Document Type : Original Article
Author
Department of Chemistry, College of Science, University of Thi-Qar, Iraq
Abstract
The present study includes the synthesis and characterization of thiazolidinedion containing indole ring. Two compounds were prepared: (4-fluorophenyl)-2-(1H-indol-3-yl)thiazolidin-4-one (I) and(4-( dimethylamino)phenyl)-2-(1H-indol-3-yl)thiazolidin-4-one(II), the prepared compounds diagnosed by using infrared spectra, NMR spectra (1H-NMR), and the results were identical to what is expected in practice. The Gaussian program was used for the computational study of thiazolidinedion and the theoretical calculations of the thermodynamic variables showed that compound (I) is the more softness with the lowest hardness. Meanwhile, compound (II) the more hardness with less softness.
Graphical Abstract
Keywords
Main Subjects
Introduction
Thiazolidinones are considered the saturated form of a thiazole, 1,3-thiazolidin-4-one. Thiazolidinones consist of a five-membered ring containing a sulfur, nitrogen, and carbonyl group [1,2] (Figure 1). Sulfur occupies position 1 in the five-membered ring, while nitrogen is in position 3, and the carbonyl group is in position 4 [3-5].

Figure 1: Thiazolidinone ring
Thiazolidinones have three isomeric [6], and they have the following structures:

Figure 2: Isomeric structures of thiazolidines
The thiazolidinedione ring is of great importance because it is a structure in many naturalproducts and medicines, for example thiazolidine-4-one derivatives (Figure 3). It indicates the activity against inflammatory, analgesic, and anti-ulcer [7].

Figure 3:3-phenyl-2-(ρ-toyl)thiazolidin-4-one
Also, the prepared compound 2-aryl-4-oxothiazoilidin-3-ylamides showed the activity against prostate cancer cells (Figure 4) [8].

Figure 4: Prepared thiazolidine compounds as against prostate cancer cells
Materials and methods
Synthesis of imines [9,10]
(E)-N-(4-fluorophenyl)-1-(1H-indol-34-yl)methanimine (a)
It was prepared by the reaction of indol-3-carboxyaldehyde (1 g, 6.89 mmol) with 4-fluroanline (0.77 g, 6.89 mmol) and (10 drops) of (CH3COOH) glacial acetic acid was added, and then they were refluxed in water bath for 16 hours. The product was precipitated and recrystallized with the addition of methanol droplets, (M.p=223-225°C), (Rf = 0.8), IR (KBr disk): (1632.62) cm-1 (C=N), yield = 74% (Figure 5).

Figure 5: Preparation of (E)-N-(4-fluorophenyl)-1-(1H-indol-34-yl)methanimine
(E)-4-(((1H–indol-3-yl) methylene)amino)–N,N-dimethylaniline (b)
It was prepared by the reaction of Indol-3-carboxyaldehyde (1 g, 6.9 mmol) with N, N-dimethylbenzen-1,4-diamine (0.93 g,6.9 mmol) and (10 drops) of (CH3COOH) glacial acetic acid was added, and then they were refluxed in water bath for 20 hours. The product was precipitated and recrystallized with the addition of methanol droplets, (M.p = 220-222°C), (Rf = 0.9), IR (KBr disk): (1603.65) cm-1 (C=N), yield = 80% (Figure 6).

Figure 6: Preparation of (E)-4-(((1H–indol-3-yl) methylene)amino)–N,N-dimethylaniline
Synthesis of thiazoledinones [11,12]
3-(4-fluorophenyl)-2-(1H-indol-3-yl)thiazolidin-4-one(I)
It was prepared by the reactant (1 g, 4.2 mmol) (E)-N-(4-fluorophenyl)-1-(1H-indol-3-yl)methanimine with (0.39 g, 4.2 mmol) thioglycolic acid acid in (15 ml) chloroform, and then it was refluxed for 18 hours with stirring. The product was precipitated and recrystallized with the addition of ethanol Rf =0.7, yield=70%, M.p=229-231 °C (Figure 7).

Figure 7: Preparation of 3-(4-fluorophenyl)-2-(1H-indol- 3-yl)thiazolidin-4- one
3- (4- (dimethylamino)phenyl)-2-(1H-indol-3-yl)thiazolidin-4-one(II)
It was prepared by reactant (1g, 3.8 mmol) (E)-4-(((1H-indol-3-yl) methylene)amino)-N,N-dimethylaniline with (0.36 g, 3.8 mmol) thioglycolic acid acid in 15 ml chloroform, and then it was refluxed for 18 hours with stirring. The product was precipitated and recrystallized with the addition of ethanol Rf =0.8, yield = 75%, M.p=230-232 °C (Figure 8).

Figure 8: Preparation of 3-(4-(dimethylamino)phenyl)-2-(1H-indol-3-yl)thiazolidin-4-one
Quantum chemical calculations
Gaussian 09 W program was used to calculate EHOMO, ELUMO, energy gap (ΔE), and other parameters of the prepared compounds by implementing density function theory (DFT).
Results and discussion
The mechanism of preparing imines
Imine formation is a reversible process that starts with the nucleophilic addition of a primary amine to the carbonyl group of an aldehyde or ketone. Next, a proton transfer forms a neutral amino alcohol called a carbinolamine. Acid protonation of the carbinolamine oxygen converts it into a better leaving group which is subsequently eliminated as water producing an iminium ion. De-protonation of nitrogen gives the final imine product, as displayed in Scheme 1.

Scheme 1: Mechanism of imines formation
Table 1 shows the melting point measurement for the prepared compounds (a and b) diagnosed by (FT-IR) (Table 2), whose bands correspond to the vibration bands aliphatic (C-H), (aromatic C-H), (C=C)), and (azomethine band C=N). These bands occur (2925, 2923), (3037, 3042, 3097), (1591, 1567), and (1603, 1632), respectively.
Table 1: physical properties of imines
|
Yield% |
Reaction time |
Rf |
Color |
Melting point |
Compound |
|
74 |
16h |
0.8 |
Yellow |
223-225 |
a |
|
80 |
20h |
0.9 |
Yellow |
220-222 |
b |
Table 2: IR spectra of imines
|
Aromatic )C=C( stretching )cm-1( |
Azomethine )C=N( Stretching )cm-1( |
Aliphatic )CH) stretching )cm-1( |
Aromatic )CH( stretching )cm-1( |
Compound |
|
1591 |
1632 |
2925 |
3042 3104 |
a |
|
1567 |
1603 |
2923 |
3037 3097 |
b |
Synthesis of Thiazolidinones
The mechanism of preparing thiazolidinediones includes the reaction of the prepared Schiff bases with thioglycolic acid. The following scheme shows the reaction mechanism involving cycloaddition to form thiazolidinediones [13].

Scheme 2: Mechanism of thiazoldinone formation
FT-IR data
The prepared compounds (I,II) melting point and physical properties for prepared compounds are represented in Table 3. Compounds diagnosed specifying (FT-IR) listed in Table 4, demonstrated the featured packages most notably, C-H aromatic, aromatic C=C, aliphatic C-H, and carbonyl amide group which occur within (3041.48, 3104.91), (1574.91, 1576.47), (2976.37, 2811.03), (1686.24, 1646.81), respectively.
Table 3: Physical properties of thiazoledinones
|
Compound |
Melting point |
Color |
RF |
Reaction time |
Yield% |
|
I |
229-231 |
White |
0.7 |
18 h |
70 |
|
II |
230-232 |
Yellow |
o.8 |
18 h |
75 |
Table 4: FTIR spectral data of thiazolidinones
|
Compound |
Aromatic (C- H) stretching cm-1 |
Aromatic (C= C) stretching cm-1 |
Aliphatic (C- H) stretching cm-1 |
Amide (C= O) stretching cm-1 (thia-) |
(C-N) stretching |
(C-S) Bending cm-1 |
|
I |
3041. 48 3104.91 |
1576.47 |
2976.37 |
1686.24 |
1334.15 |
787.97 |
|
II |
3033.01 3089.35 |
1574.91 |
2972.70 |
1646.81 |
1355.51 |
884.82 |
1H NMR spectral
1H-NMR data of thiazoledinones are indicated in Table 5.
Table 5: 1H-NMR data of thiazolidinones
|
NH Indol |
C2H Indol |
Aliphatic proton |
Aromatic proton |
Thiazolidinone ring |
Compound |
|
|
(C-H2) ring |
(C-H) ring |
|||||
|
11.61 ppm |
11.12 ppm |
--- |
6.72-8.69 ppm |
5.58 ppm |
3.18- 3.41 ppm |
I |
|
12.6 ppm |
11.17 ppm |
3.66 ppm |
7.01-8.70 ppm |
5.21 ppm |
3.40- 3.44 ppm |
II |
Quantum chemical calculations
In computational study of the prepared compounds, Table 6 indicates the calculation of the most important chemical parameters of the prepared compounds.
Whether a molecule is hard or soft, the HOMO-LUMO energy gap represents the chemical reaction of that molecule. Compound 2 is characterized by a small energy gap (ΔE gap=1.17 eV), while compound 1 has a slightly higher energy gap (ΔE gap=1.8 eV). Therefore, compound 2 has a large susceptibility to polarization because its excitation energy is small, so compound 2 is soft [14].
In addition, compound 2 has a low ionization potential IP=4.23 and this indicates a high reactivity of the molecules [15,16].
Low electron affinity values enhanced the electron-donating property of the molecule. Thus, the most donating molecule is compound 2 (EA = 3.06 eV) and the most acceptable molecule is compound 1 (EA = 3.12 eV) [17,18].
The stability and reactivity of the molecule can be known by the factors of hardness (ɳ) and chemical softness (ς) [19-22]. Hence, compound 1(ς =1.11 eV) is the more softness with the lowest hardness. Meanwhile, compound 2 (ς =1.71 eV) is the more hardness and less softness molecule (Figure 10-13).
Table 6: Quantum chemical parameters of the prepared compounds
|
Compound |
HOMO |
LUMO |
Energy gap (Eg) |
Ionization potential (IP) |
Electron affinity (EA) |
Hardness (ɳ) |
Electronegativity (χ) |
Ductility (ς) |
Chemical potential (μ) |
|
1 |
-4.92 |
-3.12 |
1.8 |
4.92 |
3.12 |
0.9 |
4.02 |
1.11 |
-4.02 |
|
2 |
-4.23 |
-3.06 |
1.17 |
4.23 |
3.06 |
0.585 |
3.65 |
1.71 |
-3.65 |

Figure 10: (HOMO) of compound 1

Figure 11: (LUMO) of compound 1
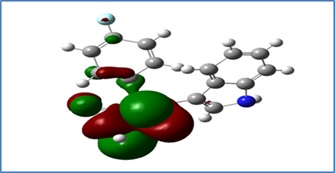
Figure 12: (HOMO) of compound 2

Figure 13: (LUMO) of compound 2
Conclusion
The present study includes the synthesis and characterization of imine compounds, in addition to the synthesis of thiazolidinones from the reaction of the corresponding imine compounds with thioglycolic acid. These compounds were characterized with various spectral methods like (IR) and (1H-NMR). Gaussian program was used to computational study thiazolidinones and the thermodynamic variables were calculated for these compounds.
Acknowledgment
The author thanks the College of Science/ University of Thi-Qar.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Authors' contributions
All authors contributed to data analysis, drafting, and revising of the paper and agreed to be responsible for all the aspects of this work.
Conflict of Interest
The author declared that they have no conflict of interest.
ORCID:
Auhood Kadhim Zaid
https://orcid.org/0000-0002-5912-5372
HOW TO CITE THIS ARTICLE
Auhood Kadhim Zaid. Synthesis, Characterization, and Computational Study of Novel Thiazolidinone Derivatives Containing the Indole. J. Med. Chem. Sci., 2023, 6(2) 346-354


