Document Type : Original Article
Authors
- Eman H. Al-Rikabi 1
- Rana A. K. Al-Refai 1
- Sadiq J. Baqir 1
- Angham G. Hadi 1
- Ahmed Kareem Al-Qayyim 2
1 Department of Chemistry, College of Science, University of Babylon, Babylon 51002, Iraq
2 Medical Laboratories Techniques Department, Al‐Mustaqbal University College, Babylon, Iraq
Abstract
Organotin carboxylates have gotten a lot of attention in recent years because of their structural intrigue and several uses. The production and characterization of organotin carboxylates, as well as their action against cancers, fungi, bacteria, and other microbes have been described by a number of researchers. Some di- and triorganotin carboxylates have been found to have the potential as anticancer drugs. The amount and kind of organic groups bound to the tin core and carboxylate ligand appear to have a significant impact on their anticancer efficacy. Two new organotin complexes of di and triorganotin carboxylate were successfully synthesized by refluxing reaction of 2-[(2,3-dimethylphenyl) amino] benzoic acid (ligand) with tri phenyl tin chloride and dimethyl tin dichloride salts to give the corresponding substituted tin complexes with high yields. The chemical structures of the complexes were confirmed by different techniques included elemental analysis, proton, carbon and Sn119-NMR, and FT-IR-spectra. The activity of each complex has been examined against the target cell line A-172 compared with the ligand alone. It was found that the complex 1 and 2 have higher cellular cytotoxicity than ligand.
Graphical Abstract
Keywords
Main Subjects
Introduction
Cisplatin has been widely used in cancer treatment, and it was the first inorganic compound used since 1978. Cisplatin had a great effect in the treatment of cancer of the bladder, ovary, esophagus, breast, stomach, and others [1-3]. After that, unfortunately, it was found that the cisplatin use is not safe because of the appearance of side effects and several defects such as neurotoxicity, nephrotoxicity, and ototoxicity for patients who received treatment [4, 5]. Subsequently, platinum derivatives such as carboplatin and oxaliplatin were used. Until the present time, platinum and its derivatives are as an antitumor treatment. Despite the emergence of metal compounds derived from the elements tin, gold, palladium, and copper where these compounds showed cytotoxic properties [6-8]. Tin derivatives have shown significant efficacy when used as a drug for cancer treatment, especially organotin compounds, as the use of their low doses has better or similar potential to the approved and used drugs [9, 10].
The effectiveness of using organic tin compounds depends on the type of bond, the type, and number of organic groups associated with the central atom of tin [11,12]. The genetic changes and the accumulation of mutations led to the formation of a malignant tumor, and this led to attracting many to search for antitumor drugs that are metallic and non-platinum based, such as binary and triple derivatives of organic tin, which showed the strong effects against the proliferation of cancer cells [13-19].
Dicarboxylate and organotin tricarboxylates showed anticancer activity in solid and solution phase [20, 21]. Studies have demonstrated that tertiary organotin compounds are more biologically active than their mono- and binary counterparts due to their ability to bind with proteins depending on the number of coordination and the nature of the groups attached to the tin atom [22-24]. In general, the coordination structure of organotin compounds is of great importance for the antitumor activity [25-30]. The subsequent studies reported that the compounds of di-organotin carboxylate have more antitumor activity in vivo than cis platinum [31, 32]. Likewise, alkyl or aryl tin derivatives are more active when R is phenyl also, the di-butyltin derivatives are more active than their tri-butyltin counterparts [33].
In this study, by refluxing the reaction of 2-[(2,3-dimethylphenyl) amino] benzoic acid (ligand) with tri phenyl tin chloride and dimethyl tin dichloride salts, it was possible to successfully synthesize two new organotin complexes of di- and triorganotin carboxylate, which led to the high yields of the corresponding substituted tin complexes.
Materials and Methods
Synthesis of triphenyltin Tin carboxylate complex 1
A reaction of a 1:1 mixture (M: L) was obtained by dissolving of (0.48 g, 2 mmol) 2-[(2,3-dimethylphenyl) amino] benzoic acid in 20 ml methanol, and then the equivalent moles of NaOH (0.08, 2 mmol) was added with stirring for 30 min. A (0.84 g, 2 mmol) triphenyltin chloride in boiling (20 mL MeOH) was added to the first solution and it was allowed to reflex for 8 hours, left to evaporate, washed with diethyl ether, and collected to provide complex 1 with 75% yield (Scheme 1) [34-37].

Synthesis of dimethyl Tin complex 2
Synthesis of dimethyl tin complex 2 is carried out according to Kovala and Demertzi et al. [38]. (0.96 g, 6 mmol) of 2-[(2,3-dimethylphenyl) amino] benzoic acid in methanol (30 mL) and (6 mmol) NaOH were stirred for 30 minutes at room temperature. A solution of dimethyl tin dichloride (3 mmol, 0.66 g) with 20 ml of boiling methanol was added to the first mixture. The mixture was allowed to reflux for 8 hours while being constantly stirred. The white precipitated was evaporated under vacuum. The product was washed with diethyl ether and collected to provide complex 2 with 70% yield (Scheme 2) [39].

Results and Discussion
The physical properties of 2-[(2,3-dimethylphenyl) amino] benzoic acid and the prepared complexes were listed in Table 1.
FT-IR spectroscopy of complexes 1 and 2
The FT-IR spectroscopy is able to grasp the vibrations of carbonyl groups in the range where stretching vibration modes appear 3500–1400 cm−1. In complexes 1 and 2, a new peak was appeared in 520-535 cm-1 and 440-450 cm-1 region related to tin-carbon and tin-oxygen groups. Carbonyl group vibrations were appeared clearly at (1685-1697 cm-1). Likewise, the broad peak of hydroxyl of carboxyl group was disappeared due to the complexation. Figure 1 and Table 2 indicate the important FT-IR spectrum numbers of complexes 1 and 2.
NMR spectroscopy of complexes 1 and 2
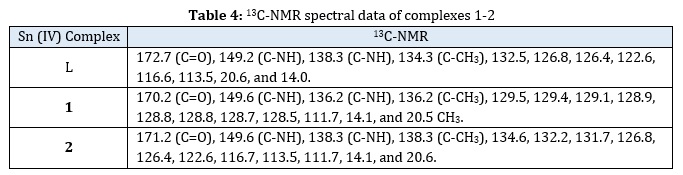
The 1H-NMR spectra showed a singlet signal at the 9.45–9.77 ppm region that was related to the NH proton (Figure 2 and Table 3). The aromatic protons were appeared with multiple signals at 7.90–6.67 ppm region. The protons of methyl-tin atom of complex 2 were appeared as a singlet in 0.78 ppm of a high field due to the shielding effect. Also, multiple signals are visible in the 13C-NMR spectra of 1 and 2 inside the aromatic area (Figure 3 and Table 4).




Cytotoxic activity
To demonstrate the binding efficiencies of each complex, the activity of each one was examined against the target cell line A-172, as compared with the ligand alone. It was found that the complexes 1 and 2 have a higher cellular cytotoxicity than unconjugated ligand as follow (Figure 4).

The data obtained that the prepared complexes 1 and 2 caused more inhibition on growth of target cells A-172 (45.7 %), (49.2 %) than ligand (30.6 %) (Figure 5). In addition, (IC50) was calculated for complexes and unconjugated ligand to be 8.1µM for ligand alone and 3.1µM and 2.8 µM for complexes 1 and 2, respectively. Thus, it indicated the important role for each compound to improve the activity of ligand as anticancer agent. Complex 2 shows a high activity than complex 1, this may be related to the existence of methyl groups besides a high symmetry in complex 2 and the high contents of tin as compared with complex 1 containing tri phenyl group.
Conclusion
Cisplatin or cis-amino dichloroplatinium (II) (CDDP) is a platinum-based chemotherapy drug. Cisplatin is used to treat different cancers, including sarcomas, some carcinomas (such as small cell lung cancer and ovarian cancer), lymphomas, and germ cells. Cisplatin is an anti-neoplastic (anti-cancer) drug from the class of alkylating agents and is used to treat various types of cancers. The reason for naming alkylating agents is because of their ability to add alkyl groups to many electronegative groups in cells. They stop tumour growth by directly attacking the DNA and creating a cross-link between the guanine bases in the double-stranded DNA strand. This prevents the chains from being able to separate from each other, which is essential in the transcription process, so the cells cannot be divided and multiplied. In this study, we found that the anticancer activity depends on the geometrical structure of the prepared complexes and type of substituted organotin groups. The two prepared complexes show a higher antitumor activity than the ligand derived from.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Authors' contributions
All authors contributed to data analysis, drafting, and revising of the paper and agreed to be responsible for all the aspects of this work.
Conflict of Interest
The author declared that they have no conflict of interest.
HOW TO CITE THIS ARTICLE
Eman H. Al-Rikabi, Rana A. K. Al-Refai, Sadiq J. Baqir, Angham G. Hadi, Ahmed Kareem Al-Qayyim. Synthesis, structure, and in vitro cytotoxic activity of two organotin complexes of 2- [(2, 3-dimethylphenyl) amino] benzoic acid. J. Med. Chem. Sci., 2023, 6(6) 1230-1238

![Synthesis, Structure, and in vitro Cytotoxic Activity of Two Organotin Complexes of 2-[(2, 3-Dimethylphenyl) Amino] Benzoic Acid](data/jmcs/coversheet/571668137739.jpg)
