Document Type : Original Article
Authors
1 Department of Chemistry, College of Education for Pure Science, University of Mosul, Mosul, Iraq
2 Manager of Mosul Oncology and Nuclear Medicine Hospital, Nineveh Directorate of Health, Ministry of Health, Mosul, Iraq
Abstract
A molar pregnancy is the result of an abnormally fertilized egg or placental tissue expansion. The goal of this research was to investigate the potential linkage between some biochemical tests, antioxidant enzymes, oxidative stress, and fatty acid compositions in patients with a molar pregnancy compared with healthy pregnancy subjects. An observational case-control study was conducted in Mosul, Iraq. In this study, 141 women participated, and they were split into three group: 45 patients with molar pregnancy, 47 women with healthy pregnancy, and 49 healthy control women. Antioxidant enzymes, lipid peroxidation, hCG, ALP, trace elements, and fatty acid compositions were all investigated. The results revealed that MAD and hCG levels were increased in molar pregnancy compared with healthy pregnancy, while paraoxonase and GSH levels were decrease in molar pregnancy women than in healthy pregnant women. Likewise, the results showed that the increase of saturated fatty acids in molar pregnancy women was greater than in healthy pregnancy. This study found a significant rise in oxidative stress and poor monounsaturated and polyunsaturated fatty acid status in molar pregnancy as well as increased saturated fatty acids.
Graphical Abstract
Keywords
Main Subjects
Introduction
Molar pregnancy (MP) is a hereditary disease caused by improper fertilization. It is marked by abnormal placental villi swelling and severe trophoblastic hyperplasia [1]. The most common form of gestational trophoblastic illness is it (GTD) and can proceed to gestational trophoblastic neoplasia (GTN), a malignant disorder [2]. Based on clinical, morphological, and genetic criteria, HM is divided into two types: partial HM and complete HM (CHM) [1]. MP's etiology is yet unknown. However, because it causes deoxyribonucleic acid damage and faulty placentation, oxidative stress (OS) may play a key role in its pathophysiology [3]. Patients with CMP had higher OS marker concentrations and reduced antioxidant defense element, as compared with healthy pregnant and non-pregnant women [4].
In living organisms, the oxidative stress is described as an imbalance between oxidant substances (reactive oxygen species) and antioxidant defenses. Various pathologic and physiologic situations, including pregnancy and its consequences are linked to oxidative stress [5]. Cells have evolved antioxidant systems to limit the generation of reactive oxygen species (ROS), inactivate them, and repair cell damage. Women's reproductive health is affected by OS throughout their lives, even after they have stopped having children. OS may play a role in the age-related decline in fecundity, according to some researchers [6, 7]. Pregnancy, healthy parturition, and the commencement of premature labor are all influenced by it [8, 9]. Lipid breakdown, protein synthesis inhibition, and ATP depletion are only a few of the processes that have harmful consequences [10, 11]. Various physiological processes (including oocyte maturation, ovarian steroidogenesis, ovulation, implantation, blastocyst development, luteolysis, and luteal maintenance in pregnancy) are known to be impacted by ROS [12].
During a Molar pregnancy, the oxidative stress is known to rise. Blood lipid hydroperoxide levels are higher and overall antioxidant capacity is lower in healthy pregnancies [13], although erythrocyte glutathione peroxidase activity and its cofactor selenium are lower [13, 14]. However, this mechanism's nature remains unknown. As a result, the purpose of this study is to assess oxidative stress in patients with molar pregnancy, and as we know, no study has looked at the oxidative/antioxidative status of plasma in the pseudopregnant condition of CMP. This research aims to contribute to that body of knowledge.
Materials and Methods
Patients
The participants in this study were 141 women who attended oncology and Nuclear medicine hospital between June 2020 and August 2021. There were 49 healthy non-pregnant women and 47 with healthy normal pregnant women (controls) in their third trimester with a single viable fetus (mean gestational age 12.8 weeks as estimated by ultrasonography). MP was performed on the remaining 45 individuals (the mean gestational age 13.2 weeks as estimated by last menstrual period). Obese, underweight, and smokers were not included in the study. A histopathological examination of molar tissue revealed abnormal trophoblastic tissue proliferation, the absence of an identifiable foetus, chorionic villi with generalized hydatidiform swelling, and diffuse trophoblastic hyperplasia caused by the abnormal fertilization, leading to the diagnosis of molar pregnancy. All of the participants signed formal consent papers stating that they were aware of the situation.
Sampling
Molar pregnancy, healthy pregnancy, and the healthy control group each gave 10 mL of venous blood for the research. The serum was then retained in plane tubes and centrifuged for 5 minutes at 3000 rpm to separate it, before zbeing frozen at -20 °C.
Biochemical analysis
The HCG levels were measured in the blood serum of all subjects using a kite applied by Diagnostic Automation/Cortez Diagnostics Inc (USA). Serum paraoxonase (PON) activity was quantified spectrophotometrically in a microplate format using unique enzyme phenol acetate, according to Richter et al. techniques [15]. The biochemical parameter of liver alkaline phosphatase (ALP) was examined by conventional procedures utilizing an automated COBAS INTEGRA 800VR (Roche Diagnostics GmbH, Mannheim, Germany) (USA). Magnesium (Mg) and Zinc (Zn) were measured using the ZEEnit 700 atomic absorption spectrometer (Analytik Jena, Germany). For measurements, flame atomization was applied and a Deuterium lamp was used as a background correction. The resonance lines of hollow cathode lamps were used as research elements (HCL, Analytik Jena, Germany). Acetylene (99.5%, Air Liquide, Germany) and nitrous oxide (99.5%, Air Liquide, Germany) were utilized as the fuel and oxidant gases, respectively.
Estimation of lipid peroxidation and antioxidant levels
Malondialdehyde concentrations have been measured using thiobarbituric acid reactions (MDA) by the Halliwell and Chirico technique (1993), and the GSH level was measured using the Mohammed and Kakey method (2020).
Analysis and identification of fatty acids
Before being extracted using the Folch procedure (chloroform: methanol, 2:1, v/v), the serum was refrozen for 30 minutes at room temperature. 100 μL aliquots of serum were mixed with 10 μL of internal standard and 1 μL of Folch reagent. To measure recovery, one or two minutes of vortexing and one or two extractions were performed. The standard fatty acid combination was spiked into serum in amounts of 10, 25, and 50 μL for all of the recovery experiments. Layers were separated by centrifuging samples at 2400 g for 5 minutes after vortexing samples with Folch reagent. The sample was dried in a Speed-vac at 37 °C after the coating of chloroform was removed, with care given to avoid the interface containing precipitated proteins. Various reagents can then be used to accomplish transesterification of fatty acid methyl esters. MethPrep II is a one-step reaction that may be conducted at room temperature, requires no extraction prior to analysis, and is faster than using sodium methoxide reagents. According to early research, longer-chain fatty acids like eicosapentanoic acid (EPA) and docsahexanoic acid (DHA), both of which elute late in the chromatogram, showed enhanced peak shape after derivatization using MethPrep II in a combination of hexane and chloroform (data not shown). Since n-3 fatty acids are not heat stable, the Meth-Prep II reagent (methanolic m-trifluoromethylphenyltrimethylammonium hydroxide) was utilized at room temperature. The Folch extractant was prepared by combining 90 μL of chloroform: hexane (1:1) with 10 μL of Meth-Prep II to derivatize dried lipid samples or standards. Prior to being put into autosampler vials, samples were sealed, briefly vortexed, and given 20 minutes to react at room temperature. The derivatized samples were stable at room temperature for more than a week. Supelco SP2330 column with film thickness of 30 m X 0.32 mm X 0.2 m was the one that was utilized (Cat. No.24073, Sigma-Aldrich, St. Louis, MO). GC and autosampler models HP 7673 were used for gas chromatography. Helium was used as the carrier gas, with a column head pressure of 10 psi. The split vent had a total flow rate of 50 mL/min, 2.5 mL/min via the column, and 2.5 mL/min by the septum purge.
One-microliter injections were done using the splitless injection mode while the injector was heated to 220 °C. A linear temperature rise of 11 °C/min from 70 °C to 170 °C, a slower linear increase of 0.8 °C/min to 175°C to separate closely eluting fatty acids, and a final 2.5 minute hold were all performed. From beginning to end, the race took 20.1 minutes. At 250 °C, an HP 5971 mass spectral detector operating in single ion monitoring mode was employed.
Statistical analysis
To draw up cross-tabs and make relevant conclusions, data was acquired, processed, and statistically analyzed using IBM SPSS statistical® 26.0 software. The mean standard deviation was used to describe the outcomes of continuous variables. The observed data was tabulated, and variable groups were evaluated using an independent t-test. When the p(t-test) value was less than 0.05, it was considered as significant, and when it was greater than 0.05, it was considered as non-significant.
Results and Discussion
The results of MDA, GSH, HCG, and PON among molar pregnancy, normal pregnancy, and Healthy controls are summarized in Table 1. The results showed that MDA and HCG levels in the Molar pregnancy were higher (2.195±0.626, 260.835±64.254), respectively, than that of healthy pregnancy group, (0.697±0.387; 64.549±11.110), respectively, compared with healthy controls group, whilst the GSH and PON, results in the molar pregnancy group was showed significantly lower (0.630±0.341, 168.841±39.010), respectively, than that of normal pregnancy group (3.163±0.801, 428.621±25.250), respectively, compared with the group of healthy controls (Table 1).
The findings indicated that the considerably higher of the outcomes by age groups of MDA, GSH, HCG, and PON in healthy pregnancy with age group 24-32 years old (0.703±0.392; 3.205±0.860; 68.065±7.800; 435.481±26.562), respectively, than the age group 33-47 years (0.693±0.390; 3.128±0.765; 62.289±12.405; 422.860± 23.060), respectively. With molar pregnancy, the results revealed that the MAD, GSH, and HCG were significantly lower in age group of 24-32 years old (2.129±0.541; 0.616±0.244; 281.176±78.119), respectively, than 33-47 years old (2.237±0.682; 0.639±0.395; 245.188±46.988), respectively. While the PON was higher in age group of 24-32 years (185.450±44.228) than 33-47 years old (158.164±31.663) (Table 1).
Furthermore, the statistical significance of MDA, GSH, HCG, and PON in serum of among groups under study (molar study and normal pregnancy) and the group of healthy controls were consistent with clinical significances and demonstrated clinical significance at (P <0.05) (Table 2).


Table 3 indicates the results APL, Mg, and Zn among molar pregnancy, healthy pregnancy, and healthy controls. The results showed that ALP and Zn levels in the healthy pregnancy were higher (0.334±0.180; 170.260±10.164), respectively, than that of molar pregnancy group (0.311±0.216; 118.104±6.274), respectively, compared with healthy controls group, whilst the Mg, results in the healthy pregnancy group was showed lower (2.217±0.482) than that of Molar pregnancy group (2.522±0.212) compared with the group of healthy controls (Table 1).
The findings indicated that the considerably higher of the outcomes by age groups of ALP, Mg, and Zn in healthy pregnancy with age group of 24-32 years old (0.361±0.175; 2.277±0.549; 170.944± 10.281), respectively, than the age group of 33-47 years old (0.334±0.180, 2.217±0.482, 170.260 ±10.164), respectively. With molar pregnancy, the results revealed that the ALP, and Mg were significantly higher in age group of 24-32 years old (0.289±0.198;2. 520±0.225), respectively, than 33-47 years old (0.289±0.198; 2.520±0.225), respectively. While the Zn results were lower in molar pregnancy with age group of 24-32 years old (117.902±6.356), respectively, than the age group of 33-47 years old (118.274±6.331), respectively (Table 3).
Furthermore, the statistical significance of ALP, Mg, and Zn in serum of among groups under study (molar study and healthy pregnancy) and the healthy control group were in conformity with the clinical significances and demonstrated clinical significances at (P < 0.05) (Table 4).

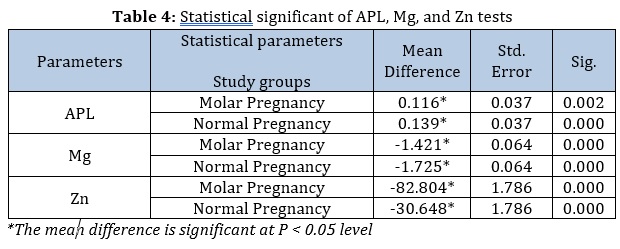
Table 5 reported the results of the composition of the serum fatty acids. The results showed the major fatty acids of statured FA were Butyric acid, stearic acid, Undecylic acid (94.564±2.1709, 1.1145±0.00303, and 0.0445±0.00302), respectively higher in molar pregnancy than healthy pregnancy (0.0605±0.0767, 0.145±0.3028, and 0.02545±0.0003), respectively, while the fatty acids of saturated of FA were palmitic acid, myristic acid, and lauric acid lower in molar pregnancy (0.1242±0.00301, 0.03356±0.0511, and 0.0845±0.00301), respectively than the healthy pregnancy (0.345±0.03027, 0.1925±0.00716, and 0.145±0.03028), respectively, as presented in Table 5. With mono unsaturated fatty acids, the results showed that the major FA of mono unsaturated FA were Palmitoleic acid, Gondoic acid, Nervonic acid, Erucic acid, and oleic acid higher in molar pregnancy (0.2645±0.00302, 0.1445±0.00302, 0.0945±0.00302, 0.0745±0.00302, and 0.0245±0.00302), respectively, than that in healthy pregnancy (0.01245±0.0003, 0.045±0.03027, 0.0545±0.0303, 0.02145±0.0003, and 0.01245±0.0003), respectively, as reported in Table 5. Moreover, the results showed the composition of poly unsaturated FA in serum with molar pregnancy compared with the healthy pregnancy that the major FA of poly-unsaturated FA were Linoleic acid in molar pregnancy (0.1949±0.00314) higher than healthy pregnancy (0.01745±0.0003), while the FA of poly-unsaturated were α-Linolenic acid and Eicosapentaenoic acid lower in molar pregnancy (0.01445±0.0003 and 0.0145±0.00302), respectively, than healthy pregnancy (0.05315±0.00236 and 0.0945±0.00302), respectively (Table 5).

A molar pregnancy (MP) is a rare lump or growth that appears at the beginning of a pregnancy inside the uterus (womb). It is a trophoblastic condition that affects expectant mothers (GTD) as a result of improper oocyte fertilization (egg). It leads to a fetus that is abnormal. The implantation and development of fertilized ovum outside the uterine cavity occurs with around 2% of all pregnancies. The ampullary section is the most prevalent place for ectopic pregnancies, accounting for almost 95 percent of all ectopic pregnancies [16]. According to several studies from developed countries, the prevalence of ectopic pregnancies has increased in the last 30 years as a result of an increase in the incidence of sexually transmitted infections, tubal surgery, and more frequent use of ovulation induction and assisted reproductive technologies [17]. The serum hCG levels in a molar pregnancy were found to be significantly higher than those in a normal at the same gestational age, which is consistent with another study that found serum Liver function tests and hCG levels to be significantly higher in a molar pregnancy than healthy pregnancy compared to a healthy controls at the same gestational age [18]. Since of the association between hCG levels and trophoblastic tumor burden, human chorionic gonadotropin (hCG) is employed as a marker in postmolar surveillance, enabling for early detection and treatment of GTN [19]. The development of GTN has been linked to abnormal hCG regression patterns [20]. In our study, we reported that the namely increased oxidative stress was present in patients with molar pregnancy more than with healthy pregnant, as compared with a healthy control subjects. The higher levels of oxidative stress and lower levels of antioxidants have previously been identified in women with MP, indicating that OS may have a role in the disease's pathogenesis [4]. The elevated oxidative stress may cause placental vasculature to change, resulting in early miscarriage. Diabetic patients and spontaneous miscarriage have higher glutathione peroxidase levels but lower selenium levels, indicating a relationship between inadequate antioxidant defenses and fetal mortality [21].
The reduced glutathione functions as a free radical scavenger and maintains the placenta's functional store of antioxidants that contain sulfhydril groups (SH). Antioxidants with the SH structure are essential for the onset of preterm labor which starts before 37 weeks of pregnancy. Glutathione peroxidase and glutathione reductase levels in the placenta were shown to be greater in a prospective study of individuals with preterm labor. These levels were compared with those seen in healthy patients who had never had a preterm birth. This antioxidant system showed a statistically significant increase, implying that such patients are in a condition of elevated demand to counteract the consequences of oxidative stress [22].
Recurrent abortion patients showed greater glutathione lipid peroxides and lower vitamin E levels in their blood [23]. Plasma glutathione levels were considerably low in pregnant individuals with a history of recurrent pregnancy loss [24]. Increased lipid peroxidation has also been linked to the pathogenesis of antiphospholipid syndrome-related recurrent pregnancy loss. In individuals with antiphospholipid syndrome, antioxidant treatment with vitamins C and E resulted in lower levels of anticardiolipin antibodies. Magnesium is the body's fourth most abundant mineral and a cofactor in over 325 enzymes. As a result, several disorders such as hypertension, cardiac arrhythmias, and dyslipidemia are linked to Mg deficiency. Magnesium shortage contributes to the formation of oxidative stress by acting as an antioxidant, acting as a cofactor in various enzymes, maintaining cell membrane integrity, and reducing the consequences of oxidative stress [25]. The zinc depletion is a dangerous condition that causes delayed wound healing, immune system malfunction, anorexia, poor protein synthesis, night blindness, skin problems, hair loss, and growth retardation, among other symptoms. The blood zinc concentration is often used to measure zinc status. However, the serum zinc concentration is reduced not just in true zinc shortage, but also in stress. More than 200 enzymes have zinc as a component. Zinc-containing metallo-enzymes frequently do not operate properly in the presence of zinc shortage. One of these zinc-dependent metallo-enzymes is alkaline phosphatase (ALP). The ALP activity in the blood can be used to estimate zinc levels, however ALP activity is impacted by a variety of other variables [26]. Mg and Zn are key trace elements that induce a reduction in ALP activity and should be supplemented with these minerals. In a research by Ray et al., it was discovered that low ALP patients had lower Zn and Mg levels than controls. In addition, a verified positive association between Mg and Zn and ALP was discovered [27].
In this study, the fatty acid content of serum molar pregnancy was compared with that of a healthy pregnancy. As compared with a healthy pregnancy, the levels of saturated fatty acids in Molar pregnancy were significantly higher. Several maternal variables have already been demonstrated to influence fatty acid levels, including gestational age, socioeconomic position, education, smoking, and supplement usage [28]. In research conducted in Western countries, alterations in fatty acid content were also identified [28, 29]. Enhancing Stearoyl CoA Desaturase (SCD) activity may help with stored fat, while inhibiting Elongase 6 activity may help with health promotion by lowering the negative effects of insulin resistance [30]. In conclusion, this research presented a striking increase in the oxidative stress, low monounsaturated, and polyunsaturated fatty acid status in molar pregnancy with increased saturated fatty acids than in healthy pregnancy compared with healthy control subjects.
Conclusion
In conclusion, this research presented a conspicuous increase in free radicals damage and depressed level for both monounsaturated and polyunsaturated fatty acid status in molar pregnancy with increase in the saturated fatty acids more than that in healthy pregnancy in compare with those of healthy control.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Authors' contributions
All authors contributed to data analysis, drafting, and revising of the paper and agreed to be responsible for all the aspects of this work.
Conflict of Interest
The author declared that they have no conflict of interest.
ORCID
Luma Abd Almunim baker
https://orcid.org/0000-0003-3611-7991
HOW TO CITE THIS ARTICLE
Luma Abd Almunim baker, Shaymaa Z. Jalal Aldin, Abduqader Salim Ahmed. Role of Oxidative Stress in Molar Pregnancy. J. Med. Chem. Sci., 2023, 6(6) 1383-1393


