Document Type : Original Article
Authors
1 Pharmacy Department, Al-Mustaqbal University College, Babylon, Iraq
2 Department of Pharmacology, College of Medicine, Al-Nahrain University, Baghdad, Iraq
3 Radiological Techniques Department, Al-Mustaqbal University College, Babylon, Iraq
Abstract
Sepsis is an upregulated systemic inflammatory reaction that has been identified as a global health concern. It can lead to multiple organ toxicity, including cardiac, hepatic, and renal dysfunction. Sepsis can result in cardiomyocyte impairment, hypertrophy, and heart failure by increasing oxidative stress and the expression of pro-inflammatory cytokines. Furthermore, elevated cardiac troponin-I (cTn-I) during sepsis impairs heart contractile performance by decreasing myofilament response to calcium. The current study aimed to examine the possible effects of levosimendan against sepsis-induced cardiotoxicity. Forty male mice 8-12 weeks old and weighing 25-30 grams. After two weeks of acclimation, the mice were separated into four groups (n = 10): (1) Healthy group (n = 10). (2) CLP group: CLP operation was performed on mice. (3) DMSO group (4) Levosimendan group: 10 mg/kg intraperitoneally (IP) in 2 divided doses for 5 consecutive days. All groups underwent the CLP procedure on the fourth day, were sacrificed on the fifth day, and then samples were taken. The Levosimendan group exhibited a significant (p < 0.05) decrease in myocardial troponin-I concentration compared to the CLP group. In addition, the inflammatory cytokines (TNF-α, IL-6, and IL-1β) serum levels in the levosimendan group were significantly (p < 0.05) lower than in the CLP group. In addition, the levosimendan group demonstrated a significant (p0.05) increase in myocardial SOD activity and a decrease in MDA level compared to the CLP mice. Histopathologically, the levosimendan group demonstrated a statistically significant (p0.05) reduction in cardiac tissue damage. Levosimendan attenuates sepsis-induced cardiotoxicity via multiple protective effects, including anti-inflammatory and antioxidant effects, according to the findings of the present study. In addition, the direct cardiac protective effect via the decreased cardiac troponin-I level and the attenuation of histopathological alterations during sepsis-induced cardiotoxicity.
Graphical Abstract
Keywords
Main Subjects
Introduction
Sepsis can be defined as an upregulated systemic inflammatory response resulting from bacterial invasion [1]. Sepsis is regarded as a global health concern, and infection is an important cause of death. Therefore, early recognition and diagnosis of sepsis can lead to the prevention of septic shock, which has a high mortality rate of about 40% and even more [2]. As a systemic immune response, sepsis can produce multiorgan failure, including cardiac, renal, hepatic, and cerebral dysfunction. Cardiac dysfunction, as an essential organ attack, can occur in humans in addition to rodents during sepsis [3, 4]. The pathophysiology of cardiomyopathy during sepsis, includes complicated different systemic factors, molecular, metabolic, and even structural changes in the myocardial cells [5]. Cardiotoxicity is a frequent result of sepsis accompanied by more significant mortality. It has been linked to elevated inflammation, inhibition of both oxidation of glucose and fatty acid oxidation, ATP depletion, and impairment of cardiac adrenergic, all of which worsen heart functions [1]. Following bacterial infection, the body's inflammatory reaction can progress to severe sepsis and septic shock, characterized by ischemia, decreased blood pressure, multiorgan injury, and even death [2]. As a severe health consequence, sepsis can potentially raise patient death and healthcare costs [6].
Because of its complexity, there is no complete explanation or assumption that can give the exact mechanisms of sepsis-induced cardiotoxicity. Furthermore, pro-inflammatory cytokines have a key role in myocardium depression; other mechanisms that may explain sepsis-induced cardiotoxicity may include oxidative stress pathway, calcium transport deterioration, energetic dysregulation, autonomic dysregulation, and apoptosis pathway [7-10]. Additionally, hypotension, hypovolemia, acidosis, and hypoxia may contribute to the pathophysiology of sepsis-induced- cardiotoxicity [10]. On the other hand, cardiac troponin release into circulation during sepsis is due to the increase in membrane permeability of cardiomyocytes [11]. A more obvious explanation regarding the pathophysiology of sepsis-induced cardiotoxicity is an interaction between different mechanisms, such as inflammation, oxidative stress, metabolism, and neuroimmunomodulation pathways [7].
Levosimendan mainly acts as a calcium sensitizer in addition to its action as a potassium channel opener. It is widely used to relieve the symptoms of acute heart failure, consequently to its effect on both systemic and pulmonary hemodynamics [12, 13]. Additionally, it is indicated in the case of decompensated heart failure due to its inodilator effect, which the following mechanisms can obtained: the first mechanism is by increasing the troponin C sensitivity to calcium within the cardiomyocytes, thereby positively modulating the cAMP-independent inotropic impact, the second mechanism is by its effect as a vasodilator through activating of ATP sensitive K channel within the vascular smooth muscle, the third mechanism is by the stimulation of ATP sensitive K channel within cardiac mitochondria hence protect the cardiomyocytes against IR injury [13, 14]. As far as we know, there is little information on the involvement of levosimendan in attenuating sepsis-induced cardiotoxicity. So, this study aimed to study the possible protective impact of levosimendan during sepsis-induced cardiotoxicity.
Materials and Methods
Drugs and chemical
Levosimendan (Simenda®) 12.5mg/vial for injection was obtained from Lupin LTD, Mumbai, India. Simenda® was reconstituted according to manufacturer instruction and then given intraperitoneally 10 mg/kg/day in two divided doses [15].
Animals of study
Forty male mice, aged 8 to 12 weeks and weighing 25 to 30 g, were employed in the current study. These mice were confined to their cages in an animal house with 12:12 light: dark cycles, 25 °C room temp, 60-65% humidity levels, and the availability of free food and water.
Study design
The forty mice were left for two weeks for adaptation, and then after mice were assigned to the following groups (n equal to 10 in each group):
Normal group: Apparently healthy mice.
CLP (Septic) group: the induction process was done without treatment.
DMSO group: Animals in this group injected an equivalent volume of DMSO as a vehicle.
Levosimendan group: Animals in this group received levosimendan 5 mg/kg IP twice daily for 5 consecutive days, followed by a CLP operation on the fourth day.
The experimental model of induced sepsis (CLP)
In this work, animals were chosen to develop polymicrobial sepsis. Depending on previous studies, sepsis was induced via needle using the method of cecal ligation and puncture (CLP) [2, 16]. Briefly, a needle with 18-G was applied in the twice puncture method to evoke organ (cardiac) impairment during the initial stage of polymicrobial sepsis (1 day). 1.5 ml/kg of a one: two ratio solution of xylazine and ketamine was administered to induce anaesthesia [17]. The cecum was exposed when the abdomen was going to open with a 1.5 cm midline abdominal incision. The cecum was then ligated and punctured just behind the ileocecal area before being returned to its anatomical position. A small portion of the faeces was extracted to verify the patency of the puncture sites. The abdominal wall was then sutured. All animals have received a subcutaneous dose of saline solution (20 mL/kg body weight) for resuscitation.
Collection of samples
On the fifth day, mice were sacrificed with anaesthesia, then both blood and tissue samples were obtained as follows:
Blood samples
After the scarification process had been done, blood was withdrawn using the direct cardiac puncture technique. For serum collection, withdrawn blood was left aside in a gel tube for about 20 minutes until the clotting process had occurred, then after, centrifuged at 10000 rpm for about 10 minutes. The supernatant was preserved at -20 °C until outcome analysis was done.
Myocardial tissue samples
After blood sample collection, a thoracic operation was done to obtain myocardial tissue. The heart was split into 2 halves; first part was kept at -20 °C until tissue homogenate analysis was done, while the other part was fixed with formalin until histopathological analysis.
Myocardial tissue homogenization
Tissue homogenization was done according to the previous study [18]. Briefly, a razor blade was used to cut tissue pieces, then 0.1 g was weighed from each sample, meanwhile placed the sample was on the ice to keep it from getting warm, followed by chopping the tissue using a single-edge razor; all these procedures were done on ice. After that 3 mL of phosphate buffer (pH=7.2) was added, then ice homogenization by grinding using a pestle until no more chunks were noticeable. The resultant sample was relocated to an Eppendorf tube, then centrifuged at 10,000 rpm for 10 minutes, and the supernatant was carefully obtained.
Outcome measurement
Inflammatory cytokines (TNF-α, IL-1 β, and IL-6)
Enzyme-Linked Immunosorbent Assay (ELISA) measured the TNF-α, IL-1β, and IL-6 serum levels following the manufacturer's recommendations. Mouse TNF-α, IL-6, and IL-1β antibodies had been pre-coated onto an ELISA kit plate. The wells were coated with TNF-α, IL-1β, and IL-6, which were present in the samples after they had been added and bound to antibodies. Once the samples contained TNF-α, IL-6, and IL-1β, biotinylated Mouse TNF-α, IL-6, and IL-1β antibodies were added and bound to them. The addition of streptavidin-HRP then bound the biotinylated TNF-α, IL-6, and IL-1β antibodies. Unbound Streptavidin-HRP has been removed during the washing process that follows incubation. Then the substrate solution was added, and the color changed based on the amount of Mouse TNF-α, IL-6, and IL-1β. The reaction was stopped by adding an acidic stop solution, and the absorbance was recorded at 450 nm.
Cardiac troponin I (cTn-I)
According to the manufacturer's instructions, an ELISA was used to find the myocardial concentration of myocardial troponin I (cTn-I). Mouse cTn-I antibody had been used to pre-coat the ELISA kit's plate. The sample's cTn-I was done add, and the wells were lined with cTn-I-linked antibodies. Afterward, the sample was incubated with a mouse cTn-I antibody that had been biotinylated and bound to cTn-I. The Streptavidin-HRP was then coupled with the biotinylated cTn-I antibody. Unbound Streptavidin-HRP flushed away following incubation during a washing procedure. The substrate solution was added, and the colour developed depending on how much Mouse cTn-I was present. After introducing an acidic stop solution, the process was stopped, followed by the absorbance measurement at 450 nm.
Oxidative stress markers
Myocardial superoxide dismutase (SOD)
Using a colorimetric technique a UV-VIS spectrophotometer was used to measure superoxide dismutase activity [19]. The strategy is based on the potency of SOD to suppress this interaction through free radical dismutation and the generation of pyrogallol-quinone from a reactive intermediate, the semiquinone radical. Brown substance pyrogallol-quinone absorbs the light at 420 nm.
Myocardial malondialdehyde (MDA)
The Buege & Aust Thiobarbituric Acid (TBA) detection method was used to measure malondialdehyde (MDA) on a spectrophotometer [20]. This technique measures the aldehyde byproducts of lipid peroxidation to quantify lipid peroxides. One molecule of malondialdehyde reacts with two molecules of thiobarbituric acid to produce a red MDA-TBA complex that can be observed at 535 nm. This reaction is the fundamental working principle of the method.
Histopathological study
As mentioned, the cardiac tissue was preserved in 10% formalin for one day [21]. In summary, 5 µm thick sections were paraffin-embedded according to the usual method. After that, hematoxylin and eosin (H&E) were used to stain the samples. Under an optical microscope, each heart slice (n = 3 per heart) was assessed for cardiac damage, and images were taken. Histological slices from all samples were analyzed and graded according to Zingarelli's technique to semi-quantify the change in heart damage [22], as shown in Table 1.
Table 1: Myocardial damage scoring system according to Zingarelli system
|
Score |
Tissue findings |
|
Score zero |
no damage, normal architecture |
|
Score one (mild) |
focal necrosis and interstitial oedema |
|
Score two (moderate) |
diffuse myocardial cell swelling |
|
Score three (severe) |
contraction bands and leukocyte infiltration are present |
|
Score four (highly severe) |
the presence of haemorrhage, leukocyte infiltrate, and contraction bands |
Statistical analysis
The SPSS 26 statistical analysis program was used. The analysis of variance (ANOVA) with LSD posthoc test was used to look into group differences, and the Kruskal-Wallis with Mann-Whitney U-test was used to look into histological differences. The significance of the current data was determined statistically to be p < 0.05.
Results and Discussion
Levosimendan diminished cardiac troponin
ELISA outcomes demonstrated that the myocardial cTn-I level was significantly higher (p < 0.05) in CLP and vehicles groups compared with healthy groups. In contrast, the levosimendan group showed a significantly lower level (p <0.05) of cTn-I compared to the untreated CLP group, Figure 1. The current study finding of the lower level of cTn-I in the levosimendan group may be attributed to its anti-inflammatory action is linked to its ability to inhibit the Rho-kinase signaling cascade, which may contribute to its cardioprotection against sepsis-induced cardiotoxicity [23]. Another proposed pathway can explain levosimendan cardioprotective effects and lower level of cTn-I by its effect as an inodilator via its work as a calcium sensitizer and activator of ATP-sensitive K channel within the vascular smooth muscle and cardiac mitochondria hence protecting the cardiomyocytes against [13, 14].
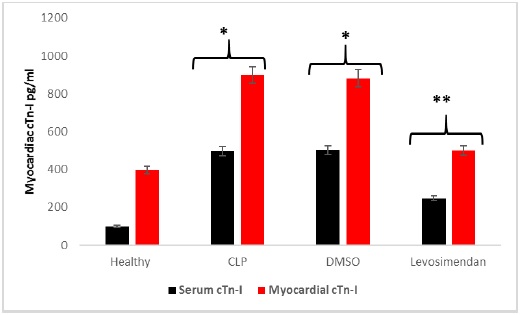
Figure 1: Mean serum & myocardial level of cTn-I (pg/L): Data are expressed as mean ± SD; *P <0.05 versus the healthy group; **P <0.05 versus the CLP group
Levosimendan attenuates inflammatory response
The serum levels of inflammatory cytokines such as TNF-α, IL-6, and IL-1β were measured using the ELISA technique 24 after CLP-induced polymicrobial sepsis to determine the effect of Levosimendan on the inflammatory response that takes place during sepsis. The current study showed that when compared to the healthy group, the serum levels of the inflammatory cytokines TNF-α, IL-6, and IL-1β were significantly higher in the CLP group (p < 0.05). In contrast to the CLP group, the levosimendan group displayed significantly lower (p 0 < 0.5) levels of inflammatory cytokines (TNF-α, IL-6, and IL-1β) (Figure 2). Levosimendan has anti-inflammatory properties that are associated with the suppression of the Rho-kinase signaling system, which may help to add value to its cardioprotection against sepsis-induced cardiotoxicity, as shown by the significantly lower plasma concentrations of cardiac troponin-I [24]. Additionally, it functions as an inhibitor for the main inflammatory kinase enzymes like activation of ERK, p38 MAPK, and protein kinase B (Akt/PKB), all of which can enhance the cellular production of inflammatory cytokines [25].
Levosimendan alleviated oxidative stress injury
The level of malondialdehyde (MDA) and the activity of the enzyme superoxide dismutase (SOD) in the myocardial tissue were also measured spectrophotometrically as part of the current study. The current study’s results demonstrated that, compared to healthy subjects, myocardial SOD activity was significantly lower in the CLP and vehicle groups (p < 0.05). While compared to the CLP group, the levosimendan group displayed significantly more SOD activity (p < 0.05). In addition, the CLP group's myocardial.
MDA level was significantly higher (p < 0.05) than the healthy group. Figures 3 shows that the levosimendan group showed a significantly (p < 0.05) lower MDA level than the CLP group. In earlier studies, Levosimendan can stop cardiomyocytes losing their mitochondrial membrane potential and from opening their transition pore [26]. It may alter ROS release in this manner, and levosimendan's maintenance of mitochondrial function may serve as the starting point for reducing ROS generation and restoration heart energy and cell survival [27].

Figure 2: Mean serum level of inflammatory cytokines (TNF-α, IL-6, and IL-1β): Data are expressed as mean ± SD; *P <0.05 versus the healthy group; **P <0.05 versus the CLP group

Figure 3: Mean myocardial activity of SOD (U/ml) and myocardial level of MDA (mol/ml): Data are expressed as mean ± SD; *P <0.05 versus the healthy group; **P <0.05 versus the CLP group
The effects of levosimendan sepsis-induced histopathological changes
A histological investigation of myocardial tissue was conducted to provide more information on the effects of levosimendan heart damage. In this histopathological analysis, serial sections of cardiac tissue were obtained one day after CLP-induced sepsis and subjected to Hematoxylin and Eosin staining (H&E). CLP myocardial tissue demonstrated a substantial significant (P < 0.05) myocardial damage as compared to the healthy mice, with the presence of contraction bands and polymorphonuclear leukocytes (PMN) infiltration, as well as necrosis, interstitial edema, and localized red blood cell extravasation. CLP group had a highly severe histological grading from normal cardiac tissue. In contrast, the levosimendan group exhibited a more significant (P<0.05) reduction in myocardial injury compared to the CLP mice. The histological changes were mild with a different number of mice. These findings agree with a previous study on mice to examine the cardioprotective effects of levosimendan during induced-polymicrobial sepsis [24, 28]. These findings are summarized in Figures 4, 5, and 6.
levosimendan significantly reduced cardiac tissue injury compared to the CLP and vehicle groups, the histopathological damage scores for the levosimendan group were usually nearly normal to mild. These cardioprotective effects of levosimendan may be related to its anti-inflammatory effect as a potent inhibitor for different MAPK kinase pathways such as ERK and p38MAPK as well as the Rho kinase pathway inhibition [25, 29-33].

Figure 4: Myocardial tissue sections of mice in the normal group: showed normal myocytes histology with clear cell borders and structures

Figure 5: Myocardial tissue sections of mice in the CLP group: showed inflammation (black arrow), contraction bands (red arrow), and necrotic karyolysis (white arrow)

Figure 6: Myocardial tissue sections of mice in the levosimendan group: showed a clear nucleus (black arrow) with nearly normal cellular borders (blue arrow)
Conclusion
Levosimendan ameliorated sepsis-induced cardiotoxicity in mice via exhibiting strong anti-oxidant (elevated SOD and decreased MDA), anti-inflammatory (suppressed TNF-a, IL-6, and IL-1β), and cardioprotective (decreased cTn-I), and improved heart histopathological changes. Levosimendan could be a good candidate therapeutic agent for protection against sepsis-induced cardiotoxicity.
Acknowledgments
Thank you to all contributors to this article.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Authors' contributions
All authors contributed to data analysis, drafting, and revising of the paper and agreed to be responsible for all the aspects of this work.
Conflict of Interest
The author declared that they have no conflict of interest.
ORCID:
Qassim A Zigam
https://www.orcid.org/0000-0002-1425-3450
Adeeb A. Al-Zubaidy
https://orcid.org/0000-0002-5207-383X
Zahraa Sami
https://orcid.org/0000-0003-4863-2237
Weaam J Abbas
https://orcid.org/0000-0003-0223-2924
HOW TO CITE THIS ARTICLE
Qassim A Zigam, Adeeb A. Al-Zubaidy, ZahraaSami, Weaam J Abbas. The Effect of Nanoparticles and Plant Growth Regulators on Germination of Miaze Seeds: A Comparative Study. J. Med. Chem. Sci., 2023, 6(3) 634-644


