Document Type : Original Article
Authors
1 Department of Chemistry, Payame Noor University, PO BOX19395-3697 Tehran. I.R of Iran
2 Department of Chemistry, Kosar University of Bojnord, Bojnord, Iran
3 Associated Professor of Dermatology NKUMS, Bojnord, Bojnord, Iran
4 Department of Biology, Payame Noor University, PO BOX19395-3697 Tehran. I.R of Iran
Abstract
The study of alkaloids from methanolic extract of Crucianella sintenisii from Rubiaceae family has not been studied yet. In this research, the methanolic extract was initially prepared from the aerial parts of Crucianella sintenisii, and then the resulting extract was acidified by acetic acid to remove neutral compounds. The neutral compounds were extracted by EtOAc (ethyl acetate). The resulting extract was alkalized with NH3 (25%), and then extracted with chloroform solvent. Finally, the crude alkaloid is extracted and ready to be injected into the MPLC device. One fraction was isolated, and the structure of 2-methyl-3-phenyl indole from the category of indole alkaloids was confirmed by using the spectroscopic techniques (NMR, ESI-MS, and IR). The therapeutic activities of the plant indole alkaloids are extensive, and most of these activities are related to the indole core in these structures.
Graphical Abstract
Keywords
Introduction
Since obsolete times, nature has been a significant source of the pharmaceutical products with a wide range of pharmacological activities due to the variations in their skeletal formula properties and target biomolecules [1]. Rubiaceae is one plant family with a considerable chemical potential. Rubiaceae, with 13143 species and distribution in 611 regions, is one of the prominent plant families [2].
The secondary metabolites of the Rubiaceae family have a high medicinal potential, and this factor can be used as a marker for genera and subfamilies in chemical taxonomy [3]. Different species of this family have been investigated in terms of the potential for biological activities, such as antiplasmodial [4], antibacterial [5, 6], anti-inflammatory [7], and anti-diabetic activities [8].
The genus Crucianella is a flowering plant that belongs to the Rubiaceae family. Iiridoids [9], Coumarins [10], Quinone compounds, triterpenoids, and flavonoids [11] are among the compounds that have been extracted and identified from this genus.
Alkaloids as the secondary metabolites can create different drugs with different therapeutic effects [12]. The study of alkaloids extracted from the family (Rubiaceae) shows that different species of this family can be a promising source of new compounds with the significant medicinal effects [9].
In general, medicinal plants containing alkaloids are widely used in traditional medicine. Some of them have shown anti-inflammatory, antibacterial, antiviral, antioxidant effects on the vascular diseases [10], and activity on the central nervous system [13].
Most terpene indole alkaloids are produced from plants of the Rubiaceae, Apocynaceae, Loganiaceae, and Nyssaceae families. These compounds can show the complexity of alkaloid biosynthesis. Most of the indole alkaloid compounds have medicinal effects, among which the compounds Ajmalicine (hypertension treatment), Yohimbine (adrenergic receptor blocker), Ajmaline (cardiac arrhythmia treatment), and Vincristine (anti-cancer) can be mentioned [14].
In addition, alkaloids in the plant can have a protective role and prevent it from being eaten by animals. However, some animals have adapted to eat plants containing alkaloids and even use these compounds in their metabolism [15].
Different procedures, such as electrophoresis and chromatography, were used for separating these naturally occurring biopharmaceutical products [16].
There are two rings in the structure of indole alkaloids, the six-membered ring of benzene, and the five-membered ring of pyrrole. The basic properties of indole alkaloids are related to the presence of a nitrogen atom in the structure of pyrrole, which leads to the therapeutic activity [17].
Concerning the richness of alkaloid compounds in the Rubiaceae family [18], we decided to investigate the alkaloid compounds of Crucianella sintenisii plant. So far, no report has been done on the extraction and identification of alkaloids in the Crucianella sintenisii plant. Our research started with the chloroform extract of the Crucianella sintenisii plant. The present research led to the isolation of 3-methyl-2-phenylindole (Scheme 2) as an alkaloid reported for the first time from this plant and the identification of its structure from the chloroformic extract of this species. Alkaloid separation was done by the medium-pressure liquid chromatography (MPLC). Likewise, FTIR, NMR, and spectrophotometer techniques were used to interpret the skeletal formulas of these isolated products [19].
Martials and methods
The spectroscopic devices and the low-pressure liquid chromatography device used in this research are as follows:
NMR: BRUKER 400MHZ spectrometer.
FTIR: Avatar 370 FTIR Thermo Nicolet.
ESI-MS: HP5973 Series Mass spectrometer.
MPLC: Büchi 861 equipment (Switzerland).
Plant material
The aerial parts of the Crucianella sintenisii plant in the flowering stage were collected from the Derkash region of North Khorasan province, Iran, in May 2020. They were identified in the Plant Science Research Centre of Ferdowsi University of Mashhad. A voucher specimen has been deposited in the Environmental Department of Bojnord Herbarium (EDBH00104) (Scheme 1).
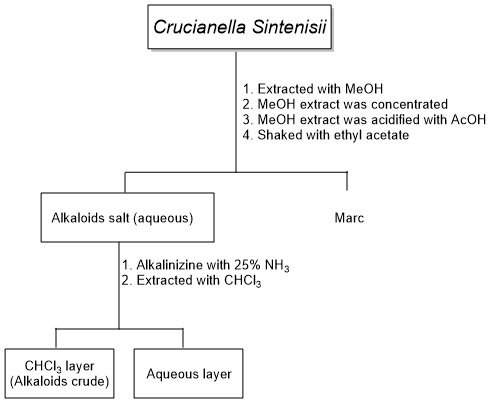
Scheme 1: The extraction steps of crude alkaloid
Extraction and isolation of alkaloid
The powdered (1 kg) of the Crucianella sintenisii plant was extracted with MeOH (2.0 L) by maceration method. The extraction process was repeated until the alkaloid test (Dragandrof's reagent) was negative. The methanolic extract was concentrated, and 270 gr of crude extract was obtained with a yield of 27.0%.
The methanolic extract was diluted with the aqueous acetic acid 60% (pH = 2) to a volume of 400 ml. To remove the neutral compounds, the extraction was done with ethyl acetate (3 × 200 mL).
After the removal of neutral compounds, the aqueous layer was alkalized with NH3 25% (pH=10-12), and then extracted with chloroform solvent (10×300 mL). The obtained crude alkaloid was dried at 30 °C. For MPLC analysis, 2 g of the crude alkaloids were dissolved in dichloromethane/methanol (10:1). Then, 10 µl were injected onto a column chromatography on silica gel, eluting with dichloromethane/methanol (10:1) to give a fraction.
3-methyl-2-phenyl indole alkaloid
Solid, IR (KBr) (νmax/ cm-1): 3500, 300, 3035, 3075, 1000-1350, 1475, and 1600. EI-MS 70 eV, m/z: 207. 1H-NMR (400 MHz, Chloroform): δ 7.01 (1H, m), 7.06 (1H, m), 7.37 (1H, m), 7.81 (1H, d), 7.43 (1H, d), 7.44 (1H, m), 7.47 (1H, m), 9.34 (1H, s) 2.42 (3H, s). 13C-NMR (100 MHz, Chloroform): δ 136.5, 135.9, 130.4, 129.3, 127.9, 127.8, 126.0, 122.5, 118.6, 118.7, 111.2, 109.8, 14.1.
Results and Discussion
In this investigation, the aerial parts of the Crucianella sintenisii plant were collected from Darkash protected area located in North Khorasan province in Iran during the flowering stage in June 2020, and then alkaloids were exhaustively extracted (Scheme 1).
The crude alkaloids were extracted by CHCl3 solvent and were subjected to MPLC. The solvent system (CH2Cl2/MeOH) (10:1) was selected for medium-pressure liquid chromatography according to the best separation mode tested by TLC. By performing a separation step by MPLC device on the crude extract, one fraction was obtained. Finally, the pure compound weighing 29 mg was obtained. IR (Figure 1), 1H-NMR (Figure 2), 13C-NMR (Figure 3) and EI-MS (Figure 4) spectroscopic methods were used to determine the structure of this compound.

Figure 1: IR spectrum of 3-methyl-2-phenyl indole

Figure 2: 1H-NMR spectrum of 3-methyl-2-phenyl indole

Figure 3: 13C-NMR spectrum of 3-methyl-2-phenyl indole

Figure 4: Mass spectrum of 3-methyl-2-phenyl indole
The IR spectrum (Figure 1) is dominated by the 3035 cm-1, and 3075 cm-1, and 680-1000 cm-1 absorption bands in the spectral region correspond to the C-H stretching vibrations of the aromatic ring. The aromatic C-C bond appears as a single absorption band at 1595 cm-1. The stretching absorptions of the dual-band of the ring appear in pairs at 1600 cm-1 and 1475 cm-1. The stretching absorption of C-N appears in the area of 1000-1350 cm-1. The N-H stretching vibration is a high-intensity peak by about 3500 cm-1. The stretching vibrations of the -CH3 group are the firm peaks at about 3000 cm-1. This compound was isolated as a colourless solid (mp 91-92 °C). The 1H-NMR spectrum showed signals at 7.01 (1H, m), 7.06 (1H, m), 7.37 (1H, m), 7.81 (1H, dd, J=7.3, 1.7Hz), 7.43 (1H, dd, J=7.5, 1.6 Hz), 7.44 (1H, m), 7.47 (1H, m) in the aromatic region , N-H at δ 9.34 (1H, s) and methyl δ 2.42 (3H, s).
The signals present in the 13C-NMR spectrum (Figure 3) were related to the aromatic ring carbons at δ 109.8-136.5 and the aliphatic carbons at δ 14.1.
The mass spectrum (Figure 4) for this compound is dominated by the m/z 206 fragments as the base ion (C15H12N+ fragment) and the m/z 130 fragment (-C6H5+) with very little molecular ion at m/z 207, corresponding to the formula of C15H13N with a very little molecular ion at m/z 77, corresponding to the formula of -C9H8N.
Conclusion
Due to the importance of extracting and isolating alkaloids from the natural sources, a new era has been created in which the synthetic drugs have largely replaced the herbal drugs due to the serious side effects caused by the use of synthetic drugs. The chemical profile for each subfamily, as expressed by the occurrence of the major categories of the secondary metabolites (indole alkaloids, iridoids, triterpenes, and anthraquinones) is remarkably distinctive. Regarding the distribution of the major secondary metabolites in Rubiaceae, indole alkaloids are indicated as the main chemical markers of this family. Indole alkaloids occur just in families belonging to the Gentianales order (Loganiaceae, Rubiaceae, Apocynaceae, and Naucleaceae), where one observes monoterpene indole alkaloids mainly. Several studies have been conducted concerning their chemical compositions, showing that this family is a potential source of alkaloids. In summary, one indole alkaloid was isolated from Crucianella sintenisii with MPLC. Isolation and determination of the structure of alkaloid 3-methyl-2-phenylindole from the Crucianella sintenisii are reported for the first time. According to the therapeutic effects of alkaloids explained in the introduction, the extraction and identification of alkaloids from plants can be of interest for the pharmacists and doctors.
Acknowledgements
The authors are grateful to Dr. Nematullahi for her cooperation in this research.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Authors' contributions
All authors contributed to data analysis, drafting, and revising of the paper and agreed to be responsible for all the aspects of this work.
Conflict of Interest
The author declared that they have no conflict of interest.
HOW TO CITE THIS ARTICLE
Malihe Nasrabadi, Majid Halimi khalilabad, Hamid Soorgic, Mohabat Nadaf. Isolation and Structure Elucidation of Methylphenylindole Alkaloid from Crucianella Sintenisii Growing in Iran. J. Med. Chem. Sci., 2023, 6(4) 771-777
https://doi.org/10.26655/JMCHEMSCI.2023.4.8
URL: http://www.jmchemsci.com/article_158625.html


