present article, mixed ligand metal (II) complexes have been synthesized with Schiff base (1E, 5Z, 6E)-1,7 bis (4-hydroxy-3-methoxyphenyl)-5-(3-hydroxyphenyl) imino) hepta-1,6-dien-3-one derived from Curcumin and 3-aminophenol as primary ligand and L-dopa as a secondary ligand. The Schiff base act as bidentate and arrange to the metals through the azomethine (C=N) nitrogen and (C=O) oxygen atom. The mode of bonding of the Schiff base has been affirmed on the infrared by the UV-Visible, 1H, and 13C NMR spectroscopic techniques. The magnetic susceptibility and the UV-Vis data of the complexes propose octahedral geometry around the central metal ion. The information appears that the complexes have the structure of [L-M-(L-dopa)] system, where M=Mn(II), Ni(II), Cu(II), Zn(II) and Cd(II). The low conductance of all complexes supports the non-electrolytic nature of the complexes. The biological activity of the complexes was screened against bacteria, (Klebsiella and P. Staphylococuse and gives a good results anti-fungus C. Albicans). All complexes possess the biological activity which has the same activities, and good antimicrobial activity that displays higher activities against fungus compared with bacteria.
Graphical Abstract
Keywords
Introduction
Curcumin compounds have a great combination with drug or amino acids glycine-Curcumin form compounds, inhibits the growth of fungal and bacterial strains after complexation [1, 2]. The complexation behavior of di-ketones in metal complexes give information of the structural, nature of coordination, and spectral data [3, 4]. Schiff base complexes played a central role in bioinorganic chemistry because of their biological importance and industrial [5, 6]. O- and N- donors tridentate Schiff bases complexes have been reported to exhibit different activities on DPPH radicals [7]. Abu-Dief et al. synthesized and characterized Schiff bases-transition metal-complexes by the condensation of 5-bromosalicylaldehyde and six L-α-amino acids [8-10]. The theoretical study of curcumin-M = Nickel(II), Copper(II), and Magnesium(II) complexes performed by density-functional theory [11].
The most objective of this inquiry was to synthesize and characterize the Schiff base, and its mixed ligand metal(II) complexes with amino cid L-doba and, decide their antibacterial activity in the present work.
Materials and Methods
Materials and physicochemical analyses
All the chemicals were obtained from Sigma–or BDH and utilized without assistance purification. Curcumin, L-dopa, DMSO, and C2H5OH were Analar grade (BDH).
The FT-IR spectra were recorded in 400–400 cm–1 region with 8300-Shimadzu spectrophotometer, Magnetic susceptibility. Metal determination was done with spectrophotometer (A.A-160). The mass spectrum was recorded by MS Model: 5973 spectrometer, Elemental microanalysis C.H.N- 2400 elemental analyzer. The NMR spectra of proton and carbon were recorded for (L) in a solvent DMSO, by using a device (INOVA-500-MHz) in δ (ppm). The UV spectra (10−3 M) by (Shimadzu U.V 160-A), Λm 10−3 mol/L in DMSO were reached by Ino.Lab.720 digital conductivity.
Synthesis of Schiff base
The synthesis of Schiff base (L) was made concurring to the methods described in the literature.12 In 100 mL flask, the (L) was prepared by curcumin condensation, of which (3 mmol, 1.104 g) was dissolved in (25 mL) boiling ethanol. Then, NaOH (3 mmol, 0.119 g) was added and dissolved in (10 mL) of ethanol. After that, 3-aminophenol (3 mmol, 0.327 g) was dissolved in (30 mL) ethanol and added to the mixture with the constant stirring. The reaction mixture was refluxed for 12 hours at 70 °C until the completion of the reaction and the response and also, the advance of the response was checked by TLC. After the reaction was finished, the drops of glacial acetic acid were added (Scheme 1). The yellow-colored compound was precipitated. The yellow-colored accelerated compound was washed with hot ethanol, recrystallized to urge an unadulterated test, and dried desiccator in CaCl2.
Synthesis of mixed complexes of Schiff base and L-dopa
The mixed complexes (1-5) were synthesized by reacting the (L) and L-dopa with the Mn(II) (1), Ni(II) (2), Cu(II) (3), Zn(II) (4) and Cd(II) (5) ions according to the literature method [12], as depicted in Scheme 1.
Results and Discussion
Characterization of the Schiff base (L)
The yield of the prepared (L) was 87% (solid, color: orange, m.p. 295 °C). FT-IR (KBr, cm–1), as displayed in Figure S1 (Supporting information), 3242 ν (O-H), 1610 ν (C=N), 1701 v (C=O), 1589 ν (C=C), 1029 ν (C-O-C), and 1126 ν (OCH3) [13-16]. UV-Vis, 10-3 mol L–1 in DMSO, λmax nm: 284 and 364 were assigned to (π→π*) and (n→π*) transitions, respectively.
1H-NMR (DMSO, 400 MHz), in Figure S2 (Supporting information), 8.8- 8.65 ppm δ (s, 1H, OH) phenol, displays 8.8-8.65 ppm δ (OH) phenol, a multiple in the region δ (m, 7H, ArH). 6.41-7.35 ppm was corresponded to the aromatic protons other than the over peaks, the chemical move values of δ (-CH=CH-) aliphatic 4.5, 5.3, 3.45 (H-CH2), and 3.7 (H-OCH3), as illustrated in Figures S3 and 4 (Supporting information) [17, 18]. The 13C-NMR spectrum of (L) is indicated in Figure S3 (Supporting information). The 13C-NMR peaks for the azomethine carbon atom and phenolic group carbons of the coordinated Schiff base ligand were observed at 𝛿 =166.5 ppm and 158.84 ppm, respectively [13, 17].
The mass spectrum of (L), in Figure S4 (Supporting information), showed the parent ion peak at M/Z [C27H25NO6] = 459 (M + which is steady with the atomic weight of the Schiff base. The other fragments, their relative abundances, and fragmentation pathways data are depicted in Scheme 2 and strongly confirmed the formation and the structure of the Schiff base.


Characterization of [L- Metal-L-doba] Complexes
All the complexes were destined reacting the metal chlorides MCl2.nH2O and the ligands by utilizing 1:2:1 mole ratio, i.e. metal: two moles of L-dopa K: Schiff base. The synthesis of [M(L)(L-dopa)2] complexes may be represented as shown in the following overall equation:
2( L-dopa) + 2KOH → 2( L-dopa K) + 2H2O
MCl2.nH2O+L+2(L-dopaK)→[M(L)(L-dopa)2]+2KCl+nH2O Where, L=Schiff base (C27H25NO6) accrued from condensation of (Curc) and 3-aminophenol as a primary ligand, L-dopa as a secondary ligand.
L-= deprotonated of (L-dopa)
As listed in Table 1, the melting points (m.p) of the complexes were higher than that of the ligands, demonstrating that the complexes are steadier than the ligands thermally. The solubility of the complexes was examined in different solvents appeared that all complexes are dissolvable in all solvents. The test for chloride particle with AgNO3 arrangement was negative (Nil%) demonstrating that there is no (Cl-) outside to the coordination circle of the central metal. The calculated and exploratory values of metal percentage (M%) in each complex are in sensible agreement [17]. The formula weights, elemental analysis of CH.N data, and melting points are presented in Table 1. The molar conductance Λm values for the complexes were found between 2.10 and 19.00 (Ω–1 cm2.mol–1) in DMSO indicating their non-electrolytic nature [18].
FT-IR spectra
The important FT-IR data of all complexes are presented in Table 2. The FT-IR spectra of the complexes, are similar and there are no major differences in their vibrational frequencies. Generally, as compared FT-IR spectra of the complexes with those of free L-dopa and (L), we observe shift of the most of bands to the lower or higher frequencies. It is probably caused by coordination of two ligands [11-13].

The blue and red shifts of observed bands are due to the formation of M- ν (C=N-) and ν (C=O) coordination bonds. The shifts in complexes suggest that the bond formation of the metal. The oxygen of the ketone part binds metal cations [11]. De-protonation of L-dopa is neighborhood in nature, for all locales considered the changes in geometry and charge thickness are localized within the locale where the deprotonation took place. In any case, the impact of carboxyl gather deprotonation is transmitted to the C–NH2 locale and leads to the collection of the abundance charge thickness on N iota in NH2 group. This point is of extraordinary significance when concerned. The spectra of the complexes displayed two different absorption bands in the 1627 and 1427 cm−1, which correspond to the difference between the ν asym (COO) and ν sym (COO) vibrational modes of the carboxyl groups, distinction Δν = [ν asym(COO−) – ν sym (COO−)] > 200 cm-1 falls in the range of 274–262 cm-1. This results for carboxyl group associated with monodentate manner coordination [14-16] with the metal ions. L-dopa acts as bidentate ligand coordination by the carboxyl oxygen COO → and the ← NH2 nitrogen atom of amino group with the active donor atoms. Inclusion t of nitrogen in the coordination was backed by the appearance of the groups comparing ν (C=N) and ν (M-N) in 1512–1597 and 516–582 cm-1, which confirms coordination through the ν (C=N-) nitrogen to the M(II) moiety. The bands at 516–595 cm-1 were relegated to ν (M←N). The new low intensity bands in the region of (432–482 cm-1) are attributed to ν (M–O).

Electronic spectra and magnetic moment
All the UV-Vis spectra of the mixed L-dopa-Schiff base metal(II) complexes indicated the similar absorption spectra as the Schiff base which is either moved to the red or blue locale, as listed in Table 3. The (UV-Vis) spectrum for the (L-dopa) in DMSO (10−3 M) was characterized by 2-absorption bands at (λmax 277 nm, π ®π*) and (λmax 451 nm, n ®π*), within the transition aromatic ring and C=O, respectively, agreed with the reported data [18, 19]. The observed magnetic value of Ni(II) complex was 3.19 μB, Cu(II) complex was 1.69 μB, Mn(II) complex was 5.94 μB which revealed an octahedral geometry [19], around the central metal ions. Zn(II) and Cd(II) d10 electronic configuration complexes are diamagnetic nature [20]. The electronic spectrum of the [Mn (L)(L-dopa)2] complex gave three bands at 285 nm and 364 nm were due to the intra-ligand transition, charge transfer, and the one at band at 864 nm was were due to 6A1g→ 4Eg transition [20, 21]. The electronic spectrum of [Ni (L)(L-dopa)2] complex given three bands at 284 nm and 366 nm are due to the intra-ligand transition, and charge transfer, and the one at 910 nm was as a result of [d-d] transition for 3A2g → 3T2g [21].
[Cu(L)(L-dopa)2] complex spectrum gave four bands. The bands at 287 nm and 362 nm are due to the intra-ligand transition and C.T. and two at 826 nm and 910 were as a result of [d-d] transitions for 3T1g →3T2g [20].
[Zn(L)(L-dopa)2] complex gave bands at 266 nm (37593 cm-1) and 429 (23310 cm-1) nm were due to (intra-ligand transition) and metal to ligand charge transfer (MLCT) transition, respectively [20].
[Cd(L)(L-dopa)2] complex gave bands at 285 nm (35087 cm-1), 345 nm (28985 cm-1), and 363 nm (27548 cm-1) were gave (intra-ligand transition) and (MLCT) transition, respectively [18].
An octahedral (goodness) geometry are proposed for all complexes based on its unearthly information and explanatory conductance, which displayed coordination number of six and may be defined as [M(L)(L-dopa)2], as demonstrated in Figure 1 [9, 11].
The Proposed molecular structure for studying complexes
Studying complexes on bases of the above analysis, the spectral observations suggesting the octahedral geometry for all the prepared complexes which exhibited coordination number six and may be formulated as [M(L)(L-dopa)2]. The general structure of the complexes is 3D, as is depicted in Figure 1.


Biological activities
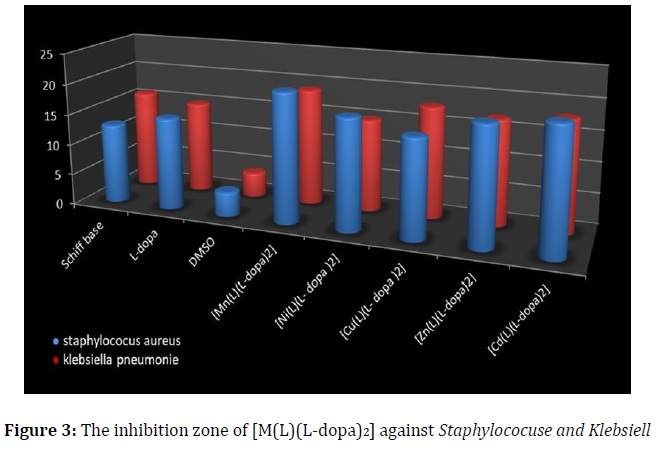
The tests were carried out on one Gram-positive (+) and one Gram-negative (-) bacterial strain concurring to the standard method [22]. All complexes are similar and there are no major differences in their biological activity. The results were represented by using bar graphs in Figures 2 and 3. The inhibition zones were measured in millimeters (mm); high activity 11-15, very high activity ≥16, and low activity for DMSO.
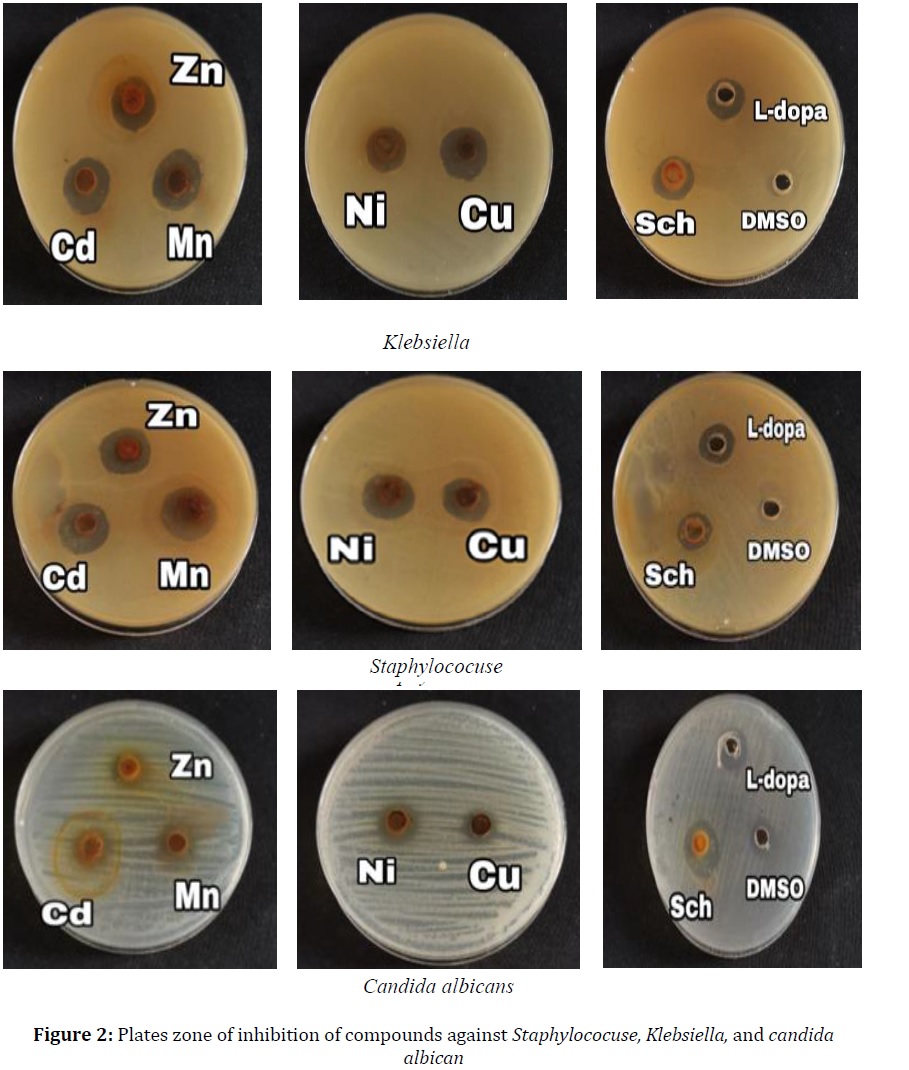
All the compounds showed a significant antibacterial activity against the tested bacteria, the ligands were found to be active against the bacterial strains and some complexes were more significant than the two ligands [11, 23].
The compounds were assessed on one strain of finding that all the test complexes initiated antifungal movement against the candida species. Figure 4 displays a really comparable antifungal movement by all the test compounds, there are numerous components increment the movement are bond length among the metal, conductivity, dipole minute, and dissolvability [22-25].

Conclusion
In this study, a Schiff base ligand (Curcumin with 3-aminophenol) was synthesized. The stable mixed ligand complexes, [L–M-(L-dopa)] system was formed with transition metal ions such as Mn(II), Cu(II), Ni(II), Zn(II), Cd(II), and chlorid, and then they were synthesized. The activity of the synthesized mixed Schiff curcumin-L-dopa complexes matches the effectiveness of some anti-microbial strain.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Authors' contributions
All authors contributed to data analysis, drafting, and revising of the paper and agreed to be responsible for all the aspects of this work.
Conflict of Interest
The author declared that they have no conflict of interest.
ORCID
Taghreed Al-Noor
https://www.orcid.org/0000-0002-6761-7131
Supporting Information
Our work includes the synthesis of new compounds, which is consistent with the goals of the journal and adds new scientific parameters to the researchers (PDF).
HOW TO CITE THIS ARTICLE
Hala Mohammed Salh, Taghreed H Al-Noor. Synthesis and Characterization with Antimicrobial Studies of mixed Curcumin Schiff Base –L-dopa with Divalent Cations Complexes. J. Med. Chem. Sci., 2023, 6(7) 1621-1631


