Document Type : Original Article
Authors
1 Bilad Alrafidain University College, Pharmacy Department, Diyala-Baquba, Iraq
2 Ministry of Education, Education directorate Baghdad, Baghdad 10001, Iraq
3 Ministry of Education, Education Directorate Babylon, Babylon, Iraq
4 Babylon Sewage Directorate, Babylon, Iraq 009647703990427
5 Baghdad University, College of Science, Chemistry Department, Baghdad, Iraq
Abstract
The present work involved preparation of new polyimides drived from chalcone derivative, chalcone compound prepared from reaction of 4-amino acetophenone with vanillin, and then the chalcone which have amine group reacted with maleic anhydride to produce the corresponding amic acid. The synthesized amic acid compound was introduced in ring closure reaction by acetic anhydride and sodium acetate to produce maleimide. The new maleimdie was polymerized by free radical polymerization to yield homo and copolyimide with two vinylic monomers (acrylonitrile and methyl acrylate). The structures of synthesized compounds and polymers have been confirmed by physical properties and spectroscopy measurements (FT-IR) Fourier Transform Infrared Spectroscopy, (1H-NMR) nuclear magnetic resonance. In addition, antibacterial activity of polymers was examined against two types of bacteria, namely Gram-negative and Gram-positive and gives good results in bacterial inhibition. Likewise, the anticorrosion activity of polymers was studied by using potensiostat measurements in acidic medium and the polymers give inhibition efficiency (94%).
Graphical Abstract
Keywords
Main Subjects
Introduction
Corrosion inhibitors for steel in acidic conditions are vital for some industrial facilities and also intriguing from a theoretical one. Carbon steel is an iron alloy that corrodes quickly in acidic environments [1-3]. Acidic solutions are commonly employed in chemical laboratories and in a variety of industrial operations, including acid cleaning, acid pickling, oil wet cleaning, and acid descaling, among others. In the chemical and allied sectors, carbon steel is also used to handle alkaline, acid, and salt solutions under various conditions. Organic chemicals adsorb on metallic surfaces and slow down the rate of corrosion. Therefore, many organic compounds were used as corrosion inhibitors [4-8].
Chalcones are a significant group of natural compounds and their pharmacological properties have attracted a lot of attention in recent years. They are open-chain flavonoids with two aromatic rings linked via three carbon atoms and the unsaturated carbonyl system.
The inhibitory effect of chalcones on human pathogenic micro-organism can be related with the substitution patterns of the aromatic rings [9], so there are several uses for chalcones as anti-inflammatory, antibacterial, antioxidant, antifungal, antimalarial, and antitumor agents [10-12].
Polyimides one of the important compounds in organic chemistry and which have a wide range of applications in many fields (dielectrics, adhesives, separation membranes, photoresists, sealants, coatings, and optical materials) [13]. Polyimides are used as anticorrosion coatings because of their excellent thermal, chemical, and mechanical qualities [14, 15]. Researchers have recently concentrated their efforts on the development of superior anticorrosive polyimide. Therefore, in this work, we synthesize the new polyimide with important moieties (vanillin, chalcone, methyl acrylate, and acrylonitrile). All these moieties give the polymers good properties and good results when used against antibacterial activity and in iron protection from corrosion in acidic medium (HCL 1M).
Materials and Methods
The chemicals were brought from (Sigma and Fluka) were used without extra purification. The melting points determined on (Cole-Parmer Stuart) melting point apparatus.
Infrared spectra (IR) were recorded on (Shimadzu FT-IR-8400 Charles/FT-IR spectrophotometer) by using (KBr pellets), 1H-NMR spectra of chalconoe, and polymers were recorded on (nuclear magnetic resonance Bruker 400 MHz).
Synthesis of chalcone (1)
Sodium hydroxide solution 50% (w/v) (3 mL) was added to the mixture of P-aminoacetophenone (0.01 mol, 1.35 g) and 0.01 mol, 1.52 g of 4-hydroxy-3-methoxybenzaldehyde in 20 mL absolute ethanol with stirring [16]. The homogenized mixture was stirred for about 12 hours at room temperature, and then it was poured onto the crushed ice. The obtained precipitate was filtered, dried, and recrystallized from ethanol to afforded brown crystals in 81% yield and melting point 65-67 °C.
Synthesis of 1-(4-aminophenyl)-3-(4-hydroxy-3-methoxyphenyl) prop-2-en-1-one maleamic acid (2)
A solution of 0.01 mol of compound (1) dissolved in a suitable volume of acetone was added dropwise to 0.01 mol maleic anhydride solution dissolved in suitable volume of acetone with stirring [17]. The resulting mixture was stirred for additional 2 hours. Thereafter, the resulted solid was collected by filtered, washed with ether, dried, and finally recrystallized from dioxane to afford pale brown crystals. Yield: 76%, m.p 95-98 °C.
Synthesis of 1-(4-aminophenyl)-3-(4-hydroxy-3-methoxyphenyl) prop-2-en-1-one maleimide (3)
The titled compound (3) was prepared by dehydration of compound (2) via fusion method by heating (1 g of compound (2) in sand bath until melting, and then gently the temperature was increased to 10 degrees above the melting point of compound (2) for 2 hours. The resulted precipitate was recrystallized from absolute acetone to afford black crystals.Yield: 90%, mp >300 °C.
Synthesis of polyimide (4, 5, 6)
- a) Homopolymerization
Monomer (3) (0.003 mol) dissolved in THF was placed in a dark polymerization bottle and initiator (AIBN) was added to solution, and then the bottle was flushed with nitrogen and quickly stoppered. After that, the bottle was heated at 75 °C for 4 hours. After heating completion, the mixture was cooled and methanol was added with stirring, the resulted polymer was filtered, and then washed with methanol and was remained to dry [18]. Compound 4: Black, Softening point 37-45 °C, conversion ratio: 88%.
b) Copolymerization
Equal amounts of monomer (3) and vinylic momoers (methylacrylate or acrylonitrle) were dissolved in THF was placed in a dark polymerization bottle, and initiator (AIBN) was added to solution, and then the bottle was flushed with nitrogen and quickly stoppered. After that bottle was heated at 75 °C for 4 hours. After heating, the resulted mixture was cooled and stirred with methanol. Then, the resulted polymers were filtered, washed with methanol, and dried. Compound 5: Brown, Gummy, conversion ratio 79%. Compound 6: Dark brown, Gummy, conversion ratio 82%.
Anti-corrosion activity study
The potentiostat setup consists of a (Host computer, magnetic stirrer, thermostat, (EmStat 4s, Palm Sens, Holland) potentiostat, and galvanostat). 250 mL Pyrex cell is made up of an interior and exterior bowl. Three electrodes make up the electrochemical corrosion cell. The saturated calomel (Hg/Hg2Cl2 sat.KCl) is used as the reference electrode, a platinum electrode with a length of 10 cm is utilized as the auxiliary electrode, and carbon steel serves as the working electrode used to determine its potential according to the reference electrode. To achieve (steady state open circuit potential Eocp), the working electrode was submerged in the test solution for 15 min. Following this, electrochemical measurements were carried out in a potential range of ±200 mV. All experiments were conducted in a cooling-heating circulating bath at various temperatures.
The corrosion cells
The corrosion cell made of Pyrex with 250 mL capacity involves two (internal and external) vessels. Three electrodes and thermostat replaced in an internal vessel, the corrosion cell, and three electrodes are depicted in Figure 1.
Descriptions of three electrodes
A reference electrode used to set the potential of working electrode according to the potential of reference electrode. Two tubes are joined in it; the inner tube contains Hg/Hg2Cl2 sat.KCl, the reference electrode was stand at 2 mm from the working electrode.
- The Auxiliary Electrode which is made from platinum metal of exceptional purity, and measures (10 cm).
- The Working Electrode was created from 20 cm of metallic wire and attached to the specimen that was mounted.
Antibacterial activity study
The antibacterial activity of producing the prepared compounds against micro-organisms (stap. and E.coli) was studied by using the cup plate technique. In addition to DMSO, nutrient agar medium was employed. Sample volume and solution for all of the examined compounds were renamed (0.1 mL). Scooping out of cups in an agar medium enclosed in a petri dish. Previously, the bacteria were incubated. The examination was done in the cups, 0.1 Ml of compound solution was added, and the petri dishes were incubated for 48 hours at 37 °C. Zones of influence for each compound’s inhibition was measured in millimeters [19].
Results and Discussion
Chalcone and polyimide are both significant active ingredients that demonstrate a broad applicability. The current effort involves the synthesis of novel derivatives that have these two active components linked together in a single molecule before evaluating their anti-corrosion action. This target is done by four steps. In the initial step, chalcone (1) is prepared by a direct reaction between 4-aminoacetophenone and 4-hydroxy-3-methoxybenzaldehyde (vanillin). The resulted compound was a reaction in the second step with maleic anhydride to produce amic acid (2) that dehydration in third stage via (fusion method) to produce the maleimide (3). On the other hand, the maleimide was polymerized by free radical polymerization to yield homo and copolyimide with two vinylic monomers (acrylonitrile and methyl acrylate) (4, 5, 6).
The synthetic route of the synthesized compounds was displayed in Scheme 1.
Chemical structures of the synthesized compounds and polymers were confirmed by FT-IR and 1H-NMR spectral data.
Infrared spectrum of chalcone (1) presented absorption bands at 3373-3392 cm-1 owing to ʋ(O-H) and the other bands at 3221-3298 cm-1 belong to ʋ(N-H) amine. The spectrum also displayed the strong absorption bands at 1647-1672 cm-1, 1589-1637 cm-1, 1494-1545 cm-1, and 1265-1280 cm-1 which recognized to carbonyl ketone, ʋ(C=C) chalcone, ʋ(C=C) ring, and ʋ(C-O), respectively, while the absorption bands owing to ʋ(C-O-C) seemed at 1232 cm-1.
1H-NMR spectrum of chalcone (1) showed a clear signal at δ=3.76 ppm belong to protons of O-CH3 group and signals at δ=7.95-8.56 ppm belong to (CH=CH-C=O) chalcon protons as well as the signals at 6.63-7.67 ppm fit to aromatic protons, while signals belong to O-H proton and NH amine proton seemed at δ=9.25 ppm and δ=6.02 ppm, respectively [20].
Infrared spectrum of amic acid (2) presented the presence of clear absorption bands at 3479 cm-1, 3412-3429 cm-1, and 3369-3400 cm-1 owing to ʋ(O-H) carboxyl, (O-H) phenolic, and (N-H) proving the success of compound (2) formation. The other bands looked at 1670, 1645, 1577, and 1415-1512 cm-1 which are due to carboxyl group, amide group, ʋ(C=C) chalcone, and (C=C) aromatic, respectively. Besides, the bands owing to sym. and asym. ʋ(C-O-C) seemed at 1296 and 1078 cm-1, respectively.

Figure 1: Set up the corrosion cell and three electrodes

Scheme 1: Synthetic route of the prepared compounds and polymers
Infrared spectra of the synthesized imide (3) displayed the clear characteristic absorption bands at 1743, 1716, and 1624 cm-1 which owing to ester group, imide group, and ketone group, respectively. The other absorptions bands seemed at 1512-1600 cm-1, 1400-1490 cm-1, 1267 cm-1, and 1080 cm-1 which are owing to ʋ(C=C) chalcone, aromatic, asymmetric and symmetric ʋ(C-O-C) cm-1(6).
1H-NMR spectrum of maleimide (3) displayed a signal at δ = 1.91 ppm, δ = 3.84 ppm, and δ = 6.7-6.81 ppm which are fit to O=CCH3 protons, (OCH3) protons, and vinylic protons in imide, respectively, while signals fit to (C=C) aromatic protons seemed at δ = 6.91-8.23 ppm.
The infrared spectra of compound (4) demonstrated the main bands for C-H aliphatic groups which were due to the polymer formation at 2970-2941 cm-1, also absorption bands at 1763, 1716, and 1685 cm-1 owing to ester group, imide group, and ketone group, although the absorption bands owing to ʋ(C=C) chalcone, aromatic, ʋ(C-H) aromatic, asym., and sym. ʋ(C-O-C) seemed at 1508-1600 cm-1, 1417-1485 cm-1, 3007-3078 cm-1, 1263 cm-1, and 1078 cm-1.
1H-NMR spectrum of polymer (4) presented a signal at δ = 1.36-2.53 ppm, δ = 3.37 and δ = 3.88 ppm which are fit to (CH2), (O=CCH3), and (OCH3) protons, while signals fit to C=C aromatic protons looked at δ = 6.84-8.1 ppm.
The infrared spectra of the compound (5) illustrated the clear distinguishing absorption bands at 1764, 1716, and 1685 cm-1 which are belong to (C=O) ester, ʋ(C=O) imide, and ʋ(C=O) ketone, respectively. Likewise, the main absorption bands at 2943-2910 cm-1 due to C-H aliphatic groups, and band of (C≡N) group at 2241 cm-1, further absorptions bands seemed at 1602-1630 cm-1, 1452-1510 cm-1, 1267 cm-1, and 1078 cm-1 that are belong to ʋ (C=C) chalcone, ʋ (C=C) aromatic, asymmetric and symmetric ʋ (C-O-C) cm-1, respectively [20, 21].
1H-NMR spectrum of polymer (5) displayed a signal at δ = 1.36-2.34, δ = 3.35, and δ = 3.86 ppm which are due to (CH2) protons, (O=CCH3) protons, and (OCH3) protons, while signals fit to (C=C) aromatic protons seemed at δ = 6.88-8.1 ppm.
The infrared spectra of compound (6) presented the clear characteristic absorption bands at 2997-2848 cm-1 owing to C-H aliphatic, also showed the bands at 1743-1766 cm-1, 1735-1720 cm-1, and 1710 cm-1 belong to ʋ (C=O) eater, ʋ (C=O) imide, and ʋ (C=O) ketone, while absorption bands owing to ʋ (C=C) chalcone, ʋ (C=C) aromatic, symmetric and asymmetric ʋ (C-O-C) looked at 1602-1687 cm-1, 1419-1510 cm-1, 1267, and 1079 cm-1.
1H-NMR spectrum of polymer (6) presented a signal at δ = 1.24-2.34 ppm, δ = 3.35 ppm and δ= 3.78-3.88 ppm which fit to (CH2) protons, (O=CCH3) protons, and (OCH3) protons, while signals due to (C=C) aromatic protons seemed at δ = 6.88-8.08 ppm.
Antibacterial activity of synthesized polymers
One of the wide spectrums of applications that imides compounds possess is the noticeable antibacterial activity. The antibacterial activity of synthesized polyimides was tested by studying these activities against two types of bacteria, namely, gram-negative (Escherichia coli) and the gram-positive (Staphylococcus aurous), by using the well-diffusion approach which includes tracking the inhibition level of these compounds by using the lowest inhibitor concentration [22].
As presented in Table 1, the polymer number (6) has the best activity against gram-positive and gram-negative bacteria compared with the standard material. The antibacterial results are listed in Table 1.
Anticorrosion measurements
From the resultant data in Tables and Fgures, the corrosion parameters were assessed. The corrosion potential (Ecorr.) and corrosion current density (icorr.) were got by the extrapolation of the anodic and cathodic Tafel in presence and absence of the inhibitors molecules in HCL solution. The (anodic ba) and (cathodic bc) Tafel slopes were calculated from the measurements as depicted in Figures 2, 3 and 4. Table 2 displays the resulting data of corrosion current density icorr (A/cm2) and corrosion potential Ecorr (mV), cathodic and anodic Tafel slopes (mV/Dec), and the protection efficiency PE%. Tafel plot exposes that (Ecorr.) for C.S in the presence of the inhibitors shifts to higher (noble) position related with blank solution, suggesting that the protection is an anodic protection.
Inhibition efficiency was calculated through following equation.

Where, (icorr) is the corrosion current density in the presence of inhibitors, and (icorr)o is the corrosion current density in the absence of inhibitors [23].
Polymers give an excellent portective for carbon steel in 303 K, the protective efficiency gives by polymers (4) and (6) is (94%), while polymer (5) gives 92% protective efficiency.


Figure 2. Polarization curves for corrosion of blank solution and 4 compounds in different temperatures

Figure 3: Polarization curves for corrosion of blank solution and 5 compounds in different temperatures
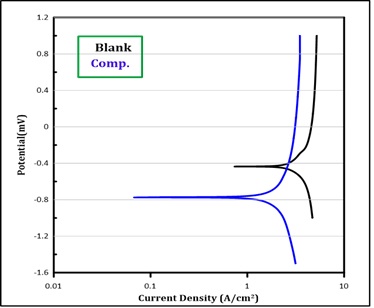
Figure 4: Polarization curves for corrosion of blank solution and 6 compounds in different temperatures

Conclusion
In conclusion, we have designed and prepared new polymers drives from chalcone derivative in a good yield. Likewise, these polymers have a simple structure and different functional groups which give the anti-corrosion activity of polymers. The new polymers have a good activity as the corrosion inhibitor for carbon steel in acidic medium with protection activity for polymers is 94%, and the protection is an anodic protection.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Authors' contributions
All authors contributed to data analysis, drafting, and revising of the paper and agreed to be responsible for all the aspects of this work.
ORCID:
Mustafa D. Husseini
https://www.orcid.org/0000-0002-1723-1855
HOW TO CITE THIS ARTICLE
Baraa H. Latief, Raghad H. Ismail, Bahaa F. Hamzah, Mustafa D. Husseini, Noor A. Khudhair. Antibacterial and Anticorrosion Activity Evaluation of New Polymaleimide Derived from Chalcone Derivative. J. Med. Chem. Sci., 2023, 6(4) 755-763


